Abstract
Ordinary Portland Cement (OPC) is a major contributor to global CO2 emissions due to the energy-intensive calcination process required for its production, highlighting the critical need for alternative binder systems that can reduce or replace cement while still meeting the practical applications. The development of this alternative binder opens opportunities for researchers to design solutions for various purposes, not only reducing or replacing cement but also offering additional benefits, such as CO₂ absorption. This research explores the synergistic of a sustainable non-OPC binder system incorporating high volumes of fly ash, limestone powder, gibbsite powder, and biomass ash (BA), presenting a sustainable local alternative for construction materials. By evaluating workability, compressive strength, and CO2 uptake. The results show that low BA content enhances flowability, while higher BA content improves compressive strength (up to 29 MPa at 56 days) by facilitating pozzolanic reactions and the formation of carboaluminate phases. Additionally, the high alkalinity of BA enhanced CO2 sequestration, with the highest absorption (2.7%) observed at 15% BA content. XRD and DTA analysis confirm active pozzolanic reactions, calcium aluminate formations, and the influence of carbonation reactions. These findings highlight the potential of this non-OPC binder for practical applications in environmentally friendly construction, offering reduced reliance on cement while effectively sequestering CO2.
Similar content being viewed by others
Introduction
The construction industry faces increasing pressure to adopt sustainable practices and reduce its environmental footprint, driven in large part by global net-zero emissions targets by 20501. Ordinary Portland Cement (OPC), as a major in construction materials, is responsible for approximately 7% of global CO2 emissions due to the energy-intensive calcination process required for its production2. This has created a critical need for alternative binder systems that can reduce or replace cement usage while still meeting the performance demands of structural applications. Recent research3 has explored non-ordinary portland cement (OPC) alternatives that consume less energy during production, emit lower levels of CO2, and offer greater sustainability. In Thailand, non-OPC development has been the subject of in-depth research. Jaturapitakkul and Roongreung4 investigated the potential of a supplementary cementitious material (SCMs) derived from calcium carbide residue and rice husk ash without using Portland cement. These materials primarily contain Ca(OH)2 and SiO2, respectively. The cementing properties were attributed to the pozzolanic reaction between them, enabling the samples to achieve a maximum compressive strength of 15.6 MPa at 28 days, which further increased to 19.1 MPa at 180 days. This strength level makes the material suitable for practical applications that do not require high compressive strength. According to the study, supplementary cementitious materials (SCMs) play a crucial role in replacing OPC completely. Various SCMs are sourced from industrial by-products such as silica fume, bottom ash, granulated blast-furnace slag, and limestone powder due to their magnitude, compatibility, and relatively low cost5. Among these, fly ash stands out as one of the most abundant SCMs, particularly in Northern Thailand, where coal-fired power plants generate significant quantities of this material. Due to its pozzolanic reactivity and self-cementing properties, high calcium fly ash serves as a key component to replace OPC in binder systems.
Over the last 10 years, extensive research has been conducted on replacing cement with fly ash to create binders that match or improve the properties of OPC. This has resulted in the development of high-volume fly ash binder, with cement replacement levels ranging from 50 to 80%6. Bhatt et al.7 reviewed the high calcium fly ash, which is classified as class C fly ash from ASTM standard, provides self-cementing properties, where its reactive silica (SiO2) and alumina (Al2O3) components react with calcium hydroxide (Ca(OH)2) released from pozzolanic activity to form calcium silicate hydrate (C–S–H) and calcium aluminate hydrate (C-A-H). This suggests that high-volume fly ash with a high calcium content has the potential to fully replace OPC, making it a viable non-OPC binder. However, to completely replace the OPC binder by increasing the amount of fly ash, a larger proportion of calcium is consumed to form additional calcium silicate hydrate (C–S–H) and calcium aluminate hydrate (C–A–H). To compensate for OPC binder reactivity, limestone powder is incorporated as a calcium source, which provides calcium carbonate (CaCO3) to react with the calcium aluminate hydrates (C–A–H) to form calcium carbo-aluminate hydrates through the additional pozzolanic reaction. This synergy was carried out by De Weerdt et al.8, which demonstrates the effect of ternary composite cement incorporating limestone powder and fly ash. The results showed that fly ash/limestone blended cement, due to the additional alumina provided by the fly ash reaction, promoted the formation of hemi- and monocarboaluminate, resulting in stabilization of ettringite compared to the limestone-free cement, which led to an increase of the volume of the hydrate products9. Once the silicates are depleted from the reaction between fly ash and the continuous supply of calcium from limestone powder, the pathway shifts toward the formation of more aluminate-rich phases, causing the increased alkalinity of the system and creating an opportunity to enhance the formation of carbonation products.
Biomass waste from agro-industrial residues such as rice husk ash, corn cob ash, wheat straw ash, sugarcane bagasse ash, and sugarcane filter cake ash, with the incorporation of SCMs, not only contribute to strength development but also helps to mitigate environmental pollution by enhancing CO2 absorption during the carbonation process. Lei and Pavia10 investigate the use of olive pit bottom ash (OBA) as biomass waste for producing carbon-negative cement. By mixing OBA with waste ground granulated blast furnace slag (GGBS) without calcination or thermal curing, the resulting cement can incorporate up to 60% OBA, achieving an impressive carbon sequestration capacity of up to -97.45 kg CO2 e/m3. The K₂O and CaO content in OBA provide significant activation and alkalinity, facilitating the release of Ca2+, Si4+, and Al3+ from GGBS. The high alkalinity of these materials is particularly valuable, as it promotes the absorption and conversion of CO2 into stable carbonates within the concrete matrix. Therefore, the use of alkaline waste as CO2 absorbents involves leveraging waste materials with high pH and the ability to react with CO2 through chemical reactions11.
By fully replacing cement with a non-OPC binder derived from SCMs while incorporating CO2 adsorption capability, this mixture offers a sustainable alternative binder system that actively captures CO2 and significantly helps reduce global emissions. This study aims to develop and evaluate a non-OPC binder mixture utilizing various local industrial and agricultural wastes, including high volumes of fly ash, and limestone powder. Additionally, gibbsite powder is introduced to activate the aluminate phase12 and biomass ash, a key component of the potential to produce non-OPC binder with an emphasis on CO2 sequestration. The primary objective is to demonstrate the practicality of this binder by assessing key properties such as workability and compressive strength, supported by reaction analysis using X-ray Diffraction (XRD). Additionally, the research evaluates the CO2 sequestration potential of the mixture through CO2 uptake measurements obtained from Differential Thermal Analysis (DTA). By developing the synergies of these material properties and showcasing their ability to sequester CO2, this study seeks to establish a sustainable binder that offers an eco-friendly for construction practices.
Materials
The materials used in this study include Ordinary Portland Cement (OPC) Type I, a class C ASTM fly ash (FA), Biomass ash (BA), limestone powder (CC), and gibbsite powder (GP). The chemical composition of each material determined by XRF is given in Table 1. Fly ash is obtained from the Mae Moh power plant, in Thailand and then grounded by a laboratory ball mill to have an average particle size of 10 microns. Biomass ash (BA) or corn cob ash is a by-product of corn cobs from animal feed production plants in Thailand. The ground cobs were burnt at 600 °C with a duration of 2–4 h and subsequently milled to an average particle size of 70 microns. The main chemical components of BA include potassium oxide (K2O), calcium oxide (CaO), and silicon dioxide (SiO2). Limestone powder (CC), which was used as a calcium source in this binder, is an industrial-quality grade that has been reduced by a laboratory ball mill to a size of less than 10 microns. Gibbsite powder (GP) also has a particle size of less than 10 microns with the primary component of aluminum oxide (Al2O3), which is present in the form of aluminum hydroxide (Al(OH)3).
Mix design
The study consists of nine different mixtures shown in Table 2, including a control sample from ordinary Portland cement (OPC) type I. The experimental mixtures incorporated varying proportions of fly ash (FA) as the primary replacement binder for OPC, combined with three different percentages of limestone powder (CC) from 10%, 20%, and 30% and biomass ash (BA) from 5%, 10%, and 15%. Additionally, Gibbsite powder (GP) was added as an alkaline activator to promote the formation of aluminate phases13, which is typically formed during the hydration reaction of OPC as AFm14, by ensuring that sufficient alumina (Al2O3) is available to facilitate the C3A to react with calcium hydroxide (CH) and carbonate ions (CO32−) from limestone powder (CC), facilitating the formation of carboaluminate hydrates, as shown in Eqs. 1 and 2.15.
Monocarboaluminate:
Hemicarboaluminate:
Workability and compressive strength testing methods
The testing involved the evaluation of both workability and compressive strength of binder. For workability, a flow table test16 was used to measure the spread diameter of the mixture after being tamped and dropped 25 times, following an ASTM C1437 standard. The setting time was assessed according to ASTM C19117, where the initial and final setting times were determined by measuring the penetration of a Vicat needle into the paste. For hardened properties, compressive strength tests were conducted on 5 × 5 × 5 cm cube specimens according to ASTM C10918, with samples cured at ambient temperature and tested at 3, 7, 28, and 56 days to evaluate their strength development over time. Confirm the strength results by using X-ray diffraction (XRD) to evaluate the reactivity of materials.
CO2 absorption
After 24 h of curing, the carbonation test was conducted in a chamber with a CO2 concentration of 100%. The pastes were carbonated at room temperature, and their CO2 absorption capacity was evaluated through differential thermal analysis (DTA). CO2 uptake represents the percentage of CO2 absorbed during forced carbonation, relative to the initial mass of the cement19. This is determined by comparing the CaCO3 content of exposed and non-exposed samples, as obtained from DTA results. First, the CaCO3 content of each sample is calculated using Eq. 3. Subsequently, CO2 uptake after exposure is determined using Eq. 4, based on the methodology outlined in20.
where:
-
CaCO3 refers to calcium carbonate in (g/100 g).where:
-
100.1/44.0 is the molar weight ratio of CaCO3 to CO2.where:
-
Mc is the cement’s initial mass (g) in the TGA sample.where:
-
\({\text{M}}_{\text{start}}^{\text{CaCO3}}\) and \({\text{M}}_{\text{end}}^{\text{CaCO3}}\) are the masses (g) of the sample at the start and endpoint for CaCO3 decomposition, respectively.
$${\text{CO}}_{{2}} \left( {{\text{g}}/{1}00{\text{g}}} \right) = \left[ {\left( {{\text{CaCO}}_{{3}}^{{{\text{exp}}{\text{.smaple}}}} } \right){ } - {\text{(CaCO}}_{{3}}^{{{\text{nonexp}}{\text{.smaple}}}} {)}} \right]\cdot\frac{{{44}{\text{.0}}}}{{{100}{\text{.1}}}}$$(4)where:
-
CO2 uptake is the amount of carbon dioxide in g/100 g.
-
44.0/100.1 is the molar weight ratio of CO2 to CaCO3.
-
\({{\text{CaC}}{\text{O}}_{3}}^{\text{exp.smaple}}\) and \({{\text{CaC}}{\text{O}}_{3}}^{\text{nonexp.smaple}}\) are the calcium carbonate contents of the exposed and non-exposed samples, respectively. These values are obtained by obtained by Eq. 3
Results and discussion
Workability
Flow table test
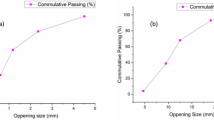
Figure 1 presents the results of the Flow Table test, indicating the flow value decreased as the percentage of biomass ash (BA) increased. Specifically, for samples CC20BA5, CC20BA10, and CC20BA15, the flow values dropped from 91.33% to 73.02% and 37.81%, respectively. Similarly, for CC15BA5, CC15BA10, and CC15BA15, the flow values were 100.00% to 50.59% and 35.61%, respectively. Lastly, the flow values for CC10BA5, CC10BA10, and CC10BA15 were 100.25%, 62.35%, and 43.60%, respectively. Generally, an increase in fly ash (FA) content enhances flowability due to the ball-bearing effect created by the spherical shapes of the fly ash particles21. However, in this study, the reduced flowability may be attributed to the corn cob ash, which absorbs more water than cement, resulting in lower slump values for fresh concrete compared to standard fresh concrete slump values22. Adesanya23 and Olafusi24 also noted that the decrease in slump values was linked to the larger size of irregularly shaped particles.
Setting time
Table 3 presents the test results for the initial and final setting times of control cement paste samples and non-OPC binder including CC20BA5, CC20BA10, CC20BA15, CC15BA5, CC15BA10, CC15BA15, CC10BA5, CC10BA10, and CC10BA15. The initial setting times were found to be 97, 142, 65, 132, 271, 96, 123, 116, and 42 min, respectively, while the final setting times were 175, 233, 141, 205, 420, 210, 180, 195, and 135 min, respectively. It was observed that the BA content increased from 5 to 10%, the setting time increased, whereas further increasing BA to 15% resulted in a shorter setting time with both initial and final stages. This can be attributed to the imbalance between FA and BA content. Both materials contain reactive silica (SiO2) and calcium oxide (CaO), but BA is primarily composed of potassium oxide (K2O). Since the pozzolanic reactions in the binder mainly rely on FA dissolution, the decrease in FA and the increase in BA interrupt the dissolution of FA, leading to a slower pozzolanic reaction rate and extending the setting time. However, at 15% BA, the balance is restored, as the alkalis (K2O) enhances the dissolution of reactive components, creating an environment for continued pozzolanic reactivity between the SiO2 and CaO from both FA and BA25. This behavior was explained by Laskar and Talukdar26, who developed the geopolymer mortars prepared from ultrafine ground granulated blast-furnace slag incorporated with fly ash and alkali activators composed of (1) sodium silicate (SS) and sodium hydroxide (SH), and (2) only a sodium hydroxide (SH) solution. The results indicated that the setting times of mixes containing only SH as an alkali activator were longer than those containing SS and SH in combination as an alkali activator for the mixes containing a high amount of FA.
Compressive strength
The compressive strength test results, presented in Table 4 demonstrate the performance of samples CONTROL, CC20BA5, CC20BA10, CC20BA15, CC15BA5, CC15BA10, CC15BA15, CC10BA5, CC10BA10, and CC10BA15 at 3, 7, 28, and 56 days. The control samples exhibited the highest compressive strength at all ages compared to the other samples, which is typical due to the reduced clinker content limiting hydration reactions. However, pozzolanic activity still occurred, contributing to the compressive strength development, even though their strength levels remained significantly lower than the CONTROL sample.
Figure 2, derived from Table 4, provides clearer results. Most samples exhibited limited strength gain beyond a certain point. For instance, CC20BA5 demonstrated an early increase in compressive strength, reaching 15.35 MPa at 3 days, 20.54 MPa at 7 days, 23.73 MPa at 28 days, and 24 MPa at 56 days, which showed minimal further development. This is attributed to the high limestone powder (CC) content, which primarily acts as a filler rather than actively contributing to strength through the pozzolanic reaction of fly ash (FA). This effect is further evidenced by the slower strength progression in CC20BA15, which recorded the lowest strength development, increasing from 5.48 MPa at 3 days to only 13 MPa at 56 days. However, a distinct trend is found in mixtures containing BA10. CC20BA10 exhibited significant growth, increasing from 5.76 MPa at 3 days to 25.5 MPa at 56 days. Similarly, CC15BA10 improved from 4.52 MPa at 3 days to 28 MPa at 56 days. Notably, CC10BA10 achieved the highest compressive strength among the non-OPC binders, increasing from 4.7 MPa at 3 days to 29 MPa at 56 days. These results highlight the importance of achieving an optimal balance between pozzolanic activity and the reaction of limestone powder with aluminate hydrates to form carboaluminate phases, contributing to the strength development.
These results align with the findings of Ibrahim, et al.27, which studied the effect of alkaline activators and binder content of natural pozzolan-based alkali-activated concrete. The results show that the compressive strength development is significantly influenced by binder content and the sodium silicate-to-sodium hydroxide (SS/SH) ratio. Optimal strength gain occurs when sufficient alkaline activators promote the dissolution of reactive components. However, excessive binder content or extended curing may negatively impact strength due to microstructural instability or unreacted particles. This suggests the need for a balanced binder content to achieve the best results.
X-ray diffraction (XRD)
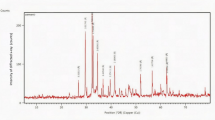
Figure 3 shows the XRD results of CC20BA10, CC15BA10, and CC10BA10 at 3, 7, 28, and 56 days which show the significance of the compressive strength results. CC (CaCO3), Gb (Al(OH)2), An (CaSO4), and Sy (K2Ca(SO4)2·H2O) are the significant mineral compositions found in the samples. The broader intensity of the CC peak and the ongoing development of compressive strength over curing age indicate active pozzolanic reactions despite the absence of portlandite (Ca(OH)₂) in the XRD results. Furthermore, carbonation reactions increase the intensity of CC peak, as portlandite reacts with CO₂ during curing, as illustrated in Eq. 3.28 At curing ages of 28 and 56 days, the hydrocalumite (Hy) phase, a layered-double hydroxide (LDH) material acting as a catalyst and CO2 capture agent, forms due to the alkaline activation of reactive alumino-silicate-based materials from the assistance of the gibbsite activator. The lamellar structure of hydrocalumite is similar to portlandite layers (Ca(OH)2), with part of the calcium being replaced by aluminum29,30,31, as demonstrated in Eq. 4. Similarly, the intensity of the Gb peak increases over the curing period, indicating the ongoing hydraulic reactions of calcium aluminates formed during initial hydration. These reactions gradually convert the calcium aluminate components into the stable garnet phase C₃AH₆ and gibbsite32.
CO2 absorption
Figure 4 shows the DTA results of the CC20BA15, CC15BA15, and CC10BA15 after 24 h of CO2 exposure. The temperature range of 200–300 °C corresponds to the decomposition of gibbsite (Al2O3·3H2O)33, while the temperature range of 600–750 °C shows the decomposition of calcite (CaCO3)34. The results show a distinguished increase of CaCO3 at 2%, 2.7%, and 2.6% for CC20BA15, CC15BA15, and CC10BA15 respectively, which is the highest BA content among all the samples. The chemical composition of BA is mainly K2O and CaO, which potentially influence the carbonation of cement-based materials since the carbonation reaction in cement binder is known to occur within capillary pores with a pore solution at high pH35. In this research, BA likely enhances the reactivity of Ca2+ and CO2 from external sources, allowing them to enter and dissolve to form CaCO3 in the pore solution36.
Figure 5 shows the CO2 uptake after 24 h of carbonation test as a function of biomass ash (BA) and limestone powder (CC). The result confirms that increasing BA content leads to higher CO2 uptake, reaching up to 2.7% with the optimum percentage of BA of 15% which is due to the high alkalinity of BA. Furthermore, gibbsite in the non-OPC binder transforms into more stable calcium aluminate phases37, indirectly enhancing CO2 penetration by increasing porosity during carbonation. This result aligns with the study of Moro et al.38 that examines the impact of nano-TiO2 on CO2 sequestration in hardened cement paste, considering the water-to-cement ratio (w/c) and pore structure, samples with higher w/c ratios exhibit greater porosity, and the decrease in porosity due to nano-TiO2 does not notably restrict the space for carbonation. Consequently, the enhanced reactivity of calcium hydroxide (CH) from nano-TiO2 results in a greater CO2 uptake in these samples.
The CC15 and CC20 sample groups exhibited a wider green zone of the contour compared to the CC10 group, indicating their suitability as CO₂ absorption mixtures, with at least 1% CO₂ captured. This suggests the biomass ash (BA) and limestone powder (CC) content in these mixtures affect their carbonation potential, likely due to the synergistic effects of high alkalinity, increased porosity, and reactive phases among the mixed materials.
Conclusion
This study highlights the synergistic benefits of incorporating high volumes of fly ash (FA), limestone powder (CC), biomass ash (BA), and gibbsite powder (GP) in non-OPC binder, offering a sustainable alternative for construction applications to create an environmentally friendly material. The key contributions are summarized as follows:
-
Samples with low biomass ash (BA) content exhibit high flowability due to the majority of fly ash (FA) in the binder. However, as BA content increases, the flowability decreases, likely due to its irregular particle shape causing higher water absorption compared to FA. Additionally, BA affects FA dissolution due to its alkalinity, initially extending the setting time. At higher BA levels (15%), the alkali content (K2O) enhances the dissolution of reactive silica and calcium oxide, restoring pozzolanic reactivity and reducing setting time.
-
BA also influences compressive strength, with these samples (CC20BA5, CC20BA15), limestone powder (CC) primarily acts as a filler rather than actively contributing to pozzolanic reactions. Samples with 10% BA (CC20BA10, CC15BA10, CC10BA10) demonstrated significant strength improvements, reaching 25.5–29 MPa at 56 days. This is due to the balance between pozzolanic activity and the formation of carboaluminate phases, which played a crucial role in strength development.
-
XRD result confirms the interactions of the material at a microstructure level. The broader intensity of the calcite (Cc) peak with the continuous compressive strength increases and the formation of the hydrocalumite (Hy) and gibbsite (Gb) peaks confirm the pozzolanic reaction and calcium aluminates are actively reacting.
-
CO2 absorption results confirm that increasing biomass ash (BA) content enhances CO2 uptake, with the highest absorption observed at 2.7% in CC15BA15. The decomposition of calcite (CaCO3) indicates that carbonation occurs significantly in mixtures with high BA content. This is attributed to the high alkalinity of BA (rich in K2O and CaO), which facilitates the dissolution of Ca2⁺ and its reaction with CO2 to form stable carbonates. Additionally, gibbsite (Al2O3·3H2O) transitions into stable calcium aluminate phases during carbonation, indirectly increasing CO2 penetration by modifying the pore structure.
These findings highlight the multiple advantages of moderate workability properties, compressive strength development, and active carbon sequestration, making these binders ideal for applications such as pavements, precast elements, and low- to moderate-strength structures. To enhance the non-OPC binder, further research is needed to optimize the composition of alternative binders, balancing the workability, strength, durability, and carbon sequestration potential using advanced characterization techniques. This could facilitate the development of sustainable alternatives to portland cement. Additionally, a deeper understanding of the carbonation reaction between various non-cementitious components could significantly advance the non-OPC negative carbon construction materials.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Liang, Y., Kleijn, R. & van der Voet, E. Increase in demand for critical materials under IEA Net-Zero emission by 2050 scenario. Appl. Energy 346, 121400. https://doi.org/10.1016/j.apenergy.2023.121400 (2023).
Chaudhury, R., Sharma, U., Thapliyal, P. C. & Singh, L. P. Low-CO2 emission strategies to achieve net zero target in cement sector. J. Clean. Prod. 417, 137466. https://doi.org/10.1016/j.jclepro.2023.137466 (2023).
Sharma, R., Kim, H., Lee, N. K., Park, J.-J. & Jang, J. G. Microstructural characteristics and CO2 uptake of calcium sulfoaluminate cement by carbonation curing at different water-to-cement ratios. Cem. Concrete Res. https://doi.org/10.1016/j.cemconres.2022.107012 (2023).
Jaturapitakkul, C. & Roongreung, B. Cementing material from calcium carbide residue-rice husk ash. J. Mater. Civil Eng. 15, 470–475. https://doi.org/10.1061/(ASCE)0899-1561(2003)15:5(470) (2003).
Orozco, C., Tangtermsirikul, S., Sugiyama, T. & Babel, S. Examining the endpoint impacts, challenges, and opportunities of fly ash utilization for sustainable concrete construction. Sci Rep 13, 18254. https://doi.org/10.1038/s41598-023-45632-z (2023).
Krishnya, S. et al. Modeling of hydration products and strength development for high-volume fly ash binders. Constr. Build. Mater. 320, 126228. https://doi.org/10.1016/j.conbuildmat.2021.126228 (2022).
Bhatt, A. et al. Physical, chemical, and geotechnical properties of coal fly ash: A global review. Case Stud. Constr. Mater. 11, e00263. https://doi.org/10.1016/j.cscm.2019.e00263 (2019).
De Weerdt, K., Kjellsen, K. O., Sellevold, E. & Justnes, H. Synergy between fly ash and limestone powder in ternary cements. Cem. Concrete Compos. 33, 30–38. https://doi.org/10.1016/j.cemconcomp.2010.09.006 (2011).
Thongsanitgarn, P., Wongkeo, W., Chaipanich, A. & Poon, C. S. Heat of hydration of Portland high-calcium fly ash cement incorporating limestone powder: Effect of limestone particle size. Constr. Build. Mater. 66, 410–417. https://doi.org/10.1016/j.conbuildmat.2014.05.060 (2014).
Lei, Z. & Pavia, S. Biomass ash (BA) waste as an activator to produce carbon-negative cement. Cement https://doi.org/10.1016/j.cement.2024.100112 (2024).
Pan, S.-Y., Chang, E. E. & Chiang, P.-C. CO2 capture by accelerated carbonation of alkaline wastes: A review on its principles and applications. Aerosol Air Quality Res. 12, 770–791. https://doi.org/10.4209/aaqr.2012.06.0149 (2012).
Wang, Y. et al. Alumina-rich pozzolan modification on Portland-limestone cement concrete: Hydration kinetics, formation of hydrates and long-term performance evolution. Constr. Build. Mater. 258, 119712. https://doi.org/10.1016/j.conbuildmat.2020.119712 (2020).
Lin, Y., Alengaram, U. J. & Ibrahim, Z. Effect of treated and untreated rice husk ash, palm oil fuel ash, and sugarcane bagasse ash on the mechanical, durability, and microstructure characteristics of blended concrete: A comprehensive review. J. Build. Eng. https://doi.org/10.1016/j.jobe.2023.107500 (2023).
Ipavec, A. et al. Carboaluminate phases formation during the hydration of calcite-containing Portland cement. J. Am. Ceram. Soc. 94, 1238–1242. https://doi.org/10.1111/j.1551-2916.2010.04201.x (2010).
Bentz, D. P., Stutzman, P. E. & Zunino, F. Low-temperature curing strength enhancement in cement-based materials containing limestone powder. Mater. Struct. 50, 173. https://doi.org/10.1617/s11527-017-1042-6 (2017).
in Standard Test Method for Flow of Hydraulic Cement Mortar (ASTM International, West Conshohocken, PA, 2020).
in Standard Test Methods for Time of Setting of Hydraulic Cement by Vicat Needle (ASTM International, West Conshohocken, PA, 2021).
in Standard Test Method for Compressive Strength of Hydraulic Cement Mortars (Using 2-in. or [50-mm] Cube Specimens) (ASTM International, West Conshohocken, PA, 2020).
Zhuge, Y., Ong, P. B., Wong, H. S. & Myers, R. J. Natural fibre-enhanced CO2 transport and uptake in cement pastes subjected to enforced carbonation. J. CO2 Util. https://doi.org/10.1016/j.jcou.2024.102983 (2024).
Lopez-Arias, M., Moro, C., Francioso, V., Elgaali, H. H. & Velay-Lizancos, M. Effect of nanomodification of cement pastes on the CO2 uptake rate. Constr. Build. Mater. https://doi.org/10.1016/j.conbuildmat.2023.133165 (2023).
Kondraivendhan, B. & Bhattacharjee, B. Flow behavior and strength for fly ash blended cement paste and mortar. Int. J. Sustain. Built Environ. 4, 270–277. https://doi.org/10.1016/j.ijsbe.2015.09.001 (2015).
Ahmad, J., Arbili, M. M., Alabduljabbar, H. & Deifalla, A. F. Concrete made with partially substitution corn cob ash: A review. Case Stud. Constr. Mater. https://doi.org/10.1016/j.cscm.2023.e02100 (2023).
Adesanya, D. A. & Raheem, A. A. A study of the workability and compressive strength characteristics of corn cob ash blended cement concrete. Constr. Build. Mater. 23, 311–317. https://doi.org/10.1016/j.conbuildmat.2007.12.004 (2009).
Olafusi, O., Adewuyi, A. P., Sadiq, O., Adisa, A. & Abiola, O. Rheological and mechanical characteristics of self-compacting concrete containing corncob ash (2017).
Akmalaiuly, K., Berdikul, N., Pundienė, I. & Pranckevičienė, J. The effect of mechanical activation of fly ash on cement-based materials hydration and hardened state properties. Materials (Basel) https://doi.org/10.3390/ma16082959 (2023).
Laskar, S. M. & Talukdar, S. Development of ultrafine slag-based geopolymer mortar for use as repairing mortar. J. Mater. Civ. Eng. 29, 04016292. https://doi.org/10.1061/(ASCE)MT.1943-5533.0001824 (2017).
Ibrahim, M., Megat Johari, M. A., Rahman, M. K. & Maslehuddin, M. Effect of alkaline activators and binder content on the properties of natural pozzolan-based alkali activated concrete. Constr. Build. Mater. 147, 648–660. https://doi.org/10.1016/j.conbuildmat.2017.04.163 (2017).
Tang, Y. et al. Re-cementation effects by carbonation and the pozzolanic reaction on LWAs produced by hydrated cement paste powder. J. Clean. Prod. 377, 55. https://doi.org/10.1016/j.jclepro.2022.134529 (2022).
Abd-El-Raoof, F., Tawfik, A., Komarneni, S. & Ahmed, S. E. Hydrotalcite and hydrocalumite as resources from waste materials of concrete aggregate and Al-dross by microwave-hydrothermal process. Constr. Build. Mater. 207, 10–16. https://doi.org/10.1016/j.conbuildmat.2019.02.105 (2019).
Abd-El-Raoof, F., Youssef, H. F., El-Sokkary, T. M., Abd El-Shakour, Z. A. & Tawfik, A. Fabrication and characterization of calcium aluminates cement via microwave-hydrothermal route: Mayenite, katoite, and hydrocalumite. Constr. Build. Mater. https://doi.org/10.1016/j.conbuildmat.2023.132988 (2023).
Rossi, T. M., Campos, J. C. & Souza, M. M. V. M. An evaluation of calcined hydrocalumite as carbon dioxide adsorbent using thermogravimetric analysis. Appl. Clay Sci. https://doi.org/10.1016/j.clay.2019.105252 (2019).
Christensen, A. N. et al. Thermal decomposition of monocalcium aluminate decahydrate (CaAl2O4.10H2O) investigated by in-situ synchrotron X-ray powder diffraction, thermal analysis and 27Al, 2H MAS NMR spectroscopy. Dalton Trans https://doi.org/10.1039/b712684k (2008).
Kloprogge, J. T., Ruan, H. D. & Frost, R. L. Thermal decomposition of bauxite minerals: infrared emission spectroscopy of gibbsite, boehmite and diaspore. J. Mater. Sci. 37, 1121–1129. https://doi.org/10.1023/A:1014303119055 (2002).
Lu, B., Shi, C., Zhang, J. & Wang, J. Effects of carbonated hardened cement paste powder on hydration and microstructure of Portland cement. Constr. Build. Mater. 186, 699–708. https://doi.org/10.1016/j.conbuildmat.2018.07.159 (2018).
Jang, J. G., Kim, G. M., Kim, H. J. & Lee, H. K. Review on recent advances in CO2 utilization and sequestration technologies in cement-based materials. Constr. Build. Mater. 127, 762–773. https://doi.org/10.1016/j.conbuildmat.2016.10.017 (2016).
Mohammed, T., Torres, A., Aguayo, F. & Okechi, I. K. Evaluating carbonation resistance and microstructural behaviors of calcium sulfoaluminate cement concrete incorporating fly ash and limestone powder. Constr. Build. Mater. https://doi.org/10.1016/j.conbuildmat.2024.137551 (2024).
Hidalgo, A., Petit, S., García, J. L., Alonso, C. & Andrade, C. in Studies in Surface Science and Catalysis Vol. 170 (eds Ruren Xu, Zi Gao, Jiesheng Chen, & Wenfu Yan) 1617–1628 (Elsevier, 2007).
Moro, C., Francioso, V. & Velay-Lizancos, M. Modification of CO2 capture and pore structure of hardened cement paste made with nano-TiO2 addition: Influence of water-to-cement ratio and CO2 exposure age. Constr. Build. Mater. https://doi.org/10.1016/j.conbuildmat.2020.122131 (2021).
Acknowledgements
The authors gratefully acknowledge the financial support of Chiang Mai University in supporting the practical work and facilities in this paper.
Author information
Authors and Affiliations
Contributions
P.W.: Conceptualization, Formal analysis, Methodology, Data curation, Writing-original draft, Writing-review and editing, Resources, Supervision, Project administration. K.K.: Methodology, Investigation, Writing-original draft, Writing-review and editing S.C.: Methodology, Investigation, Writing-review and editing T.K.: Methodology, Investigation, Writing-review and editing. K.P.: Conceptualization, Formal analysis, Methodology, Resources, Validation, Data curation, Supervision
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wattanachai, P., Kochchapong, K., Chaiwithee, S. et al. Development of non-OPC binder using fly ash, limestone powder, gibbsite powder, and biomass ash for workability, strength, and CO2 capture. Sci Rep 15, 11098 (2025). https://doi.org/10.1038/s41598-025-95482-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-95482-0
Keywords
This article is cited by
-
Optimizing calcium efficiency for sustainable cement with GGBFS-fly ash systems
Scientific Reports (2025)