Abstract
Open-label placebos (OLPs), which are transparently administered inert treatments, have gained increasing attention in the research community. They have the potential to harness placebo effects without deceiving the patient. The purpose of this study was to examine and synthesize prior research discussing ethical issues in OLPs. A systematic qualitative literature review was conducted (last search on January 2023) across multiple databases (e.g., MEDLINE, Embase, PsycInfo) using keywords related to OLPs. Publications were screened and assessed for eligibility. Seventeen articles explicitly addressing OLP ethics were included. Data were extracted and thematically analyzed following established qualitative content analysis procedures. 37 distinct ethical issues were identified, grouped into five overarching themes: (1) Sociocultural and Contextual Factors, (2) Implementation and Logistics, (3) Informed Consent, (4) Patient Health Behavior and Dynamics, and (5) Therapeutic Relationship. Frequently mentioned concerns focused on uncertainty of OLP efficacy, the need for further research, and complexities of dose-extending OLPs. The effective integration of OLPs into healthcare necessitates thoughtful consideration and responsible communication. This analysis shows that demonstrating the efficacy of OLPs becomes pivotal for their widespread adoption, underscoring the necessity for additional research to validate their effectiveness. This review was pre-registered on January 26th, 2023, in the Open Science Framework Registry under the identifier: DOI https://doi.org/10.17605/OSF.IO/R6NKG.
Similar content being viewed by others
Introduction
The use of placebos in clinical practice has traditionally been a subject of ethical controversy due to several reasons: It often involves deception, conflicting with the ethical principle of informed consent and violating patient autonomy by not allowing informed decision-making1,2. Furthermore, administering a placebo without the patient’s knowledge violates their autonomy by not allowing them to make an informed decision about their treatment. This can be seen as paternalistic and undermines respect for the patient’s right to self-determination3. Additionally, placebos can prevent patients from receiving effective treatments, leading to potential harm4. Their use may compromise the integrity of the medical profession and erodes patient trust5,6. The efficacy of placebos is also highly variable, making them unreliable as a standard treatment7. The use of placebos without consent may have legal implications, potentially constituting malpractice or fraud8. Historically, placebos were administered covertly9,10, with patients unaware of the inert nature of the treatment. In recent years, open-label placebos (OLPs) have gained prominence as an innovative approach for medical treatment, effectively addressing a fundamental concern associated with traditional placebos—the element of deception. Unlike traditional placebos, OLPs are administered transparently, with patients knowingly receiving placebos. This approach leverages the psychological effects of belief, expectation, and hope in healing while avoiding deception. Several studies have shown that OLPs can still improve symptoms and promote well-being11,12,13,14,15,16,17. However, significant obstacles to the methodological integrity of OLP research have been identified in a number of other recent articles18,19,20,21,22. The effectiveness of OLPs in various clinical contexts and the psychological mechanisms underlying their effects are subjects of extensive inquiry23.
OLPs bring forth the question of whether they represent a mere adjunct to conventional treatment, potentially reducing the need for pharmacological interventions24,25,26,27. Conversely, some studies argue that open-label placebos might serve as an important complementary therapeutic tool, particularly in conditions with a strong psychological component28,29. The question of whether OLPs have the potential to revolutionize healthcare by harnessing the mind’s ability to heal30 is a topic of ongoing interest.
A substantial body of research has discussed the ethical implications of open-label placebos1,26,27,31,32,33,34,35,36,37,38,39,40,41,42,43,44, . Nonetheless, there is currently no comprehensive overview available, and many researchers may not be fully cognizant of the potential ethical concerns that arise with OLPs.
Ethical considerations surrounding OLPs encompass a wide spectrum of topics, such as the necessity of unambiguous informed consent27,39,43,45 and the impact of OLPs on patient-doctor relationships and autonomy1,27,34,38,41,46,47. The question of whether open-label placebos are ethically sound for specific medical conditions, like (chronic) pain11,39,40,46,48,49 and irritable bowel syndrome50,51,52, remains an area of active debate. An empirical literature review provides concrete evidence and real-world examples to support the conceptual exploration of ethical concerns surrounding open-label placebo usage. Our primary objective was to explore which ethical principles and concerns have been identified and discussed in the context of OLPs-.
Methods
Study design and rationale
We conducted a systematic qualitative review to synthesize ethical concerns specific to OLPs. This approach was selected because much of the OLP ethics literature is conceptual or theoretical, and the empirical studies available lack sufficient homogeneity for quantitative pooling. Hence, a meta-analysis was not feasible. A systematic qualitative review allows for an in-depth exploration of heterogeneous evidence—ranging from opinion pieces and conceptual analyses to empirical research—focusing on emergent ethical themes53,54.
This review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines55, adapted where necessary for qualitative evidence56. Where relevant, we also drew on previous systematic ethics review approaches for identifying and synthesizing conceptual issues in biomedical literature57,58,59,60.
Search strategy; databases and search terms
On January 5th, 2023, we searched multiple databases via the Ovid platform—including MEDLINE (1946–2023), Embase (1974–2023), PsycInfo (1806–2022), and additional databases listed in Table 2—using the following abstract keywords: (“open label placebo*” OR “open-label placebo*” OR “non-deceptive placebo*” OR “non deceptive placebo*”).
We replicated the same search strategy on PubMed to account for variations in indexing and platform-specific algorithms61. Table 1 details the sources and date cutoffs.
All search results from Ovid (n = 539) and PubMed (n = 187) were exported into Covidence62 for screening and deduplication.
Eligibility criteria; inclusion criteria
153 publications were eligible because they explicitly addressed an ethical concern, conflict, or controversy arising from OLP use. Articles needed to either:
-
Discuss OLP in the context of at least one core ethical principle (e.g., autonomy, beneficence, non-maleficence, or justice), or.
-
Discuss whether OLPs presented a clash between multiple ethical principles.
We employed principlism63 as our overarching ethical framework, consistent with prior systematic ethics reviews57,58,59,60. Only explicitly stated ethical arguments or reflections were included; implicit or “between the lines” inferences were excluded.
Eligibility criteria; exclusion criteria
136 publications were excluded because of three reasons: there was no ethical dilemma discussed in the paper that concerned OLPs (114 excluded), there were only abstracts available (16 exclusions) or there were not OLPs applied at all (wording in the article might have been similar but not the same topic, 6 exclusions).
Screening and selection process
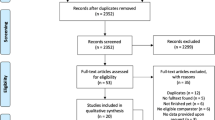
After Covidence removed duplicates (n = 416 total across both Ovid and PubMed results), 279 unique abstracts remained. One reviewer (M.R) screened all abstracts (n = 279) for relevance based on predefined inclusion and exclusion criteria. During this initial screening, 126 abstracts were excluded for being off-topic, typically because they addressed topics like “open-label medication” rather than specifically OLPs. Subsequently, the full texts of the remaining 153 articles were retrieved for detailed review. Upon thorough examination of these 153 articles, an additional 136 studies were excluded for reasons such as being available only as abstracts (n = 16), not involving the application of open-label placebos (n = 6), or failing to discuss ethical dilemmas specifically in the context of open-label placebos (n = 114). Ultimately, 17 studies satisfied all criteria and were included in the review.
A PRISMA flow diagram (Fig. 1) depicts the entire selection process. For each of the 17 included studies, we extracted Publication Details: author(s), year, study design, sample size, population, aim and journal, which can be consulted in Table 3.
Figure 1 PRISMA flowchart overview.
Data analysis
We employed conventional qualitative content analysis64,65 to synthesize ethical themes across the included studies. This process was performed using MAXQDA software66 to ensure consistency. A subset was double coded (for a discussion on intercoder reliability see67). Table 2 shows a step-by-step breakdown on the analysis.
Results
From the initial pool of 279 non-duplicate records, 17 articles met the inclusion criteria (see Fig. 1). As shown in Table 4, most of these articles were published between 2019 and 2023 (11 out of 17). The studies vary in design, with most being qualitative studies or conceptual analyses, and one randomized controlled trial (Publication 12). Sample sizes and populations differ widely; most papers (10 out of 17) were conceptual pieces while the empirical ones focus on healthcare professionals (e.g., surgeons, physicians) or patients with specific conditions such as chronic pain.
Ethical issues
In total, we inductively derived 37 discrete ethical issues (“codes”) from the 17 articles. Each distinct concern was assigned a single code. Through iterative qualitative analysis, these codes were grouped into five overarching themes:
-
1.
Sociocultural and contextual factors.
-
2.
Implementation and logistics.
-
3.
Informed consent.
-
4.
Patient health behavior and dynamics.
-
5.
Therapeutic relationship.
Each theme captures related ethical discussions that cut across multiple articles. In the following section, we inspect the five themes with their 37 corresponding codes. In Table 4 we list the theme, code, corresponding publication (numbered as per Table 3), a brief explanation of the code and an example quote68,69,70,71. This allows readers to see both a concrete instance (via the quote) and the scope (which publications) for each code (a discussion on using quotes in qualitative studies can be found here72). After Table 4 we provide a summary text for each theme, where codes are printed bold, and publication numbers are mentioned again.
Sociocultural and contextual factors
The theme of Sociocultural and Contextual Factors became apparent in the discourse on placebo use. This theme encompasses the intricate and often contentious perspectives that various stakeholders hold regarding the legitimacy and utility of placebos within healthcare settings.
Publications coded under Alternative Medicine (Publications: 17, 5, 6, 9, 10, 13) reveal a recurrent concern regarding the potential for placebo use to foster or reinforce beliefs in the efficacy of the placebo itself or the broader concept of mind-body healing. A salient quote from publication 17 encapsulates the apprehension that such beliefs could be mistakenly held as sufficient for curing serious illnesses, thus neglecting established treatments. This suggests a risk of placebos contributing to an overvaluation of “positive thinking” in healing, possibly leading to the avoidance of medically proven interventions. Publications 3, 7, 13 and 14 also suggest a possible paradigm shift that pertains to the cultural and ethical discourse around placebo treatments potentially advancing and redefining their role within medical practice. A forecasted change, as detailed in publication 13, might see placebo therapy integrated into a legitimate clinical method, albeit in specific situations delineated by clear guidelines. The theme further engages with the influence of social norms (Publications: 3, 13) on patient-physician interactions, particularly how patients’ expectations of treatments are constructed and sometimes confronted in the context of OLPs. Publication 3 provides an account of a participant suggesting that OLPs could challenge social conventions, indicating a potential shift in the dynamics of patient-clinician relationships. Lastly, publications 8, 13 and 17 contribute to a discussion on how the use of placebos might affect patient and parental attitudes view on medicine. A noteworthy concern is that an increased reliance on placebos could inadvertently escalate the expectation for immediate “treatments” for every symptom, potentially exacerbating issues within medical practice as highlighted by publication 17. In summary, this theme underscores a complex interplay between sociocultural and contextual factors and the use of placebos to justify the use of alternative medicine. The data indicates both concern for the potential misappropriation of placebos and an optimistic outlook for their ethical integration into medical practice, subject to ongoing cultural and ethical discourse.
Implementation and logistics
The exploration of OLPs within the systematic review highlighted a nuanced spectrum of implementation and logistical considerations critical to their integration into medical practice. Acceptability was a pivotal concern, with intervention preference varying widely; some patients exhibited a readiness to embrace OLPs as an extension of their current treatment regimen (Publications: 2, 3, 4, 5, 6, 7, 8, 9). However, the potential for OLPs to serve as a dose extending OLP alongside active medication was met with both intrigue and scepticism, prompting calls for more definitive evidence to validate this application (Publications: 2, 3, 4, 8, 9, 11, 13, 15, 17). The issue of blinding in placebo-controlled trials was also broached, with publications indicating a challenge in maintaining the rigour of double-blind placebo research design when comparing open with hidden placebos and evaluating them against the best available treatment (Publications: 8, 9, 14). Doubts concerning the clear efficacy of OLPs were prevalent, with several studies suggesting a strong imperative for future research to provide solid evidence for their effectiveness, as OLP efficacy not clear yet (Publications: 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 14, 16, 17). Indeed, future work is needed by large-scale, pragmatic randomized trials to assess the true potential of OLPs, which was a recurrent theme, emphasizing the importance of robust, evidence-based conclusions (Publications: 1, 4, 5, 6, 7, 8, 9, 11, 12, 16). Legal aspects emerged as a barrier to the prescription of OLPs, with practitioners expressing concern over the consequences of possible negative outcomes from OLP use (Publications: 3, 4). Opinions were divided; some viewed OLPs as comparable to other prescriptions, while others called for additional provider training or patient education to ensure appropriate use, which is summarized under the code ‘logistics’ (Publications: 3, 7, 8, 9, 17).
‘OLP by Proxy’ was acknowledged as a phenomenon, particularly in paediatric care, where the belief systems of parents could significantly influence the placebo response in children, highlighting a complex dynamic in placebo use (Publications: 10, 11). The cost of OLPs was another factor affecting their acceptability, with patients hesitant to incur costs for an inert substance, suggesting economic factors need to be considered in the implementation strategy (Publications: 2, 7, 14).
Lastly, the notion of replacing drugs by OLPs was contemplated, especially in the context of treatments for conditions such as ADHD, where the goal might be the gradual withdrawal from active drugs. This theme suggested a careful and considered approach to introducing OLPs into the treatment regimen (Publications: 2, 9, 11).
Throughout this theme, it was apparent that while there is an underlying positive sentiment towards OLPs, their acceptance and practical application hinge on a multitude of factors that span the spectrum of ethical, legal, logistical, and economic considerations. The synthesis of these insights portrays a healthcare landscape cautiously navigating the potentialities of OLPs, with a clear consensus on the need for continued investigation and dialogue.
Informed consent
In the context of informed consent within the use of OLPs, this theme has uncovered several dimensions of concern and consideration. The ambiguity inherent in the disclosure process was highlighted, with some practitioners opting for vague language to introduce placebos to patients. This strategy involves offering OLPs with phrases like “here’s something you could try and let’s see if it works,” which, while avoiding definitive claims about the treatment keeping up ambiguity, could lead patients to assume they are receiving an active medical intervention (Publications: 1, 6, 7). The necessity for clinician OLP literacy is underscored, pointing to a need for comprehensive training to ensure that the wider adoption of OLPs is accompanied by accurate patient understanding and expectation management (Publications: 5, 7, 8, 9, 11). Publications suggest that OLPs are consistent with IC values and it is both achievable and necessary to mitigate the ethical risks associated with OLP use (Publications: 5, 7, 13, 16, 17).
The definition of OLPs was also a focal point of debate. It was proposed that recent discussions around OLPs, which often include considerations of communication, empathy, and other contextual factors, should not blur the conceptual boundaries of OLP and other non-specific effects (Publications: 3, 9, 15). OLP Disclosure practices are central to the ethical deployment. Evaluating whether the disclosures made in open-label placebo studies would be ethically acceptable in clinical encounters first requires a clear understanding of what is divulged to participants. Publications stressed that the disclosures and informed consent in clinical trials must be subject to careful moral scrutiny to differentiate them from disclosures in other therapeutic contexts (Publications: 1, 2, 5, 7, 9, 11, 15). Furthermore, the influence on decision-making was considered important. It was posited that the way in which OLPs are presented could independently influence how patients understand their illness and symptoms, and thus their maintenance and healthcare-seeking behaviour (Publications: 1, 11). A critical concern raised was the potential for patients not to fully understanding the consent and hence the concept of OLPs, raising alarms about the consistency of messaging and comprehension across care teams and between clinicians and patients (Publications: 7, 8, 11). Moreover, the notion of patients’ right to ‘not know’ or to know less was discussed, emphasizing the ethical complexity in ensuring that patients can consent to OLP use without forfeiting their right to fully informed consent or the benefits of a safe and transparent patient-clinician relationship (Publication: 13). In synthesizing these insights, it becomes evident that informed consent for OLPs is a multifaceted issue that intersects with ethical, communicative, and educational domains, each demanding careful attention to uphold the integrity of patient care and the ethical standards of clinical practice.
Patient health behaviour and dynamics
In the sphere of patient health behaviour and dynamics, this theme presents a multifaceted view informed by patient expectations and the perceived harms versus benefits of OLPs. Expectations play a crucial role in the acceptability of placebo treatments, with outcomes depending greatly on how patients anticipate the effectiveness of treatment (Publications: 5, 9, 11). Physicians need to carefully evaluate harm vs. benefits (Publications: 3, 7, 9, 13, 15). The potential influence of OLPs on healthcare-seeking behaviour is complex. On the one hand, they can empower patients, providing a sense of control and comfort. Yet, there are once more concerns that by being prescribed placebos, some patients may lose faith in mainstream medicine, erroneously believing that placebos legitimize complementary and alternative treatments, which influences help-seeking behaviour (Publications: 5, 6, 16).
The phenomenon of nocebo effects—where an inactive substance causes negative outcomes—is also a significant concern. This includes the potential for unknown risks or ‘nocebo effects,’ where the expectation of an adverse effect by patients precipitates or exacerbates the experience of that symptom (Publications: 2, 3, 8, 9, 17). Moreover, there is the issue of overestimating the positive effects of OLPs. While considerable research remains to be done, there is a need for honesty about the current understanding of the boundaries of placebo potential and the realistic communication of what placebo responses can and cannot do for people (Publications: 2, 5, 7, 10, 11). Overprescription of drugs is a concern tied closely to the use of OLPs. The widespread use of placebos could intensify the already prevalent expectation for treatments for every symptom, potentially leading to an overreliance on medication in general, which may crowd out other forms of care or lead to the devaluation of non-pharmacological treatments and to overprescription of drugs (Publication: 17). Lastly, the issue of structural injustice was addressed. Assumptions made about patients when they are giving testimony can be rooted in biases and power imbalances between social groups. When OLPs are integrated within minority or participant groups that have been historically disbelieved or devalued, there is a risk that this might reinforce social disparities. This runs the risk of exacerbating these imbalances (Publication: 16).
Therapeutic relationship
The dynamics within the therapeutic relationship are critically reflected upon within this theme. In environments where medical expertise is rapidly expanding and the lexicon becoming ever more specialized, the relationship between healthcare providers and patients is strained, with the latter often feeling overwhelmed and isolated (Publications: 2, 3, 10, 14, 15). Agency, particularly in the context of OLPs, is emphasized as a key component of the therapeutic relationship. It is recognized that the role of agency in placebo effects is complex and traverses the clinical context, necessitating a broader view of potential for promoting placebo effects in various domains. This involves understanding the underlying mechanisms of how they can promote agency and benefit patient outcomes (Publications: 3, 9, 10, 11). Additionally, the concept of autonomy within the therapeutic relationship is underscored, highlighting the importance of patient autonomy in any placebo-centric practice. The review suggests that autonomy plays a critical role in domains far beyond the immediate clinical encounter (Publications: 7, 9, 10, 14, 17).
Deception is a contentious issue that can include offering false hope, the downplaying of risks and ambiguously formulated messages that may foster more positive expectations than the evidence justifies. The fine line between justified optimism and exaggerated confidence presents a moral dilemma, where placebo-based approaches must operate within the bounds of truthfulness (Publications: 1, 5, 8, 15).
The impact of OLPs on long-term health outcomes and the potential for blame was discussed, acknowledging the uncertainty about how different patient groups may internalize the use of OLPs. There is a risk that patients, especially those with medically unexplained symptoms, may internalize blame, which could be mitigated by how practitioners frame the use of OLPs (Publications: 6, 9, 10, 11, 14, 17). Paternalism in the therapeutic relationship was also critiqued, particularly when researchers assume a psychogenic mechanism for patients’ experiences, leading to the use of placebos rather than a conventional pill, potentially even without the patients’ full understanding or consent (Publications: 15, 16). Responsibility in prescribing OLPs is highlighted, posing the question of the ethical considerations that fall within the bounds of medicine and what is considered difficult care. The necessity for clear communication about the influences on placebo responses is emphasized, especially regarding informed consent processes (Publication: 10). Lastly, trust is identified as a cornerstone of the therapeutic relationship, with concerns that the prescription of placebo treatments could undermine trust in physicians, particularly if there is a perceived imbalance in the relationship (Publications: 5, 9, 12, 13, 14, 17).
Discussion
To our knowledge, this is the first systematic qualitative review of the full range of ethical issues discussed in the OLP literature. As can be seen in the PRISMA-Flowchart, we excluded articles that mention ethical issues surrounding OLPs but do not reflect on them. Those excluded fifty-four papers mainly mention deception as the primary or sole ethical issue with “traditional” placebo but then argue that there is no ethical concern with OLP due to the lack of deception6. However, as we have uncovered through this comprehensive analysis, there are several ethical issues that have been raised surrounding OLPs. Since, ideally, research leads to policy and implementation, it is important that researchers and ethicists grapple with these concerns as the ethical considerations associated with OLPs may not be alleviated by the mere absence of deception. On the contrary, the ethical considerations of OLP in medical practice are multifaceted, encompassing patient choices, the role of CAM, transparent communication, logistical challenges, and the complex dynamics of therapeutic relationships. Careful consideration and responsible communication are imperative to navigate these complexities and ensure the ethical and effective incorporation of OLPs in healthcare.
We did do an analysis of the most occurring codes and several articles stated that it is pivotal to further demonstrate the efficacy of OLPs as the widespread adoption of OLPs hinges on the confirmation of their effectiveness. We decided to put these results into the discussion section due to the echo-chamber concerns (explained below). i.e., we rank Table 5 according to most to least different publications that mentioned the specific ethical issue). As summarized in Table 6, “OLP Efficacy Not Clear” was the most frequently mentioned code (13 of 17 articles), closely followed by “Future Work Needed” (10 of 17), “Dose Extending OLP” (9 of 17), and “Acceptability” (9 of 17). Notably, these four most frequent codes all fall under the Implementation and Logistics theme, making it the most widely discussed theme across the included literature.
Whilst (concealed) placebos have been a topic of investigation for many decades, OLPs have emerged only in recent years and are still relatively unknown. Hence the codes that appeared the most in this review are not surprising (OLP efficacy not clear & more research needed). There has been one review and meta-analysis that pointed to the efficacy of OLPs in certain conditions compared to no-treatment23,73. This review found a significant overall effect favouring OLPs but also points out that the research is in its infancy and more work is needed. The prevailing scientific consensus is evident in our own analysis, which explicitly states that, despite the promising nature of the approach, it is too early for the clinical implementation of OLPs.
When we examined the articles included in this review, we noticed that there is a lack of author heterogeneity within the papers discussing OLPs highlighting a potential limitation in the breadth and diversity of perspectives being presented. The frequent recurrence of a limited group of scholars across multiple articles within our dataset suggests a risk of an echo chamber74, where the repetition of specific ethical considerations by the same authors could inadvertently create the illusion of a broader consensus, when it is in fact reflective of a single group’s viewpoint. In our opinion, this observation underscores the critical need for broadening the field to include a wider range of voices and perspectives, thereby enriching the ethical discourse surrounding OLPs and ensuring a more balanced and inclusive exploration of their potential and limitations.
Also, it is noteworthy that in another high occurring code in the reviewed articles many discussions revolved around implementing OLPs in form of a dose extension rather than a complete replacement of an active medication. Further research into dose extension via OLPs and possible reduction of active medication as well as minimization of side-effects of active ingredients is indicated which is an important step towards possible implementation of OLPs. Research thus far has been promising as some studies have observed OLPs can reduce prescription pain medication75. Moreover, Kirchof and colleagues found that open placebos paired with a conditioned stimulus could have a positive immunological function76.
The most controversial issue in our eyes is the role of CAM, which has also been brought up prominently by Fabrizio Benedetti in his talk at the fourth international conference of the society of interdisciplinary placebo studies (SIPS 2023): “Today many quacks say their bizarre interventions work because of a placebo effect, thus they justify them. Paradoxically, research on placebo effects supports this assertion. What can we do to avoid this worrisome tendency?”77. As per today, no such investigation has been undertaken.
Limitations
Abstract screening was performed by a single reviewer, which, despite careful adherence to predefined criteria, might have introduced unintended selection bias or oversight. Future research involving multiple reviewers could strengthen the robustness of the selection process. Additionally, variations in coding practices or differing research foci across studies included in this review could influence the identification and interpretation of ethical issues. Furthermore, it is important to recognize that ethical issues identified here may not universally apply across different healthcare settings, patient populations, and cultural contexts, highlighting opportunities for further investigation and contextual validation. Finally, potential language bias should be acknowledged, as we only included English and German papers.
Outlook
The current outlook suggests a multifaceted research agenda that addresses the complexities of OLPs through an interdisciplinary lens, blending scientific inquiry with ethical considerations and patient-centred approaches. The first step on this path is a concerted effort to expand the evidence base through larger, more comprehensive studies that can verify the effectiveness and safety of OLPs across various conditions. These studies must be designed to not only test efficacy in a controlled environment but also to understand the real-world implications of prescribing OLPs in routine clinical practice. Understanding the mechanisms through which OLPs exert their effects remains an important objective. This includes further biological research to unravel the neurobiological pathways involved, as well as psychological research to explore the role of patient expectations, the therapeutic relationship, and the power of suggestion. As the body of evidence grows, there could be a shift towards the integration of OLPs into clinical practice, necessitating robust physician education programs. These programs should focus on when and how to effectively prescribe OLPs, manage patient expectations, and navigate the complex ethical terrain that surrounds their use.
The code “Future Work Needed” emerged among the top four in Table 5, reflecting a strong consensus that further investigation is required. Table 6 distils the recommendations from these segments, encompassing:
-
Calls for Larger Studies: Multiple authors advocate for robust clinical trials to ascertain OLP efficacy and guide best practices.
-
Mechanisms of Action: Future research could delve deeper into how embodiment, conditioning, and expectancy influence OLP outcomes.
-
Physician Training: Many publications suggest integrating OLP education into medical curricula, with emphasis on communication strategies and ethical considerations.
Such recommendations align with the high frequency of codes like “OLP Efficacy Not Clear” and “Dose Extending OLP,” indicating that while the potential of OLPs is compelling, the scholars have argued it is still in its early stages and requires more empirical substantiation.
The ethical dimension of OLP research will continue to be a critical area of focus. Future research must ensure that the use of OLPs adheres to the highest standards of patient consent and autonomy, with clear communication strategies developed to inform patients about the nature of OLPs and their potential benefits and limitations. With the rise of personalized medicine, the tailoring of OLP interventions to individual patient profiles could enhance therapeutic outcomes78. This precision approach would consider genetic, environmental, and lifestyle factors that may influence the effectiveness of OLPs. In paediatric and geriatric populations, where OLP research is particularly sparse, future studies should strive to fill this gap, considering the specific ethical, physiological, and psychological considerations that are unique to these age groups. The potential for OLPs to reduce reliance on pharmacological treatments, especially in the context of chronic pain management and psychological disorders, is another promising avenue for exploration. This could have significant implications for public health, offering a cost-effective and low-risk alternative to long-term medication use.
Data availability
All data generated or analysed during this study are included in this published article.
References
Blease, C., Colloca, L. & Kaptchuk, T. J. Are open-label placebos ethical? Informed consent and ethical equivocations. Bioethics 30, 407–414. https://doi.org/10.1111/bioe.12245 (2016).
Bartley, H., Clarke, S. & Ryan, C. Ethical frameworks for the use of placebos in clinical practice: a scoping review. BMC Med. Ethics 22, 56. https://doi.org/10.1186/s12910-021-00631-9 (2021).
Foddy, B. A duty to deceive: placebos in clinical practice. Am. J. Bioethics 9, 4–12. https://doi.org/10.1080/15265160903318350 (2009).
Hróbjartsson, A. & Gøtzsche, P. C. Placebo interventions for all clinical conditions. Cochrane Database Syst. Rev. 2010, 003974. https://doi.org/10.1002/14651858.CD003974.pub3 (2010).
Miller, F. G. & Colloca, L. The legitimacy of placebo treatments in clinical practice: evidence and ethics. Am. J. Bioethics 9, 39–47. https://doi.org/10.1080/15265160903316263 (2009).
Kaptchuk, T. J. & Miller, F. G. Placebo effects in medicine. N. Engl. J. Med. 373, 8–9. https://doi.org/10.1056/NEJMp1504023 (2015).
Finniss, D. G., Kaptchuk, T. J., Miller, F. & Benedetti, F. Biological, clinical, and ethical advances of placebo effects. Lancet 375, 686–695. https://doi.org/10.1016/s0140-6736(09)61706-2 (2010).
Richard, M. et al. Noble Humbug?? Hard and soft laws on clinical placebo use. Front. Psychol. Sec Psychol. Clin. Settings 16, 664. https://doi.org/10.3389/fpsyg.2025.1520664 (2025).
Czerniak, E. & Davidson, M. Placebo, a historical perspective. Eur. Neuropsychopharmacol. 22, 770–774. https://doi.org/10.1016/j.euroneuro.2012.04.003 (2012).
Finniss, D. G. International Review of Neurobiology Vol. 139 (ed Colloca, L.) 1–27 (Academic Press, 2018).
Nestoriuc, Y. & Kleine-Borgmann, J. Der Schein Truegt nicht—klinische evidenz und neue forschungsansaetze zum Open-label-Placebo (Appearances are not deceptive: clinical evidence and new research approaches to open-label placebo). Der Nervenarzt 91, 708–713. https://doi.org/10.1007/s00115-020-00953-6 (2020).
Kleine-Borgmann, J. et al. Effects of open-label placebos on test performance and psychological well-being in healthy medical students: a randomized controlled trial. Sci. Rep. 11, 2130. https://doi.org/10.1038/s41598-021-81502-2 (2021).
Hahn, A., Gohler, A. C., Hermann, C. & Winkler, A. Even when you know it is a placebo, you experience less sadness: first evidence from an experimental open-label placebo investigation. J. Affect. Disord. 304, 159–166. https://doi.org/10.1016/j.jad.2022.02.043 (2022).
Winkler, A., Hahn, A. & Hermann, C. The impact of pharmaceutical form and simulated side effects in an open-label-placebo RCT for improving psychological distress in highly stressed students. Sci. Rep. 13, 6367. https://doi.org/10.1038/s41598-023-32942-5 (2023).
Schaefer, M. et al. Open-label placebos reduce test anxiety and improve self-management skills: A randomized-controlled trial. Sci. Rep. 9, 13317. https://doi.org/10.1038/s41598-019-49466-6 (2019).
Schaefer, M., Sahin, T. & Berstecher, B. Why do open-label placebos work? A randomized controlled trial of an open-label placebo induction with and without extended information about the placebo effect in allergic rhinitis. PLoS ONE 13, e0192758. https://doi.org/10.1371/journal.pone.0192758 (2019).
Barnes, K., Babbage, E., Barker, J., Jain, N. & Faasse, K. The role of positive information provision in open-label placebo effects. Appl. Psychol. Health Well-Being 15, 1406–1426. https://doi.org/10.1111/aphw.12444 (2023).
Maher, C. G., Traeger, A. C. & Shaheed, A. O’Keeffe, M. Placebos in clinical care: a suggestion beyond the evidence. Med. J. Aust. 215, 252–253. https://doi.org/10.5694/mja2.51230 (2021).
Blease, C., Bernstein, M. & Locher, C. Open-label placebo clinical trials: is it the rationale, the interaction or the pill? BMJ 25, 159–165. https://doi.org/10.1136/bmjebm-2019-111209 (2020).
Blease, C., Colagiuri, B. & Locher, C. Replication crisis and placebo studies: rebooting the bioethical debate. J. Med. Ethics 49, 663–669. https://doi.org/10.1136/jme-2022-108672 (2023).
Jones, C. M. P. et al. Time to reflect on open-label placebos and their value for clinical practice. Pain 164, 2139–2142. https://doi.org/10.1097/j.pain.0000000000003017 (2023).
Blease, C. R., Kaptchuk, T. J. & Locher, C. Open-label placebos for treating functional disorders: ethical challenges. J. Med. Ethics 49, 573–578. https://doi.org/10.1136/jme-2022-108379 (2023).
von Wernsdorff, M., Loef, M., Tuschen-Caffier, B. & Schmidt, S. Effects of open-label placebos in clinical trials: a systematic review and meta-analysis. Sci. Rep. 11, 3855. https://doi.org/10.1038/s41598-021-83148-6 (2021).
Morales-Quezada, L. et al. Conditioning open-label placebo: a pilot pharmacobehavioral approach for opioid dose reduction and pain control. Pain Rep. 5, e828. https://doi.org/10.1097/pr9.0000000000000828 (2020).
Krockow, E. M., Emerson, T., Youssef, E., Scott, S. & Tromans, S. Evidencing general acceptability of open-label placebo use for tackling overtreatment in primary care: a mixed methods study. BMC Med. 21, 362. https://doi.org/10.1186/s12916-023-03074-4 (2023).
Trogen, B., Caplan, A. & Klass, P. The ethics of open-label placebos in pediatrics. Pediatrics 140, 4328. https://doi.org/10.1542/peds.2016-4328 (2017).
Bernstein, M. H. et al. Treating pain with open-label placebos: A qualitative study with post-surgical pain patients. J. Pain 22, 1518–1529. https://doi.org/10.1016/j.jpain.2021.05.001 (2021).
Meeuwis, S. H. et al. Placebo effects of open-label verbal suggestions on itch. Acta Dermato-Venereol. 98, 268–274. https://doi.org/10.2340/00015555-2823 (2018).
Kirsch, I. Hypnosis in psychotherapy: efficacy and mechanisms. Contemp. Hypn. 13, 109–114. https://doi.org/10.1002/ch.57 (1996).
Colloca, L. & Miller, F. G. Harnessing the placebo effect: the need for translational research. Philos. Trans. R. Soc. Lond. B Biol. Sci. 366, 1922–1930. https://doi.org/10.1098/rstb.2010.0399 (2011).
Annoni, M. & Miller, F. G. Placebo effects and the ethics of therapeutic communication: A pragmatic perspective. Kennedy Inst. Ethics J. 26, 79–103. https://doi.org/10.1353/ken.2016.0004 (2016).
Bernstein, M. H. et al. Primary care providers’ use of and attitudes towards placebos: an exploratory focus group study with US physicians. Br. J. Health Psychol. 25, 596–614. https://doi.org/10.1111/bjhp.12429 (2020).
Bernstein, M. H. et al. How orthopedic surgeons view open label placebo pills: ethical and effective, but opposed to personal use. J. Psychosom. Res. 151, 110638. https://doi.org/10.1016/j.jpsychores.2021.110638 (2021).
Blease, C. R. The role of placebos in family medicine: implications of evidence and ethics for general practitioners. Aust J. Gen. Pract. 48, 700–705. https://doi.org/10.31128/AJGP-05-19-4939 (2019).
Bliamptis, J. & Barnhill, A. Physician perspectives on placebo ethics. J. Med. Ethics. https://doi.org/10.1136/medethics-2021-107446 (2021).
Colloca, L. & Howick, J. Placebos without deception: outcomes, mechanisms, and ethics. Int. Rev. Neurobiol. 138, 219–240. https://doi.org/10.1016/bs.irn.2018.01.005 (2018).
Druart, L., Vauthrin, O., Pinsault, N., Locher, C. & Blease, C. It’s not my Greengrocer, it’s someone from the medical profession’: A qualitative study regarding acceptability of deceptive and open-label placebo prescribing in France. Br. J. Health Psychol. 28, 273–290. https://doi.org/10.1111/bjhp.12624 (2023).
Friesen, P. Placebos as a source of agency: evidence and implications. Front. Psychiatry. 10, 721. https://doi.org/10.3389/fpsyt.2019.00721 (2019).
Locher, C. et al. Placebos are part of the solution, not the problem. An exemplification of the case of antidepressants in pediatric chronic pain conditions. Front. Psychiatry 10, 998. https://doi.org/10.3389/fpsyt.2019.00998 (2019).
Mundt, J. M., Roditi, D. & Robinson, M. E. A comparison of deceptive and non-deceptive placebo analgesia: efficacy and ethical consequences. Ann. Behav. Med. 51, 307–315. https://doi.org/10.1007/s12160-016-9854-0 (2017).
Nitzan, U., Bloch, Y. & Fennig, S. Using placebo medications in the clinical setting—an intellectual game or a possible reality? Isr. J. Psychiatry Relat. Sci. 51, 182–187 (2014).
Ratnapalan, M. et al. Placebos in primary care? A nominal group study explicating UK GP and patient views of six theoretically plausible models of placebo practice. BMJ Open 10, e032524. https://doi.org/10.1136/bmjopen-2019-032524 (2020).
Schone-Seifert, B. Placebos and placeboids in clinical practice: conceptual and ethical considerations. Nervenarzt 91, 684–690. https://doi.org/10.1007/s00115-020-00943-8 (2020).
Specker Sullivan, L. More than consent for ethical open-label placebo research. J. Med. Ethics. https://doi.org/10.1136/medethics-2019-105893 (2020).
Heiss, U. Can the Open Label Placebo Rationale Be Optimized?.
Schwartz, M. & Klinger, R. Analgesic placebo effects and implications for the treatment of chronic pain. Psychotherapeutics 67, 220–226. https://doi.org/10.1007/s00278-022-00592-z (2022).
Frey Nascimento, A. Auf dem Weg der Heilung—Die Rollen und Interaktionen zwischen Patient und Therapeut in der Psychotherapie naeher beleuchtet (DeepL) (On the Healing Pathway—Zooming in on Patient-Provider Roles and Interactions in Psychotherapy). https://doi.org/10.5451/unibas-ep83232 (2021).
Kleine-Borgmann, J. & Bingel, U. The placebo effect in pain therapy—Suitable for everyday clinical use? Yes, definitely!. Aktuelle Rheumatol. 45, 453–459. https://doi.org/10.1055/a-1165-6689 (2020).
Kleine-Borgmann, J., Schmidt, K., Hellmann, A. & Bingel, U. Reply to Amorim et al. Pain 161, 1124–1125. https://doi.org/10.1097/j.pain.0000000000001804 (2020).
Lembo, A. et al. Open-label placebo vs double-blind placebo for irritable bowel syndrome: a randomized clinical trial. Pain 162, 2428–2435. https://doi.org/10.1097/j.pain.0000000000002234 (2021).
Haas, J. W. et al. Patients’ experiences treated with open-label placebo versus double-blind placebo: a mixed methods qualitative study. BMC Psychol. 10, 20. https://doi.org/10.1186/s40359-022-00731-w (2022).
Ballou, S. et al. Open-label versus double-blind placebo treatment in irritable bowel syndrome: study protocol for a randomized controlled trial. Trials 18, 234. https://doi.org/10.1186/s13063-017-1964-x (2017).
Hannes, K. Guidance on the Conduct of Narrative Synthesis in Systematic Reviews (ed. Popay, J.) 42–48 (ESRC Methods Programme, 2011).
Thomas, J. & Harden, A. Methods for the thematic synthesis of qualitative research in systematic reviews. BMC Med. Res. Methodol. 8, 45. https://doi.org/10.1186/1471-2288-8-45 (2008).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 6, e1000097. https://doi.org/10.1371/journal.pmed.1000097 (2009).
Noyes, J. et al. Cochrane qualitative and implementation methods group guidance series—paper 1: introduction. J. Clin. Epidemiol. 75, 6–10. https://doi.org/10.1016/j.jclinepi.2015.03.013 (2015).
Kahrass, H., Strech, D. & Mertz, M. The full spectrum of clinical ethical issues in kidney failure. Findings of a systematic qualitative review. PLoS ONE 11, e0149357. https://doi.org/10.1371/journal.pone.0149357 (2016).
Mertz, M., Kahrass, H. & Strech, D. Current state of ethics literature synthesis: a systematic review of reviews. BMC Med. 14, 152. https://doi.org/10.1186/s12916-016-0688-1 (2016).
Seitzer, F., Kahrass, H., Neitzke, G. & Strech, D. The full spectrum of ethical issues in the care of patients with ALS: a systematic qualitative review. J. Neurol. 263, 201–209. https://doi.org/10.1007/s00415-015-7867-4 (2016).
Strech, D., Mertz, M., Knuüppel, H., Neitzke, G. & Schmidhuber, M. The full spectrum of ethical issues in dementia care: systematic qualitative review. Br. J. Psychiatry 202, 400–406. https://doi.org/10.1192/bjp.bp.112.116335 (2013).
Katchamart, W., Faulkner, A., Feldman, B., Tomlinson, G. & Bombardier, C. PubMed had a higher sensitivity than Ovid-MEDLINE in the search for systematic reviews. J. Clin. Epidemiol. 64, 805–807. https://doi.org/10.1016/j.jclinepi.2010.06.004 (2011).
Covidence Systematic Review Software (2021).
Beauchamp, T. L. & Childress, J. F. Principles of Biomedical Ethics, 7th edn (Oxford University Press, 2013).
Hsieh, H. F. & Shannon, S. E. Three approaches to qualitative content analysis. Qual. Health Res. 15, 1277–1288. https://doi.org/10.1177/1049732305276687 (2005).
Schreier, M. Qualitative Content Analysis in Practice (SAGE, 2012).
MAXQDA V. Version 2022 (VERBI Software, 2022).
O’Connor, C. & Joffe, H. Intercoder reliability in qualitative research: debates and practical guidelines. Int. J. Qual. Methods 19, 1609406919899220. https://doi.org/10.1177/1609406919899220 (2020).
Tong, A., Sainsbury, P. & Craig, J. Consolidated criteria for reporting qualitative research (COREQ): a 32-item checklist for interviews and focus groups. Int. J. Qual. Health Care 19, 349–357. https://doi.org/10.1093/intqhc/mzm042 (2007).
Braun, V. & Clarke, V. Using thematic analysis in psychology. Qual. Res. Psychol. 3, 77–101 (2006).
Miles, M. B., Huberman, A. M. & Saldaña, J. Qualitative Data Analysis: A Methods Sourcebook, 3rd edn (SAGE, 2014).
Patton, M. Q. Qualitative Research & Evaluation Methods, 4th edn (SAGE, 2015).
Eldh, A. C., Årestedt, L. & Berterö, C. Quotations in qualitative studies: reflections on constituents, custom, and purpose. Int. J. Qual. Methods 19, 1609406920969268. https://doi.org/10.1177/1609406920969268 (2020).
von Wernsdorff, M., Loef, M., Tuschen-Caffier, B. & Schmidt, S. Author correction: effects of open-label placebos in clinical trials: a systematic review and meta-analysis. Sci. Rep. 11, 17436. https://doi.org/10.1038/s41598-021-96604-0 (2021).
Warriner, D. How to read a paper: the basics of evidence-based medicine. BMJ 336, 1381. https://doi.org/10.1136/bmj.a186 (2008).
Estudillo-Guerra, M. A., Mesia-Toledo, I., Schneider, J. C. & Morales-Quezada, L. The use of conditioning open-label placebo in opioid dose reduction: A case report and literature review. Front. Pain Res. 2, 475. https://doi.org/10.3389/fpain.2021.697475 (2021).
Kirchhof, J. et al. Learned immunosuppressive placebo responses in renal transplant patients. Proc. Natl. Acad. Sci. U.S.A. 115, 4223–4227. https://doi.org/10.1073/pnas.1720548115 (2018).
Benedetti, F. 4th International Conference of the Society of Interdisciplinary Placebo Studies (SIPS, 2023).
Colloca, L. The placebo effect in pain therapies. Annu. Rev. Pharmacol. Toxicol. 59, 191–211. https://doi.org/10.1146/annurev-pharmtox-010818-021542 (2019).
Author information
Authors and Affiliations
Contributions
M.R. planning, writing manuscript, PRISMA, coding. M.B double coding, editing, and revising manuscript. J.G. editing, B.E. planning, editing, and revising manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and informed consent
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Richard, M., Bernstein, M., Gaab, J. et al. A systematic qualitative review of ethical issues in open label placebo in published research. Sci Rep 15, 12268 (2025). https://doi.org/10.1038/s41598-025-96425-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-96425-5
Keywords
This article is cited by
-
The mind-bending power of placebo
Nature Medicine (2025)