Abstract
In this study, graphene oxide (GO) nanosheets were chemically modified by attaching polyaniline (PAN) nanoparticles to their surfaces, creating a polyaniline partially reduced graphene oxide composite (PAN@PRGO). This synthesized PAN@PRGO nanocomposite serves as an innovative and highly effective adsorbent for removing indigo carmine (IC) dye from water. The morphology and chemical composition of PAN@PRGO were analyzed using various techniques, including scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), and X-ray photoelectron spectroscopy (XPS), confirming the successful grafting of PAN onto the GO surface. Batch adsorption tests showed that PAN@PRGO has an outstanding adsorption capacity for indigo carmine (IC) dye, achieving 490.0 mg g−1 at pH 5.0 and 298 K. This is notably higher than the adsorption capacity of GO nanosheets alone (317.25 mg g−1) and exceeds that of other materials reported in the literature. Additionally, PAN@PRGO demonstrated 100% removal efficiency for IC dye at concentrations up to 300 mg L−1. The experimental data closely matched the Langmuir isotherm model and the pseudo-second-order kinetic model, suggesting that electron-sharing interactions between IC dye and PAN@PRGO contribute to the adsorption mechanism. The adsorbed IC dye was recoverable using a 0.1 M NaOH solution, with the composite retaining near-100% efficiency even after five adsorption–desorption cycles. These results indicate that the PAN@PRGO composite is a promising, reusable adsorbent for effective IC dye removal from industrial wastewater.
Similar content being viewed by others
Introduction
The growing concerns over public health necessitate ensuring a clean water supply, as access to safe and adequate water is crucial for reducing disease burdens and improving well-being1,2,3. Unfortunately, the expansion of industries such as textiles, plastics, printing, paper, and leather has resulted in the discharge of large volumes of wastewater contaminated with hazardous organic dyes. These dyes pose significant risks to both human health and the environment, making their removal from industrial effluents a pressing challenge4,5,6.
Indigo Carmine (IC) is a water-soluble anionic dye with a molar absorption coefficient of approximately 2 × 104 M−1 cm−1 at 600 nm7,8. Due to its extensive use in textiles, pharmaceuticals, cosmetics, medicine, and the food industry, IC is frequently detected in wastewater, particularly from textile effluents. Even at trace levels, IC can alter water color, degrade water quality, and render it unfit for consumption7,8. Studies have reported IC concentrations in wastewater ranging from a few milligrams per liter to several hundred milligrams per liter, depending on the industrial source and treatment methods employed. For instance, IC concentrations of approximately 64.6 mg L−1 have been detected in textile wastewater, while levels as high as 500 mg L−1 and 200 mg L−1 have been reported in effluents from textile and dye manufacturing plants, respectively9,10,11,12.
Several methods have been explored for dye removal, including electrochemical oxidation, ion exchange, ozonation, biological treatment, photocatalytic degradation, membrane filtration, and adsorption13,14,15,16,17,18. Among these, adsorption is widely regarded as a promising approach due to its simplicity, cost-effectiveness, high removal efficiency, and ability to regenerate adsorbents for multiple cycles13,19,20. Various adsorbents, such as activated carbon21, polymers22, hydrogels23, metal oxides24, carbon nanotubes25, and metal–organic frameworks26, have been investigated for IC removal. In particular, carbon-based materials, including graphene oxide (GO) and its derivatives, have gained considerable attention due to their high surface area, chemical versatility, and superior adsorption capabilities20,27,28. GO, a two-dimensional nanomaterial with a unique layered structure, is rich in oxygen-containing functional groups (–OH, –COOH, –C=O, and epoxy), which facilitate electrostatic interactions, hydrogen bonding, and π–π stacking with dye molecules. While GO exhibits excellent adsorption properties, its strong hydrophilicity, structural instability, and tendency to aggregate in aqueous media can limit its effective surface area and hinder its reusability. Therefore, modifying GO to enhance its adsorption efficiency, selectivity, and long-term stability is essential for practical wastewater treatment applications20,27,28,29.
Among potential modifiers, polyaniline (PAN) has emerged as a promising candidate due to its unique electronic properties, ease of synthesis, and strong interactions with dye molecules via hydrogen bonding, electrostatic forces, and π–π interactions30. PAN modification of GO addresses key limitations associated with unmodified GO by improving its dispersion, preventing nanosheet restacking, and introducing additional nitrogen-containing functional groups (–NH, =N–, –NH2), which further enhance dye adsorption31,32. Despite several studies on polyaniline grapheme oxide composites (PAN-GO) for dye removal, most research has focused on general dye adsorption rather than the selective removal of IC. Thus, there remains a research gap in optimizing PAN-GO composite for IC removal from wastewater.
In this study, a polyaniline partially reduced graphene oxide (PAN@PRGO) composite was synthesized via in situ polymerization of aniline on GO nanosheets through π–π stacking, electrostatic interactions, and hydrogen bonding. The synergy between PAN and partially reduced GO (PRGO) enhances the composite’s adsorption kinetics, stability, and reusability. Comprehensive characterization using SEM, FTIR, XPS, and XRD was conducted to confirm successful composite formation. The adsorption performance of PAN@PRGO for IC dye was systematically evaluated under various conditions, including initial dye concentration, pH, contact time, adsorbent dosage, temperature, and reusability. To ensure the practical relevance of this study, IC concentrations ranging from 10 to 800 mg L−1 were selected, covering the typical contamination levels observed in real-world wastewater scenarios. This study provides a detailed investigation of PAN@PRGO composites as an advanced adsorbent for IC removal, contributing to the development of efficient and sustainable wastewater treatment technologies. Figure 1 presents an overview of the PAN@PRGO synthesis process.
Experimental section
Materials
High-purity graphite powder (99.999%) was utilized in this experiment. The oxidizing agents included concentrated sulfuric acid (H2SO4, 98%), phosphoric acid (H₃PO4, 99%), and potassium permanganate (KMnO4, 99%). Other chemicals involved were hydrogen peroxide (H2O2, 30%), ethanol (99%), potassium persulfate (K2S2O8, 99.9%), aniline (99.55%), and deionized water (DI). All chemicals were purchased from Sigma Chemical Co. For additional details on characterization, refer to the Supporting Information (S1).
Synthesis of polyaniline partially reduced graphene oxide (PAN@PRGO) composite
Graphene oxide (GO) was synthesized following the procedure outlined in Ref.2. Improved GO (GO) was prepared by mixing a 1:9 concentrated H₃PO4/H2SO4 solution (60:540 mL) with a blend of KMnO4 (27.0 g) and graphite flakes (4.5 g) while maintaining the temperature below 30 °C in an ice bath. The reaction mixture was then heated to 50 °C and stirred for 12 h, followed by cooling to room temperature. Afterward, 30% H2O2 (4.5 mL) and 600 mL of iced distilled water were added. The mixture was centrifuged, and the solid product was washed sequentially with 200 mL each of deionized water, 30% nitric acid (HNO₃), and 2% ethanol. The final product was dried in a vacuum oven at 80 °C overnight to yield GO. For PAN@PRGO preparation, 0.2 g of IGO was dispersed in 75.0 mL of DI water and sonicated to create a stable suspension. Aniline (1.3 mL) was added to the suspension, followed by 1-h sonication. Next, 7.0 mL of concentrated HCl was introduced under continuous stirring for an additional hour. Then, 2.5 g of potassium persulfate dissolved in 1.0 M HCl was added while maintaining the reaction mixture at 50 °C in an ice bath with stirring for 12 h. After centrifugation, the finished product was rinsed with ethanol and DI water until the filtrate was clear, and it was then dried in an oven at 80 °C.
Batch adsorption studies
Batch adsorption experiments for IC dye removal were conducted by stirring 10 mg of the PAN@PRGO composite as an adsorbent in 10 mL of IC dye solution inside a 20-mL glass vial under various conditions: IC concentrations (10–800 mg L−1), pH values (2–8), contact time (15–240 min), adsorbent dosage (5–30 mg), and temperatures (25–60 °C), all at a constant stirring rate of 250 rpm. Regeneration of the adsorbent was achieved using 0.1 M NaOH as a desorbing agent, and the recovered adsorbent was subsequently reused in further adsorption experiments. After each adsorption experiment, IC dye concentration was measured spectrophotometrically at ƛ = 600 nm. The adsorption capacity of PAN@PRGO and the amount of dye removed were determined using the following Eqs. (1) and (2)33:
where Ci represents the initial concentration of IC (mg L−1), Ce denotes the equilibrium concentration of IC (mg L−1), V stands for solution volume (L), m for adsorbent dosage (g), and qe for the adsorbent’s absorption capability (mg g−1).
Results and discussion
Characterization of GO and PAN@PRGO composite
The surface morphology of GO and PAN@PRGO was investigated using scanning electron microscopy (SEM). Figure 2 presents SEM images of GO and PAN@PRGO, revealing distinct morphological characteristics. GO exhibits an irregular, layered structure with uniformly stacked graphene nanosheets that possess a clean and smooth surface. In contrast, the SEM images of the PAN@PRGO nanocomposite indicate the successful formation of PAN nanoparticles, which are homogeneously distributed on the surface of the graphene nanosheets.
At higher magnifications, SEM images further reveal that PAN nanoparticles are spherical with well-defined sharp edges. Notably, the PAN matrix plays a crucial role in preventing GO sheet aggregation by acting as a spacer. This structural modification significantly enhances the effective surface area and increases the availability of active adsorption sites. Consequently, the improved morphology of PAN@PRGO contributes to enhanced adsorption efficiency. These findings confirm the successful grafting and in situ polymerization of PAN onto the surface of graphene oxide nanosheets, further validating the structural integrity and functionality of the synthesized composite.
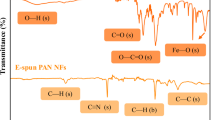
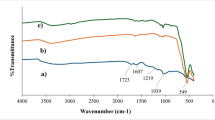
FTIR spectroscopy was used to examine the surface functional groups present in GO and the PAN@PRGO nanocomposite, as illustrated in Fig. 3A. The FTIR spectrum of PAN@PRGO displayed several distinctive new peaks not observed in GO alone. The peak at 1581 cm−1 is associated with the C=N bond vibration, while the 1501 cm−1 peak corresponds to the C=C bond vibration, which is linked to the benzene ring in polyaniline34. The peaks at 1293 cm−1 and 1117 cm−1 are attributed to the C–N stretching vibration of an aromatic secondary amine and the C=N stretching vibration, respectively34,35,36. The reduced intensity of the epoxy peak further supports the partial reduction of GO37. These observations confirm the successful attachment of PAN to the GO nanosheets through electrostatic interactions between the oxygen-containing functional groups on GO and the –C= + N– groups in PAN33,35,37.
X-ray diffraction (XRD) analysis was conducted to characterize the crystalline structure of GO and the PAN@PRGO nanocomposite. In Fig. 3B, the GO pattern shows a strong, sharp peak at 10.8°, corresponding to an interlayer spacing that confirms its nanosheet structure. In contrast, the XRD pattern of PAN@PRGO shows the disappearance of the GO peak at 2θ = 10.8°, indicating the partial reduction of GO functional groups during in-situ polymerization. Four new characteristic peaks at 2θ values of 20.76°, 25.4°, 28.8°, and 30.3°, with respective interlayer spacings of 4.273 Å, 3.503 Å, 3.091 Å, and 2.945 Å, indicate the integration of PAN nanofibers onto the GO surface, forming a crystalline composite structure37,38.
The most common method for surface chemical analysis is XPS, which is used to look at individual elements in GO and PAN@PRGO composites. The formation of PAN@PRGO nanocomposite can also be confirmed by XPS, as shown in Fig. 4. In the XPS survey spectra of GO and PAN@PRGO (Fig. 4B), the intensity of the peak assigned to C1s significantly enhanced from GO to PAN@PRGO while the intensity of the O1 peak decreased from GO to PAN@PRGO. Additionally, a new peak of N1s is clearly observed in the XPS survey spectra of PAN@PRGO. These findings show that PAN nanoparticles were successfully grafted onto GO nanosheet surfaces. Three peaks with binding energies of 288.9 eV (C in C=O), 284.0 eV (Cin C=C, C–H), and 286.8 eV (C in C–O) were deconvoluted from the C1s high-resolution scan of GO33. The C1s high-resolution spectrum (Fig. 4D) related to PAN@PRGO was deconvulated to two peaks with bending energies of about 285.7 eV (C in C–N, C=N), 287.2 eV (C in C–N–C, C=N), and 288.7 eV (C in C=O)33,36.
The O1s high-resolution spectra of GO (Fig. 4F) were deconvoluted into two peaks at binding energies 531.7 and 533.3 eV, corresponding to (O in COOH) and (O in C=O), respectively. The high-resolution scan of the N1s peak of PAN@PRGO (Fig. 4E) was deconvoluted into three peaks at binding energies of 398.8 eV (N in C=N of quinoide), 399.9 eV (N in C–N–C), and another peak at 401.5 eV (N in –NH2)33,36,39. These observations confirm the successful grafting and polymerization of PAN nanoparticles onto the surface of GO nanosheets.
Batch adsorption studies
Influence of pH
The solution pH plays a crucial role in the adsorption process. The effect of pH on IC dye adsorption onto both GO and PAN@PRGO was investigated over a pH range of 2–8 using acetate buffer (pH 3–6) and phosphate buffer (pH 2, 8), as shown in Fig. 5A. The results demonstrated that the extraction efficiency of IC dye increased as the solution pH increased from 2.0 to 5.0, followed by a decline at higher pH values above 5. The optimal pH for IC dye adsorption onto GO and PAN@PRGO was found to be 5, with removal efficiencies of 90.48% and 92.66%, respectively. At low pH values, protonation of the functional groups on the adsorbents (COOH⁺2, O=C–NH⁺) increases the positive charge density on the adsorbent surface, thereby enhancing electrostatic attraction toward the negatively charged IC dye molecules and improving adsorption. However, at pH values above 5.5, adsorption efficiency decreases rapidly. This behavior can be attributed to the electrostatic repulsion between the negatively charged dye molecules and the increasingly negative surface of PAN@ PRGO40,41,42. Furthermore, this trend is consistent with the point of zero charge (pHpzc) of PAN@PRGO, which was determined to be 5.8 (Supporting Information, S2, Fig. S1)43,44,45. Below the pHpzc, the nitrogen- and oxygen-containing functional groups on PAN@PRGO are predominantly protonated, favoring electrostatic interactions with IC dye46. Conversely, at pH values above the pHpzc, the surface of PAN@PRGO becomes negatively charged, significantly reducing the attractive forces between the adsorbent and IC dye molecules, thereby lowering the adsorption efficiency. These findings are in agreement with previous studies43,44,45,47,48.
Impact of IC dye concentration
The influence of the initial concentration on the removal of IC dye using GO and PAN@PRGO was investigated at pH 5 and 25 °C by using 10.0 mg adsorbent dose and 10.0 mL of different IC dye concentrations (10.0 to 500.0 mg L−1) for GO and (10.0 to 800.0 mg L−1) for PANI-GO. Figure 5B showed that by increasing the concentration of IC dye (10.0 to 800.0 mg L−1), qe increased from 10 to 490 mg g−1 for PAN@PRGO and from 10 to 310.0 mg/g for GO. This might be explained by the concentration gradient increasing the driving force, which improved mass movement20,49,50. Additionally, the findings also showed that the materials displayed 100% removal for concentrations up to 300 mg L−1for PAN@PRGO and 100.0 mg L−1for GO.
The Langmuir and Freundlich isotherm models models51,52, described by Eqs. (3), and (4) respectively, are very useful for describing the interaction nature between the adsorbent and adsorbate molecules. Langmuir model proposed monolayer and homogenous adsorption coverage. Whereas the Freundlich model based on heterogeneous active sites with multilayer adsorption.
where qe and qmax are the equilibrium and theoretical adsorption capacity of Langmuir monolayer respectively (mg g−1). Ce is the equilibrium concentration of dye (mg L−1). The parameter Kf (mg L−1) denotes the Freundlich constant related to adsorption capacity, and n represents the heterogeneity factor. The equilibrium adsorption constant is denoted as b (mg L−1) in the Langmuir isotherm model models51,52.
Figure 5C,D reveals the theoretical Freundlich and Langmuir adsorption isotherm which also describe the experimental data.
RL parameter is given by Eq. (5)
Table 1 shows that the correlation coefficient (R2) value for the Langmuir model is around 0.999, and the calculated qm (495.04 mg g−1) is relatively close to the experimental qexp. (490.0 mg g−1). Thus, the IC dye adsorption is more in line with the Langmuir model, indicating a greater propensity for monolayer adsorption in the IC dye adsorption by PAN@PRGO composite. Additionally, Furthermore, RL value range from 0 to 1 (Table 1), suggesting that the IC dye’s adsorption mechanism is favorable53,54,55.
Effect of adsorbent dosage
The effect of adsorbent dosage (0.5 to 3.0 g L−1) on the removal of IC dye using PAN@PRGO was studied at pH 5, temperature 25 °C, IC concentration 600 mg L−1, and agitation time 120 min. Figure 6A showed that the removal efficiency of IC dye by PAN@PRGO increased from 62.82% to 100% by increasing the adsorbent dosage from 0.5 to 3.0 g L−1. This explained that at higher adsorbent dosages, more active sites are available, which boosted the extraction of higher amounts of IC dye56.
Effect of agitation time and adsorption kinetics
The contact time plays an important role in the investigation of the adsorption pathway until it reaches equilibrium and the rate of adsorption, as depicted in Fig. 6B. The results showed that the rate of adsorption of IC dye increased rapidly with increasing agitation time. In the first 60 min, more than 70.56% and 63.08% of IC dye were adsorbed onto PAN@PRGO and GO, respectively. This is due to the availability of a large number of available adsorption sites on the surface of the adsorbent. Additionally, the higher driving force boosted fast transfer of IC dye to the adsorbent surface50. The maximum adsorption of IC was achieved within 90 min at initial IC concentrations of 600 mg L−1 and 500 mg L−1 for PAN@PRGO and GO, respectively.
Two adsorption kinetic models (Fig. 6C,D) were utilized mainly to describe the adsorption process mechanism, pseudo-first-order and pseudo-second order expressed by Eqs. (6), and (7), respectively20,57.
where qe and qt are the adsorption uptake of IC dye at equilibrium and at time t (min) and k2 (g mol−1 min−1) and k1 (min−1) and are the rate constant for pseudo-second order and pseudo first order respectively.
Table 2 summarized all the kinetic parameters calculated for adsorption of IC dye onto PAN@PRGO.The results demonstrated that the experimental values capacities (qe.exp) are in good agreement with calculated maximum uptake capacity using pseudo second order. Furthermore, the correlation coefficient (R2) of pseudo second order (0.999) is higher than those of pseudo first order as described in Fig. 6C,D. These confirmed that the adsorption of IC dye on PAN@PRGO fitted well with pseudo second order kinetic model through the sharing or exchanging of electrons between IC dye and PAN@PRGO composite. The interaction between PANG@PRGO and IC dye molecules was also confirmed by XPS analysis before and after dye adsorption. The XPS survey scans of PAN@PRGO before and after IC dye adsoprion showed two peaks, as shown in Fig. 7, which were caused by C 1s (285.7 eV), O 1s (532.9 eV), and N 1s (400.9 eV). Additionally, the presence of indigo carmine dye was indicated by an extra peak in the XPS survey spectra of PAN@PRGO---IC caused by S 2p (167 eV). These findings support the concept that the indigo carmine attaches to the PAN@PRGO via hydrogen bonding between the IC dye molecules and the –C=NH, –NH, and –C=O groups of the PAN@PRGO as well as electrostatic attraction (Fig. 7C).
Influence of temperature and thermodynamic studies
The effect of temperature parameter on the adsorption capacity of IC dye using PAN@PRGO is presented in Fig. S2A. It utilized that by increasing the temperature from 298 to 328 K, the maximum adsorption capacities of IC dye onto PAN@PRGO adsorbent increased from 490.0 to 530.98 mg g−1 at T = 318 K and from 490 to 590.93 mg.g−1 at T = 328 K. This may be attributed to the increase in the kinetic energy and mobility of IC dye molecules with the temperature2.
The thermodynamic parameters (ΔH°, ΔG°, and ΔS°) were determined by Eqs. (8), (9), and (10).
Van’t Hoff equation
ΔH°, ΔG°, and ΔS° stand for the standard enthalpy change, standard free energy change, and standard entropy change, respectively2. The symbols Kd, T, and R stand for the adsorption coefficient, temperature (k), and gas constant (8.314 J mol−1 K−1), respectively50. To create a dimensionless Kd, use ρ = 1000 g/L. The ΔS° and ΔH° values were calculated using the intercept and slope of the plot of ln Kd vs 1/T (Fig. S2B). The computed thermodynamic functions are summarized in Table 3. An rise in the solid/solute interface’s randomness throughout the adsorption process is examined by the positive entropy. A spontaneous adsorption process is suggested by a decrease in the negative value of free energy (ΔG°), while an endothermic adsorption process is indicated by a positive value of enthalpy (ΔH°).
Reusability of PAN@PRGO adsorbent
The ability of an adsorbent to be reused is one of its most crucial characteristics, as it affects both its long-term and economical viability in industrial applications. Eluent (desorbing agent) 0.1 M NaOH was used, as explained in the experimental section. The PAN@PRGO adsorbent’s regeneration and reusability across seven cycles were described in detail in Fig. 8. After seven cycles, the findings showed that the adsorption efficiency was approximately 93.0%. These results offer important information for the creation of an adsorbent PAN@PRGO with excellent stability, high efficiency, and low cost effectiveness to address dye pollution.
Application in real water samples
To evaluate the performance of PAN@PRGO under optimal conditions, adsorption experiments were conducted using different real water sources, including tap water, Nile water, and sewage water (Fig. 8B). A reference solution containing 100 mg L−1 of IC was prepared for comparison. Water samples were collected from municipal tap water, a designated section of the Nile River, and a local sewage treatment facility for laboratory analysis. The adsorption studies demonstrated that PAN@PRGO maintained a removal efficiency exceeding 99.0% across all tested water matrices (Fig. 8B). Despite the presence of various dissolved minerals and organic pollutants in river and wastewater samples, the adsorbent exhibited consistent performance. All batch adsorption experiments were performed in triplicate, yielding relative standard deviation (RSD) values below 4%, ensuring data reliability. These findings underscore the potential applicability of PAN@PRGO for efficient dye removal in diverse environmental water sources.
Comparison with literature
Table 4 provides a comparison of the qmax values for IC dye adsorption onto PAN@PRGO, GO, and other materials reported in the literature. Both PAN@PRGO and GO demonstrated remarkable adsorption capacities for removing IC dye from aqueous solutions.
Conclusions
The present study elucidates the successful synthesis of PAN@PRGO nanocomposite via in situ polymerization of aniline (AN) onto the surface of GO nanosheets. The PAN@PRGO was fully characterized using different analytical techniques FTIR, XPS, XRD and SEM. The prepared PAN@PRGO nanocomposite exhibited an effective efficiency for the removal of IC dye (490.0 mg/g) via electrostatic attraction and hydrogen bonding between –C=NH, –NH and C=O groups of PAN@PRGO and IC dye molecule. The optimum conditions for the removal of IC dye using PAN@PRGO were pH 5, 1 g/L of adsorbent, and 90 min of room temperature agitation. In addition, the pseudo-second-order model and the Langmuir isotherm model for the adsorption process are followed, suggesting a monolayer adsorption mechanism. Additionally, the outcomes demonstrated a little reduction in adsorption efficiency during seven cycles, suggesting that PAN@PRGO composite might be used as superior adsorbent for the remediation of industrial effluents containing organic dyes.
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Shin, J.-H. et al. Rapid and efficient removal of anionic dye in water using a chitosan-coated iron oxide-immobilized polyvinylidene fluoride membrane. ACS Omega 7, 8759–8766 (2022).
Awad, F. S., AbouZied, K. M., Abou El-Maaty, W. M., El-Wakil, A. M. & El-Shall, M. S. Effective removal of mercury (II) from aqueous solutions by chemically modified graphene oxide nanosheets. Arab. J. Chem. 13, 2659–2670 (2020).
Sayed, N. S. M., Ahmed, A. S. A., Abdallah, M. H. & Gouda, G. A. ZnO@ activated carbon derived from wood sawdust as adsorbent for removal of methyl red and methyl orange from aqueous solutions. Sci. Rep. 14, 5384 (2024).
El-Wakil, A., Abou El-Maaty, W. & Awad, F. Adsorption of methylene blue and rhodamine B from aqueous solutions using dry or carbonized water hyacinth plant. J. Appl. Sci. Res. 9, 3607–3619 (2013).
Ceroni, L. et al. Enhanced adsorption of methylene blue dye on functionalized multi-walled carbon nanotubes. Nanomaterials 14, 522 (2024).
Al-Rubayee, W. T., Abdul-Rasheed, O. F. & Ali, N. M. Preparation of a modified nanoalumina sorbent for the removal of alizarin yellow R and methylene blue dyes from aqueous solutions. J. Chem. 2016, 4683859 (2016).
Harrache, Z., Abbas, M., Aksil, T. & Trari, M. Thermodynamic and kinetics studies on adsorption of Indigo Carmine from aqueous solution by activated carbon. Microchem. J. 144, 180–189 (2019).
Chowdhury, M. F. et al. Current treatment technologies and mechanisms for removal of indigo carmine dyes from wastewater: A review. J. Mol. Liq. 318, 114061 (2020).
Babu, A. N. et al. Removal of hazardous indigo carmine dye from waste water using treated red mud. Mater. Today Proc. 17, 198–208 (2019).
Ristea, M.-E. & Zarnescu, O. Indigo carmine: Between necessity and concern. J. Xenobiot. 13, 509–528 (2023).
De León-Condés, C. A. et al. Sulfonated and gamma-irradiated waste expanded polystyrene with iron oxide nanoparticles, for removal of indigo carmine dye in textile wastewater. Heliyon 5, 1 (2019).
Laing, I. The impact of effluent regulations on the dyeing industry. Rev. Prog. Color. Relat. Top. 21, 56–71 (1991).
Bakry, A. M., Alamier, W. M., El-Shall, M. S. & Awad, F. S. Facile synthesis of amorphous zirconium phosphate graphitic carbon nitride composite and its high performance for photocatalytic degradation of indigo carmine dye in water. J. Market. Res. 20, 1456–1469 (2022).
Fallah, N., Bloise, E., García-López, E. I. & Mele, G. Carbon-based materials in combined adsorption/ozonation for indigo dye decolorization in constrain contact time. Molecules 29, 4144 (2024).
Tien, H. N. et al. Electrochemical degradation of indigo carmine, P-nitrosodimethylaniline and clothianidin on a fabricated Ti/SnO2–Sb/Co-βPbO2 electrode: Roles of radicals, water matrices effects and performance. Chemosphere 313, 137352 (2023).
Zein, R. et al. The improvement of indigo carmine dye adsorption by Terminalia catappa shell modified with broiler egg white. Biomass Convers. Bioref. 13, 13795–13812 (2023).
Ahmed, N. & Mir, F. Q. A comprehensive study on the development of ceramic membranes from natural Kashmir clay and its application in pH-mediated removal of Indigo carmine dye. Emerg. Mater. 1, 1–12 (2024).
Rendón-Castrillón, L. et al. Treatment of water from the textile industry contaminated with indigo dye: A hybrid approach combining bioremediation and nanofiltration for sustainable reuse. Case Stud. Chem. Environ. Eng. 8, 100498 (2023).
Arenas, C. N., Vasco, A., Betancur, M. & Martínez, J. D. Removal of indigo carmine (IC) from aqueous solution by adsorption through abrasive spherical materials made of rice husk ash (RHA). Process Saf. Environ. Prot. 106, 224–238 (2017).
Ghazy, N. M. et al. Enhanced performance of hydroxyl and cyano group functionalized graphitic carbon nitride for efficient removal of crystal violet and methylene blue from wastewater. RSC Adv. 12, 35587–35597 (2022).
Ozalp, O., Oguz, F. & Soylak, M. Activated carbon from green walnut shells as the adsorbent for the solid-phase extraction of indigo carmine from food and textiles. Instrum. Sci. Technol. 53, 167–181 (2025).
Teerasong, S. et al. Preparation of PVP-BiOBr adsorbent for efficient indigo carmine dye removal using flow-circulation systems. ACS Omega 9, 29644–29650 (2024).
Azadikhah, F., Ghaderi, M., Khorsandi, H. & Karimi, A. R. Advanced superparamagnetic cross-linked chitosan-based biosorbent hydrogel for efficient removal of indigo carmine dye from water: Adsorption isotherms, kinetics, and thermodynamic studies. Polym. Adv. Technol. 35, e6513 (2024).
Aissa, M. A. B. et al. Facile synthesis of TiO2@ ZnO nanoparticles for enhanced removal of methyl orange and indigo carmine dyes: Adsorption, kinetics. Heliyon 10, 1 (2024).
Elamin, M. R., Abdulkhair, B. Y. & Elzupir, A. O. Removal of ciprofloxacin and indigo carmine from water by carbon nanotubes fabricated from a low-cost precursor: Solution parameters and recyclability. Ain Shams Eng. J. 14, 101844 (2023).
Alanazi, K. D. et al. Citric acid-cross linked with magnetic metal-organic framework composite sponge for superior adsorption of indigo carmine blue dye from aqueous solutions: Characterization and adsorption optimization via Box-Behnken design. J. Mol. Struct. 1299, 137131 (2024).
Bakry, A. M. et al. Remediation of water containing lead (II) using (3-iminodiacetic acid) propyltriethoxysilane graphene oxide. Sci. Rep. 14, 18848 (2024).
Bakry, A. M. et al. Enhanced performance of amine and thiol chemically modified graphene oxide for effective removal of Hg (II), Pb (II), and Cr (VI) from aqueous solution. Appl. Water Sci. 14, 179 (2024).
Bakry, A. M., Alamier, W. M., Salama, R. S., El-Shall, M. S. & Awad, F. S. Remediation of water containing phosphate using ceria nanoparticles decorated partially reduced graphene oxide (CeO2-PRGO) composite. Surf. Interfaces 31, 102006 (2022).
Zeeshan, M., Shah, J., Jan, M. R. & Iqbal, M. Removal of bisphenol-A from aqueous samples using graphene oxide assimilated magnetic silica polyaniline composite. J. Inorg. Organomet. Polym Mater. 31, 2073–2082 (2021).
Verma, D. K. et al. Superlubricity of nanocomposites of polyaniline-functionalized reduced graphene oxide with yttrium and vanadium-codoped zinc oxide nanoparticles. ACS Appl. Eng. Mater. 2, 156–169 (2024).
Li, R., Liu, L. & Yang, F. Preparation of polyaniline/reduced graphene oxide nanocomposite and its application in adsorption of aqueous Hg (II). Chem. Eng. J. 229, 460–468 (2013).
Awad, F. S. et al. Polyacrylonitrile modified partially reduced graphene oxide composites for the extraction of Hg (II) ions from polluted water. J. Mater. Sci. 56, 7982–7999 (2021).
Chauhan, N. P. S. et al. High-performance supercapacitors based on polyaniline–graphene nanocomposites: Some approaches, challenges and opportunities. J. Ind. Eng. Chem. 36, 13–29 (2016).
Liu, Y., Chen, L., Li, Y., Wang, P. & Dong, Y. Synthesis of magnetic polyaniline/graphene oxide composites and their application in the efficient removal of Cu (II) from aqueous solutions. J. Environ. Chem. Eng. 4, 825–834 (2016).
Chinnathambi, A. & Alahmadi, T. A. Facile synthesis of Fe3O4 anchored polyaniline intercalated graphene oxide as an effective adsorbent for the removal of hexavalent chromium and phosphate ions. Chemosphere 272, 129851 (2021).
Ramezanzadeh, M., Asghari, M., Ramezanzadeh, B. & Bahlakeh, G. Fabrication of an efficient system for Zn ions removal from industrial wastewater based on graphene oxide nanosheets decorated with highly crystalline polyaniline nanofibers (GO-PANI): Experimental and ab initio quantum mechanics approaches. Chem. Eng. J. 337, 385–397 (2018).
Pawar, D., Malavekar, D., Lokhande, A. & Lokhande, C. Facile synthesis of layered reduced graphene oxide/polyaniline (rGO/PANI) composite electrode for flexible asymmetric solid-state supercapacitor. J. Energy Storage 79, 110154 (2024).
Ahuja, P., Ujjain, S. K., Arora, I. & Samim, M. Hierarchically grown NiO-decorated polyaniline-reduced graphene oxide composite for ultrafast sunlight-driven photocatalysis. ACS Omega 3, 7846–7855 (2018).
Hashem, H. M., El-Maghrabey, M. & El-Shaheny, R. Inclusive study of peanut shells derived activated carbon as an adsorbent for removal of lead and methylene blue from water. Sci. Rep. 14, 13515 (2024).
Aravindhan, S., Kumar, G. B., Saravanan, M. & Arumugam, A. Delonix regia biomass as an eco-friendly biosorbent for effective Alizarin Red S textile dye removal: Characterization, kinetics, and isotherm studies. Bioresour. Technol. Rep. 25, 101721 (2024).
Al-Qarhami, F., Khalifa, M. E., Abdallah, A. & Awad, F. S. Remediation of wastewater containing methylene blue and acid fuchsin dyes using 2-aminothiazole chemically modified chitosan. Int. J. Biol. Macromol. 303, 140744 (2025).
Ai, L., Zhang, C. & Meng, L. Adsorption of methyl orange from aqueous solution on hydrothermal synthesized Mg–Al layered double hydroxide. J. Chem. Eng. Data 56, 4217–4225 (2011).
Yang, L., Shahrivari, Z., Liu, P. K., Sahimi, M. & Tsotsis, T. T. Removal of trace levels of arsenic and selenium from aqueous solutions by calcined and uncalcined layered double hydroxides (LDH). Ind. Eng. Chem. Res. 44, 6804–6815 (2005).
Das, D. P., Das, J. & Parida, K. Physicochemical characterization and adsorption behavior of calcined Zn/Al hydrotalcite-like compound (HTlc) towards removal of fluoride from aqueous solution. J. Colloid Interface Sci. 261, 213–220 (2003).
Pourebrahim, F., Ghaedi, M., Dashtian, K., Heidari, F. & Kheirandish, S. Simultaneous removing of Pb2+ ions and alizarin red S dye after their complexation by ultrasonic waves coupled adsorption process: Spectrophotometry detection and optimization study. Ultrason. Sonochem. 35, 51–60 (2017).
Waly, S. M., El-Wakil, A. M., Abou El-Maaty, W. M. & Awad, F. S. Remediation of water containing mercury (ii) using poly-2-aminothiazole intercalated α-zirconium phosphate nanoplates. N. J. Chem. 49, 1314–1324 (2025).
Moheb, M., El-Wakil, A. M. & Awad, F. S. Highly porous activated carbon derived from the papaya plant (stems and leaves) for superior adsorption of alizarin red s and methylene blue dyes from wastewater. RSC Adv. 15, 674–687 (2025).
Sillanpää, M., Mahvi, A. H., Balarak, D. & Khatibi, A. D. Adsorption of Acid orange 7 dyes from aqueous solution using Polypyrrole/nanosilica composite: Experimental and modelling. Int. J. Environ. Anal. Chem. 103, 212–229 (2023).
Waly, S. M., El-Wakil, A. M., Abou El-Maaty, W. M. & Awad, F. S. Efficient removal of Pb (II) and Hg (II) ions from aqueous solution by amine and thiol modified activated carbon. J. Saudi Chem. Soc. 25, 101296 (2021).
Alrefaee, S. H. et al. Adsorption and effective removal of organophosphorus pesticides from aqueous solution via novel metal-organic framework: Adsorption isotherms, kinetics, and optimization via Box-Behnken design. J. Mol. Liq. 384, 122206 (2023).
Awad, F. S., AbouZeid, K. M., El-Maaty, W. M. A., El-Wakil, A. M. & El-Shall, M. S. Efficient removal of heavy metals from polluted water with high selectivity for mercury (II) by 2-imino-4-thiobiuret–partially reduced graphene oxide (IT-PRGO). ACS Appl. Mater. Interfaces 9, 34230–34242 (2017).
Waly, S. M., El-Wakil, A. M., Abou El-Maaty, W. M. & Awad, F. S. Hydrothermal synthesis of Mg/Al-layered double hydroxide modified water hyacinth hydrochar for remediation of wastewater containing mordant brown dye. RSC Adv. 14, 15281–15292 (2024).
Bulin, C., Guo, T., Zheng, R. & Xiong, Q. Interaction mechanism of phytic acid functionalized graphene oxide with ionic dyes. Sep. Purif. Technol. 330, 125369 (2024).
Bulin, C. et al. High efficiency removal of methyl blue using phytic acid modified graphene oxide and adsorption mechanism. Spectrochim. Acta A Mol. Biomol. Spectrosc. 307, 123645 (2024).
Alsuhaibani, A. M., Alayyafi, A. A., Albedair, L. A., El-Desouky, M. G. & El-Bindary, A. A. Synthesis and characterization of metal–organic frameworks based on thorium for the effective removal of 2, 4-dichlorophenylacetic pesticide from water: Batch adsorption and Box-Behnken Design optimization, and evaluation of reusability. J. Mol. Liq. 398, 124252 (2024).
Danish, M. et al. Application of optimized large surface area date stone (Phoenix dactylifera) activated carbon for rhodamin B removal from aqueous solution: Box-Behnken design approach. Ecotoxicol. Environ. Saf. 139, 280–290 (2017).
Asadi, E., Haroonian, P., Ghaedi, M. & Asfaram, A. Removal of tartrazine and indigo carmine with layered double hydroxide-modified graphite nanocomposite: Isotherm, kinetics and response surface methodology (RSM) optimization. J. Mol. Liq. 402, 124769 (2024).
Suvaitha, S. P. & Venkatachalam, K. Adsorptive separation of food and textile dyes from aqueous solution by SBA-15 supported polyaniline/polypyrrole composite: Isotherms, kinetics, thermodynamics and recyclability study. J. Mater. Sci. Mater. Electron. 35, 94 (2024).
Shabani, S., Mirkazemi, S. M., Mohammadi, B., Magni, M. & Trasatti, S. Spherical gamma-alumina macroscopic beads as easy to remove adsorbents for water remediation: Modeling of indigo carmine case study. Appl. Surf. Sci. 676, 161019 (2024).
Zolgharnein, J., Rajabalipour, F. & Dermanaki Farahani, S. Indigo carmine dye adsorptive removal by polyethylene glycol-modified hydroxyapatite nanoparticles as an efficient adsorbent. Water Air Soil Pollut. 234, 210 (2023).
Ahmed, M. A., Ahmed, M. A. & Mohamed, A. A. Removal of 4-nitrophenol and indigo carmine dye from wastewaters by magnetic copper ferrite nanoparticles: Kinetic, thermodynamic and mechanistic insights. J. Saudi Chem. Soc. 27, 101748 (2023).
Jamil, S. et al. Photocatalytic degradation of indigo carmine dye by hydrothermally synthesized graphene nanodots (GNDs): Investigation of kinetics and thermodynamics. RSC Adv. 14, 23973–23986 (2024).
Adam, F. A. et al. Enhanced adsorptive removal of indigo carmine dye by bismuth oxide doped MgO based adsorbents from aqueous solution: Equilibrium, kinetic and computational studies. RSC Adv. 12, 24786–24803 (2022).
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Saadia M. Waly contributed to methodology development, investigation, formal analysis, data validation, and curation, as well as drafting the original manuscript. Ahmad M. El-Wakil oversaw the project, managed funding acquisition, and participated in manuscript review and editing. Mohamed M. Waly and Weam M. Abou El-Maaty provided supervision, validation, and support in securing funding. Fathi S. Awad played a key role in conceptualization, investigation, supervision, methodology design, formal analysis, and manuscript preparation, including both drafting and critical revisions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Waly, S.M., El-Wakil, A.M., Waly, M.M. et al. Enhanced removal of indigo carmine dye from aqueous solutions using polyaniline modified partially reduced graphene oxide composite. Sci Rep 15, 15555 (2025). https://doi.org/10.1038/s41598-025-98115-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-98115-8
Keywords
This article is cited by
-
Eco-friendly tassel-derived activated carbon for efficient dye removal in wastewater treatment
Scientific Reports (2025)