Abstract
Cocaine use disorder (CUD) is a worldwide health problem with limited effective treatment options. The therapeutic potential of repetitive transcranial magnetic stimulation (rTMS) is gaining more attention following evidence of its role on craving reduction in CUD. However, the heterogeneity of results underscores a pressing need for biomarkers of treatment outcome. We asked whether brain connectomics together with clinical assessments can predict response to add-on rTMS therapy for CUD better than solely conventional clinical assessments. Forty-four randomly assigned CUD patients underwent the 2-week double-blind acute phase [Sham (n = 20, 2f./18m.) and Active (n = 24, 4f./20m.)], in which they received 2 daily sessions of rTMS on the left dorsolateral prefrontal cortex. Subsequently, 19 and 14 patients continued to an open-label maintenance phase of two weekly rTMS sessions for 3 and 6 months, respectively. Pre and post treatment resting-state brain functional connectivity as well as two clinical scores of craving were measured to predict the subsequent response to rTMS therapy. Two conventional clinical scores, namely Cocaine Craving Questionnaires (CCQ) and Visual Analogue Scale (VAS) were used as craving level assessments. We used a priori seed-driven connectivity of Left Dorsolateral Prefrontal Cortex (LDLPFC) and Anterior Cingulate Cortex (ACC) together with the connectivity from a whole-brain multi-voxel pattern analysis at each time point to predict the reduction in craving after rTMS. The combination of connectivity changes and baseline craving severity improved the prediction of individual craving compared to the prediction with only the initial craving severity. The predictive model from the combination of neuromarkers could explain 45 to 97 percent of variance in craving changes assessed by two different clinical scores. We used leave-one-subject-out cross-validation to support the generalizability of our findings. Our results indicate that employing neuromarkers from resting-state functional connectivity of pre and post condition of CUD patients receiving add-on rTMS therapy increases the power of predicting craving changes and support the idea that neuromarkers may offer improvements in precision medicine approaches.
Similar content being viewed by others
Introduction
Cocaine use disorder (CUD) is considered a worldwide public health problem, with significant contribution to over 12% of mortality, and one of the leading causes of preventable death1. This disorder is characterized by patterns of compulsive substance searching, intense craving, impulsive decision making, and psychological behaviors such as depression and anxiety2,3,4. Despite the significant morbidity associated with CUD, there is not yet an effective psychological or pharmacological therapy for it. In recent years, transcranial magnetic stimulation (TMS) has emerged as a non-invasive neuromodulating approach which had positive outcomes in substance use disorders5,6,7,8,9,10,11,12. Likewise, repetitive transcranial magnetic stimulation (rTMS) and theta burst stimulation (TBS) have been used as add-on therapies for treatment of CUD and showed promising outcomes such as reducing craving and substance use9,13,14,15.
Craving is a core clinical symptom of cocaine addiction that can be addressed as treatment target. Craving is defined as a pressing and urgent desire for an addictive behavior, motivated by internal and external cues1. There are currently various experimental subjective measurements of this state: cocaine craving questionnaires (CCQ), visual analogue scale (VAS), and non-verbal physiological measures16,17. Substance craving is an important risk factor of relapse in cocaine addiction18,19, and therefore, reducing craving has been targeted to curb the addictive behavior1.
Neuroimaging studies in humans have shown that craving is mainly involved in the activation of reward and motivation circuits as well as executive control network (ECN)7,8. The ECN most prominently involves the prefrontal cortex (PFC), including dorsolateral prefrontal cortex (DLPFC) and anterior cingulate cortex (ACC)20. It has been shown that chronic cocaine use can cause damages to the structure and function of the prefrontal cortex (PFC). Previous studies reported the microstructural changes in frontostriatal circuits21 and the impairment of the executive control functions9 in CUD. There is also evidence of the gray matter density increase in frontal and temporal regions of the brain after 1 month of abstinence in CUD22. In this regard, the left dorsolateral prefrontal cortex (LDLPFC) has been considered as a possible target for the application of rTMS in CUD patients. LDLPFC is one of the main hubs of the executive control network in the human brain 8 and plays a critical role in the addictive cycle, craving and impulsive behavior5,23. The application of rTMS to the left dorsolateral prefrontal cortex has shown promising outcomes in CUD such as reduction in cocaine use and symptoms9,11,24.
Although rTMS of the LDLPFC could be considered as an additional treatment in CUD, the ability to predict the heterogenous treatment responses is still a challenge. Identifying clinically meaningful and accessible predictors can help with the prediction of treatment response in different individuals as well as the overall treatment design. Several biomarkers achieved from the brain neuroimaging studies (neuromarkers) have previously shown the power in predicting the treatment response in brain disorders25,26,27,28,29,30. Recent studies suggest that neuromarkers can provide higher predictive power than the conventional clinical and demographic measures in rTMS outcomes31,32,33. Among these biomarkers, there are well-replicated evidence of correlational relationship between neuromarkers in LDPFC and ACC and clinical response in CUD7,12,34.
In this context, in the current study, we asked whether the neuromarkers from magnetic resonance imaging can help to predict the short and long-term response to rTMS in CUD. The previous analysis on the current dataset, had focused on the changes of resting state connectivity only in acute phase, and compared the changes in the clinical scores between the baseline, and short and long time points12. However, the power of connectivity changes in predicting the clinical outcomes has not been studied yet. In this work, we are particularly interested in the predictors from resting-state functional magnetic resonance imaging (rsfMRI), for predicting the treatment outcomes, both in short and long periods during the treatment. We aim to investigate the power of univariate as well as multivariate connectomics from rsfMRI in prediction of short term and long term rTMS impacts in CUD. First, as the LDLPFC and ACC have been regarded as the loci of dysfunction inside the ECN in CUD7,35, we hypothesized that rTMS outcome would be associated with the seed-driven resting state functional connectivity of these regions. Then, in a data-driven approach, we examined the power of the predictors from resting-state multi-voxel pattern analysis (MVPA) in predicting the rTMS response. Treatment outcome in our study was considered as pre-post change in craving measures (CCQ and VAS), for three time points during the treatment – two weeks, three months, and six months after the start of the treatment.
Materials and methods
Participants and experimental procedure
The data used in this study was acquired and has been made available on OpenNeuro (https://doi.org/10.18112/openneuro.ds003037.v2.1.0.). This data was obtained from a clinical trial conducted at the Clinical Research Division of the National Institute of Psychiatry in Mexico City, Mexico4.
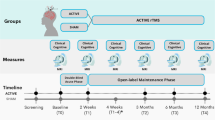
The original study on this dataset4,12 was designed with 44 CUD patients that first underwent a 2-week double-blind randomized controlled trial, with 24 patients in the active group and 20 patients in the sham group. Specifically, each patient received 2 daily sessions of 5-Hz rTMS (5,000 pulses) on the left DLPFC for two weeks. After the 2-week acute phase, 12 patients from the sham group underwent open-label rTMS sessions for 2 weeks. Therefore, 36 patients in total received active rTMS for 2 weeks as in open-label phase (24 + 12: 36 patients in total at baseline and 2 weeks). Following this phase, 19 and 14 patients out of all patients participated in twice-a-week maintenance phase of rTMS application for 3 months, and 6 months, respectively. Here, we aim to consider patients from both phases who received rTMS during short and long periods, and investigate the effects of the rTMS application in all time intervals.
The cocaine users were included in the study based on the criteria: 1) age 18–50 years, and 2) high cocaine consumption for at least 1 year. Clinical and MRI data were obtained for each patient at baseline (bs), after 2 weeks (2w), after 3 months (3m), and after 6 months (6m). An overview of the demographic data is shown in Table 1. Detailed information about recruitment, study design, and a complete demographics can be found in previous publications by Garza-Villarreal et al.4,12.
Clinical assessment
The primary clinical score for outcome measurement was craving, including Cocaine Craving Visual Scale and Cocaine Craving Questionnaire Now as described below.
Cocaine Craving Visual Scale (VAS): This score is used to evaluate the patients’ craving at the moment of assessment. A 10-cm line with “no craving” and “the most intense craving” at its ends, was shown to the patients, and they were asked to mark their craving level at the moment of assessment.
Cocaine Craving Questionnaire Now (CCQ-N): The CCQ is a 45-item questionnaire that explores cocaine craving among patients. The CCQ-Now asks participants about their craving at the moment of assessment. The items are related to the following contents: desire to use cocaine, intention and planning to use cocaine, anticipation of positive outcome, the anticipation of relief from withdrawal or dysphoria, and lack of control overuse4.
The detailed statistics of clinical measurements are provided in Table 1 and 2.
Transcranial magnetic stimulation
During the 10 weekdays of acute phase, 5-Hz rTMS was delivered every day, using a MagPro R30 + Option magnetic stimulator and a figure-eight B65-A/P coil (MagVenture, Alpharettta, GA). Stimulation was delivered with 50 trains at 5Hz at 100% motor threshold to the left DLPFC. To identify the cortical target for stimulation, vitamin E capsule fiducials were used during the MRI acquisition4.
Imaging information
MRI scans were acquired using a Philips Ingenia 3T MR system (Philips Healthcare, Best, The Netherlands, and Boston, MA, USA), with a 32-channel Head coil.
T1-Weighted (T1w) images were acquired using a three-dimensional FFE SENSE sequence, TR/TE = 7/3.5 ms, filed of view = 240 mm2, matrix = 240 × 240 mm, number of slices = 180, and voxel size = 1 × 1 × 1 mm. Resting state functional MRI (rsfMRI) sequences were acquired using a gradient recalled (GE) echo planner imaging (EPI) sequence with 36 axial slices with the following parameters: TR = 2000 ms, TE = 30.001 ms, flip angle = 75°, matrix = 80 × 80 × 37, FOV = 240 mm2, and voxel size of 3 × 3 × 3.3 mm. The whole scanning last for 10 min and a total of 300 volumes were acquired. During this time, the participants were asked to keep their eyes open, and relax, not thinking about anything in particular4.
Preprocessing of anatomical images
T1w image of each subject was preprocessed using the SPM-based CONN Toolbox36 (RRID:SCR_009550), release 20.b37 (conn-toolbox.org). First, each subject’s T1w images were segmented for Gray/White/CSF parts and then were normalized to the standard MNI space. Then, the standard Harvard–Oxford Cortical atlas (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/Atlases) was registered to each scan to obtain the connectivity measures for each subject and each region.
Preprocessing of resting state data
Resting state fMRI data were preprocessed using the default preprocessing pipeline in CONN. Specifically, spatial preprocessing of functional volumes included slice timing correction, realignment, normalization, and smoothing (8 mm FWHM Gaussian filter), using SPM8 (Wellcome Department of Imaging Neuroscience, London, UK; http://www.fil.ion.ucl.ac.uk/spm). To address potential spurious correlations in resting-state networks caused by head motion, we used the Artifact Detection Tools (ART, http://www.nitrc.org/projects/artifact_detect). Specifically, an image was defined as an outlier image if the head displacement in x, y, or z direction was greater than 0.5 mm from the previous frame, or if the global mean intensity in the image was greater than 3 standard deviations from the mean image intensity for the entire resting scan. Anatomical volumes were segmented into grey matter, white matter, and CSF areas, and the resulting masks were eroded (one voxel erosion) to minimize partial volume effects. The temporal timeseries characterizing the estimated participant motion (3 rotation and 3 translation parameters, plus another 6 parameters representing their first-order temporal derivatives) and artifactual covariates (one covariate per artifactual time point consisting of 0’s everywhere and a “1” for the artifactual time point), as well as the blood oxygen level–dependent (BOLD) timeseries within the participant-specific white matter mask (5 PCA parameters) and CSF mask (5 PCA parameters), were used as temporal covariates and removed from the BOLD functional data using linear regression, and the resulting residual BOLD timeseries were band-pass filtered (0.008 Hz < f < 0.09 Hz). The same preprocessing steps were done for the MRI data from two conditions (bs and 2w/3m/6m).
Processing
Baseline craving severity as well as the rsfMRI data were obtained for each subject at baseline, and three time points during the treatment – 2 weeks, 3 months, and 6 months after the start of the treatment. All rsfMRI and anatomical images first passed through preprocessing steps. The ultimate goal of our study was to extract connectomics measures from rsfMRI and use them as predictors in a response prediction model. In this regard, we performed two main analyses to extract the predictors, namely seed-based connectivity analysis (SBC) and whole brain multi-voxel pattern analysis (MVPA).
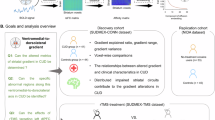
LDLPFC and ACC were chosen as seeds in SBC analysis. Next, second-level group analysis was performed in CONN toolbox to obtain connectomics-based predictors from each analysis. The significant cluster of voxels which had the largest correlation between the connectivity changes and craving changes were identified. We employed leave-one-subject-out cross-validation in our study to increase the generalizability of the results. The two neuromarkers (SBC-driven and MVPA-driven) along with the baseline craving scores were entered into a linear ordinary least square model to predict the changes in craving scores. The performance of the predictive models was assessed using adjusted R-squared and mean absolute error. The methodological overview of the processing is displayed in Fig. 1.
-
1)
Clinical data assessment
Overview of the processing pipeline. CUD patients were randomly selected for rTMS therapy. Baseline craving severity as well as rsfMRI data obtained for each subject at baseline, and three time points during the treatment – 2 weeks, 3 months, and 6 months after the start of the treatment. Following preprocessing, two analysis including seed-based connectivity (SBC) of LDLPFC and ACC, as well as whole brain multi-voxel pattern analysis (MVPA) followed by second-level group analysis were performed in CONN toolbox to obtain connectomics-based predictors at each time point. The significant cluster of voxels which had the largest correlation between the connectivity changes and craving changes were identified. Leave-one-subject-out cross-validation was employed here to increase the generalizability of the results. The combination of the two neuromarkers together with baseline craving scores were entered into a linear ordinary least square model to predict the changes in craving scores. The performance of predictive models was assessed using adjusted R-squared and mean absolute error. VAS: visual analog scale; CCQ-N: cocaine craving questionnaire now; bs: baseline; 2w: 2 weeks; 3m: 3 months; 6m: 6 months.
To test whether the clinical scores related to craving severity are statistically different among groups and time points, we performed a t-test analysis for comparing the mean score values between each of the two time points.
-
2)
Baseline craving severity
The baseline severity of clinical condition of neuropsychiatric patients is a conventional predictor of individual responsivity to different treatments in psychiatric disorders25,31,32,38. To decide for the inclusion of baseline craving score in prediction model, we first examined the correlational relationship between baseline VAS and CCQ-N and the changes in these scores at three time points during the treatment.
-
3)
Seed-based analysis
Using Seed-to-voxel bivariate correlation analysis in CONN39, the correlation maps of left DLPFC as well as ACC with the rest of the brain voxels were obtained. Specifically, the first-level correlation maps of the seed are produced by computing the Pearson’s correlation coefficients between the BOLD time course of the seed and the time courses of all other brain voxels. The resultant correlation coefficients – r-maps – were then converted to z-scores using Fisher transformation, and submitted to the second-level General Linear Model analysis. Here, we defined the average stimulation target cone mask4 as the seed mask for LDLPFC. This mask has a large number of overlapping voxels with the left DLPFC (Brodmann areas 9 and 46), the major locus of dysfunction in CUD7. The mask of ACC – extracted from the Harvard–Oxford Cortical atlas (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/Atlases) – was selected as the other seed in our SBC analysis.
Fisher transformed r-maps of the seed regions were used in a second-level analysis of covariance (ANCOVA) to regress the changes in DLPFC/ACC connectivity (pre minus post) based on the individual change in the clinical score (pre minus post condition) as a covariate of interest, controlling for the score at baseline (testing whether the correlation between connectivity-change and score-change differ from zero after controlling for the influence of baseline score value). The predictive model is compared to the null model using a standard likelihood ratio test. Results are estimated using height threshold p < 0.01 and cluster-level threshold p < 0.01.
We performed a leave-one-subject-out cross-validation (LOSOCV) to extract the connectivity values of the seed with the largest significant cluster of voxels in the brain. This would help to have a robust predictive model with the ability to be generalized to new cases. Specifically, in each iteration, the data from N-1 subjects were used to estimate the first largest cluster of voxels that has significant association with changes in the craving scores. Then, the average connectivity within this cluster was computed for the left-out subject, and this procedure was repeated for each participant. Doing so, the potential biases from selected voxels in the significant cluster is minimized25,40.
-
4)
Multi-voxel pattern analysis
To extract the neuromarkers, we did not limit our analysis to an a priori seed-driven approach, and to maximize clinical prediction, we also used a data-driven agnostic approach known as multi-voxel pattern analysis (MVPA)41 in CONN42. First, the pairwise connectivity pattern between each voxel of the brain mask and all other voxels of the brain were computed. Then, the dimensionality of this multi-voxel pattern was reduced using principal component analysis (PCA), to maximize the explained variability of the extracted patterns with a lower number of spatial components. Here, we selected only the first of four main components. Next, we performed multivariate analysis to investigate the relationship between this component’s connectivity changes and the changes in the clinical scores, controlling for the baseline scores. The first largest cluster whose connectivity patterns were significantly correlated with craving changes was identified (height threshold p < 0.01; cluster-level threshold p < 0.01). The same LOSOCV procedure as in SBC analysis was used to extract subject-specific cluster masks and MVPA connectivity values, iteratively for each subject.
-
5)
Statistical analyses
To assess whether baseline craving score can predict change in craving score, we used independent linear least square models with baseline score values as predictors. To test whether functional connectivity changes from SBC and MVPA analysis can predict the changes in craving scores, we ran linear regression models using these connectomics measures together with baseline score as predictors.
For all predictive models, we used statistical ordinary least squares (OLS) model in python. To numerically assess the quality of the predictive models, we used R2 values. These values represent the explained percent variance in score changes. Here, we calculated Adjusted R2 (Adj. R2) which minimizes the biases due to model selection, and helps to better compare the model with different numbers of the degrees of freedom and predictors43. The mean absolute error (MAE) represents the difference between the observed and predicted values, i.e., craving changes. In deciding between different models, the model with higher R2 and lower MAE is more favorable.
LOSOCV of SBC and MVPA correlation analysis were performed in MATLAB using “spm_crossvalidation” code provided with CONN toolbox. All other statistics were performed in python using statsmodels module version 0.14.0.
Ethics approval
All the procedures were approved by the Ethics Committee of the Instituto Nacional de Psiquiatría “Ramón de la Fuente Muñiz” (CEI/C/070/2016), and registered at ClinicalTrials.gov (NCT02986438)4.
Consent to participate
The study was approved by the local ethics committee and performed at the Instituto Nacional de Psiquiatría ‘Ramón de la Fuente Muñiz’ in Mexico City, Mexico. All participants provided verbal and written informed consent before participation in any study procedures, all of which conformed to the Declaration of Helsinki for research involving human subjects.
Results
Clinical data assessments
Raincloud plots for distribution of clinical scores at different time points are demonstrated in Fig. 2 (acute phase), and Fig. 3 (open-label phase). The t-test analysis for comparing the mean score values between each of the two time points (baseline vs 2w/3m/6m) are given in Tables 3 and 4. It is evident from the results in Table 3 that both craving scores have significantly different means (p-value < 0.05) between the two conditions for active compared to sham in the acute phase. Results in Table 4 also show that except for VAS at 6m, both scores are statistically different between baseline and each time point. The differences in craving scores among different time points raised this question that whether the changes in craving scores can be related to changes in brain connectomics.
Baseline craving severity
Pearson’s correlation values between baseline VAS and CCQ-N and changes in these scores at three time points during the treatment are presented in Fig. 4 and 5. The results indicate the highly linear relationship between baseline and changes in scores, especially for scores after 3 months. In this regard, we hypothesized that the baseline craving severity can explain the variability in this score, and used it as one of the predictors in our response prediction model.
Resting-state functional magnetic resonance imaging
-
1) LDLPFC/ACC seed‑based correlation analysis
Seed-based analysis was performed with LDLPFC and ACC as seeds. The mask of these regions is illustrated in Fig. 6.
Illustration of left DLPFC seed region (left) and ACC seed region (right). Maps created using CONN toolbox (release 20b37, conn-toolbox.org).
The seed-to-voxel analysis resulted in multiple clusters with significant differences in the correlations of LDLPFC and ACC between bs and 2w/3m/6m conditions which were associated with the changes in the VAS and CCQ-N scores, respectively. The first largest significant clusters obtained from this analysis are presented in Table 5 (See Figure S1) (cluster-level p < 0.01; voxel threshold p < 0.01).
-
2) MVPA analysis
The whole brain MVPA analysis resulted in multiple clusters with significant difference in connectivity of the first component between bs and 2w/3m/6m conditions that was associated with the changes in VAS and CCQ-N scores. The largest significant clusters are presented in Table 6 (See Figure S2) (voxel threshold p < 0.01, cluster threshold p < 0.01).
Prediction model for craving score changes
We used ordinary least square models for prediction of changes in clinical scores between the baseline and 2w/3m/6m conditions. Specifically, for each of the VAS and CCQ-N scores, the connectivity from the SBC and MVPA analysis together with the baseline scores were used in a linear model to explain the variance in the score changes.
The detailed statistics of each model for both craving scores is specified in Tables 7 (VAS) and 9 (CCQ-N) (acute phase) and Tables 8 (VAS) and 10 (CCQ-N) (open-label phase).
Scatter plots for VAS values computed from the predictive model and the scatter plots of the predicted versus observed values are shown in Fig. 7. The similar scatter plots for CCQ-N changes are shown in Fig. 8.
Discussion
A. Overview
The aim of this study was to determine whether changes in resting-state functional connectivity (rsFC) in brain regions associated with Cocaine Use Disorder (CUD) could explain the changes in craving scores in cocaine users receiving add-on repetitive Transcranial Magnetic Stimulation (rTMS) therapy, over both short-term and long-term periods. We used neuromarkers derived from resting-state functional MRI (rsfMRI) through both seed-based and data-driven approaches to predict the response to rTMS treatment. We assessed whether the combination of these neuromarkers and baseline severity could better predict the variability in craving than using baseline clinical scores alone. Predictions were made using clinical and neuroimaging data at three time points during rTMS treatment: 2 weeks, 3 months, and 6 months. Two cocaine craving scores were used as clinical measures: Visual Analogue Scale (VAS) and Cocaine Craving Questionnaire-Now (CCQ-N).
B. Baseline craving
Initial assessments showed a high correlation between changes in craving scores and their baseline values. Correlational analysis at acute phase (Fig. 4) suggests that baseline scores are strong predictors of changes in VAS scores, especially under active conditions. However, the effect of the intervention on CCQ-N scores is less predictable, with weaker correlations observed, particularly in the active condition.
Analysis during the open-label phase (Fig. 5) showed that baseline scores are strong predictors of changes in both VAS and CCQ-N scores over time. The VAS scores exhibit very high predictability, particularly at 3 months. The CCQ-N scores also show strong predictability, albeit with slightly lower correlations. This indicates that higher baseline scores are consistently associated with greater improvements in both VAS and CCQ-N scores at various time points. Quantitatively, baseline VAS accounts for 58%, 95%, and 44% of the variance in VAS changes after 2 weeks, 3 months, and 6 months of rTMS application, respectively (Table S1). Similarly, baseline CCQ-N accounts for 27%, 61%, and 43% of the variance in CCQ-N changes after 2 weeks, 3 months, and 6 months of rTMS, respectively (Table S2).
C. SBC results
Seed-driven connectivity changes in regions implicated in CUD and craving, were chosen as one of the predictors in our prediction model. Given the frequent evidence of dysfunction in the anterior cingulate cortex (ACC) and left dorsolateral prefrontal cortex (LDLPFC) in craving and CUD9,10,44,45,46,47 we got motivated to consider seed-based connectivity changes of these two regions as one of the connectomics-based predictors. ACC seed-based connectivity was used for VAS prediction model. Multiple studies have consistently reported the structural and functional changes of ACC in CUD48,49 and also its impacts on VAS50. As a more accurate technique with detailed scales for assessing craving level, we chose CCQ_Now. Seed-based connectivity of LDLPFC were used in prediction model for CCQ-N. We hypothesized that CCQ_N would be more related to the connectivity of DLPFC, the well-recognized loci of dysfunction in CUD.
SBC analysis for both VAS and CCQ-N showed significant brain activity changes under sham and active conditions, with more extensive and pronounced changes observed in the active conditions (Table 5). The effects of the intervention are evident from early time points (2 weeks) and become more widespread and sustained over time (3 and 6 months) (Figure S1). These effects evolve over time, with early changes observed in visual and associative areas, mid-term changes reflecting broader neural adaptations, and long-term changes indicating sustained effects in executive control and cognitive regions. The significant clusters under sham conditions may suggest placebo effects or natural variability in brain activity.
SBC analysis for open-label phase showed that the connectivity of ACC with precentral gyrus, occipital cortex, and pre/post-central gyrus predicted greater reduction in VAS after 2w, 3m, and 6m application of rTMS, respectively. Connectivity of LDLPFC with the cluster including precuneus, thalamus, nucleus accumbens, and posterior cingulate gyrus predicted greater response to rTMS after 2 weeks. The corresponding clusters after 3, and 6 months contained regions from left and right cerebellum and lateral occipital, respectively. The findings from these significant clusters are consistent with evidence from prior studies which indicate the role of regions from frontal lobe35, nucleus accumbens11,46, occipital-temporal35,46,51, and cerebellum44,52,53 in craving as well as cocaine addiction.
The choice of the ACC for the VAS analysis and the DLPFC for the CCQ-N analysis was guided by previous literature highlighting the distinct but complementary roles of these regions in drug-craving behaviors54. ACC has been extensively associated with the emotional and subjective components of craving, which aligns well with the VAS—a single-item scale designed to capture momentary subjective craving intensity55. In contrast, DLPFC is known to play a critical role in cognitive control, decision-making, and the regulation of craving56,57 processes that are more comprehensively assessed by the CCQ, a multi-item questionnaire which captures a broader and more stable measure of craving across multiple domains. Given that the DLPFC is the main target of rTMS in addiction, we hypothesized that its connectivity would be more relevant to the multidimensional nature of craving assessed by the CCQ.
While we did not initially have a priori expectations regarding the exact differences between the two measures, it is important to note that the VAS and the CCQ measure slightly different aspects of craving. The VAS reflects transient and immediate craving states17, while the CCQ captures trait-like and multi-dimensional craving, encompassing cognitive, emotional, and behavioral aspects. This distinction likely explains why specific ROIs (ACC vs DLPFC) showed stronger associations with one measure over the other.
D. MVPA results
MVPA analysis examined whole-brain voxels without any a priori seed, considering connectivity between all voxel pairs in the brain. Active conditions led to significant cerebellar activation in both VAS and CCQ-N metrics compared to sham conditions, suggesting executive control and coordination involvement under the intervention (Table 6).
Over time, initial extensive cerebellar and occipital activation at 2 weeks transitioned to more focused and sustained activation in these regions at 3 and 6 months. This indicates the neuroplastic effects of the intervention, with both immediate and long-term changes in brain activity related to control and visual processing.
Several studies previously implicated the role of cerebellum and vermis parts in CUD/craving44,52,53. Our agnostic approach confirmed these results, revealing clusters in the cerebellum common to both craving scores (VAS and CCQ-N) across all time points, indicating the predictive power of neuromarkers from these less-noted brain regions.
Several functional imaging studies have suggested the role of cerebellum in stimulant-associated behaviors, and indicated the relationship between the cerebellar activity during cocaine craving, and recall of cocaine-use experience. In one study BOLD fMRI analysis in cocaine users showed that cocaine-related cues selectively induce BOLD activation in vermis lobules 2–3 and 8–952. In this context, our study can be regarded as evidence for the locations in cerebellum and vermis that have the strongest association with the changes in craving. Our agnostic MVPA showed the association between connectivity changes in cerebellum 4–6 left and vermis 4–9, and VAS changes at 2 weeks. Similarly right parts of cerebellum 6–10, left parts of cerebellum 7–9, and vermis 8 were identified after 6 months (Table 6). For CCQ-N changes, surprisingly all the identified regions from MVPA are commonly localized in cerebellum 8 and 9 left at all three time points. Vermis 4–8 were also identified at 2 weeks and 3 months.
E. Prediction
Here, we used a multivariate prediction model from both clinical and connectomics measures to predict the response to rTMS in CUD. Our results show that adding brain connectomics from SBC and MVPA analysis to clinical scores improves the predictions accuracy, especially at open-label phase (Tables 7, 8, 9, 10) (See also Table S1 and S2).
Prediction models indicate that baseline scores are strong predictors of post-intervention outcomes for both VAS and CCQ-N across different phases. For the sham group, baseline scores were significant, while SBC and MVPA predictors were not. For the active group, both baseline scores and SBC predictors were significant predictors (Tables 7 and 9).
During the open-label phase, for VAS prediction, the influence of SBC and MVPA fluctuates, with notable significance at 3 and 6 months. Specifically, the combination of rsfMRI neuromarkers and baseline VAS accounts for 66%, 97%, and 85% of the variance in clinical benefits after 2 weeks, 3 months, and 6 months, respectively (Table 8). The models showed limitations in predicting VAS changes for very small or zero values, but residuals in these cases were small (in the interval [0,2]).
For CCQ_N prediction, SBC and MVPA show significant effects at all time points. rsfMRI neuromarkers together with baseline CCQ-N account for 45%, 82%, and 71% of the variance in craving reduction after 2 weeks, 3 months, and 6 months, respectively (Table 10).
The combined models exhibit varying degrees of explanatory power, with the open-label phase models generally showing stronger fits (higher R2) and lower MAE values, suggesting improved prediction accuracy over time. This indicates that interventions might have more profound effects in the long term, with brain connectivity metrics becoming more predictive of changes in VAS and CCQ-N scores as time progresses. Overall, the results suggest that the combined model is a valuable tool for predicting long-term changes in VAS scores following interventions, with continued refinement needed to enhance short-term prediction accuracy. It should be noted that all the statistics are repeated with including age and years of education as additional predictors; however, they did not improve models’ performance, and were removed from the final models.
Several studies provided evidence on the usefulness of neuromarkers in predicting treatment responses. Different modeling approaches were employed to link neuroimaging and behavioral data. Several studies have incorporated neuromarkers for prediction of rTMS outcomes in addiction58, Schizophrenia59, Alzheimer’s disease60, and depression31,32,61,62. Some studies have used neuromarkers to predict treatments in brain disorders such as depression26,27, schizophrenia28, dyslexia29, generalized anxiety30. As an example, Whitfield-Gabrieli et al.25 used a combination of SBC, MVPA, and diffusion MRI neuromarkers to predict treatment response to social anxiety disorder. Similarly, recent studies have demonstrated that SBC analysis of resting-state fMRI data can provide mechanistic predictions for changes in sedentary behavior63.
Although most of the studies model the relationship between neuroimaging data and outcome measures, the findings mainly fail when applied to new datasets. The ability to being generalized to new cases is one of the main features in choosing neuromarkers for clinical practice. Due to our small sample size, we used leave-one-subject-out cross-validation to ensure about the generalizability of our predictive models. Specifically, in each connectivity analysis, we extracted the connectivity measures for each individual from the cluster mask obtained from group analysis performed for the rest of the patients. Such procedure has resulted in robust models in previous studies25,63. Doing so, we can avoid non-independence in our data analysis64 and also prevent the problem of double dipping in neuroscience65.
We hypothesized that the changes in neuromarkers together with rTMS intervention would be reflected in craving level. Therefore, in our set of analyses we considered the connectivity “changes” as predictors in the models. However, from clinical point of view, it is worth to assess whether the “baseline” brain condition of patients can predict future treatment response. In this regard, in our second set of analyses, we tried to assess the power of the “baseline” connectomics measures in outcome predictions. Our findings (See Table S3 and S4) show that baseline functional connectivity is also predictive for both VAS and CCQ-N, with comparable adjusted-R2 values to the model with “connectivity changes” as predictors. The results were slightly better in VAS prediction model for 2w and 3m (higher adj. R2 and lower MAE), but generally lower in CCQ-N prediction model. This may be explained by the differences between craving measurements, as the VAS is a more visual cue whereas the CCQ-N is a questionnaire.
F. Limitations
One of the main limitations of our study is that the data only includes the sham group for the short-term acute phase, and a control group is not available for the long-term assessments. Future study designs investigating the effects of rTMS should consider including control groups for both short-term and long-term phases.
To predict long-term treatment outcomes, the entire course of changes in clinical and connectivity measures during treatment should be considered. Our analysis was limited to comparisons between scores at baseline and each time point. Despite our small sample size, the employed method showed that a linear model combining clinical scores and connectomics can be predictive in rTMS treatment. However, a more comprehensive model incorporating various neuroimaging modalities together with all behavioral, clinical and demographic data from all time points, may improve predictions. Nonlinear models such as exponential decay model62 or machine learning-based approaches31,66 could also enhance outcome prediction accuracy.
While a cluster-forming threshold of p-value < 0.05 with correction for multiple comparisons can indeed control false positives effectively in both SBC and MVPA analysis, this approach often results in overly conservative thresholds in our study, which may lead to empty or incomplete clusters. In such cases, important signals may be missed entirely, impacting the subsequent prediction analysis. By choosing p-value < 0.01, we aimed to minimize Type I errors while retaining sensitivity to detect clusters with meaningful signal (non-zero voxels).
We assessed the predictive power of our models using adjusted R2 and MAE. The R2 value compares the unexplained variance (variance of the model’s error) with the total variance of the data. While R2 is a unitless measure, it is sensitive to outliers, and as a result, different models with different configurations can result to similar R2. This fact is evident in plots for VAS prediction at 3m in Fig. 7. It can be seen that despite different predictions for the same observed values, the R2 value of the combined model is close to 1. Due to this limitation, we used MAE as the other metric for evaluation of models. This metric shows the differences in the units of clinical metrics, and makes the comparison and interpretation of models easier. However, a comprehensive assessment of models would require evaluating performance with various available metrics for linear models.
We hypothesized that the heterogeneity in patients’ brains is the substrate for heterogeneity in their response to treatment, and defined our predictors based on brain biomarkers. Therefore, our method differs from other studies that try to predict the connectivity changes from changes in behavior. Future analysis could explore predicting connectivity changes from behavioral changes.
Although we tried to link the neuroimaging data to behavioral data for rTMS treatment, one should bear in mind that this treatment was an add-on treatment to the conventional pharmacological therapies that CUD patients received. Therefore, there can be pharmacological and other factors influencing treatment outcomes. A more complete model in future would include this information in the prediction model.
We showed that the individual differences in functional connectivity changes from SBC and MVPA analysis can predict future changes in craving in cocaine users receiving add-on rTMS treatment. This study provides evidence for the power of neuromarkers in craving predictions, and highlights the idea that neurmarkers may offer improvements in precision medicine approaches.
Data availability
All raw data are freely available in BIDS format: https://doi.org/10.18112/openneuro.ds003037.v2.1.0. Clinical and cognitive data are available in Zenodo: https://doi.org/10.5281/zenodo.10409461.
References
Gay, A. et al. Repetitive transcranial magnetic stimulation (rTMS) as a promising treatment for craving in stimulant drugs and behavioral addiction: A meta-analysis. J. Clin. Med. 11(3), 624 (2022).
Kilts, C. D. et al. Neural activity related to drug craving in cocaine addiction. Arch. Gen. Psychiatry 58, 334–341 (2001).
Fryer, R. G., Heaton, P. S., Levitt, S. D. & Murphy, K. M. Measuring crack cocaine and its impact. Econ. Inq. 51, 1651–1681 (2013).
Angeles-Valdez, D. et al. The Mexican magnetic resonance imaging dataset of patients with cocaine use disorder: Sudmex conn. Sci. Data 9, 1–10 (2022).
Camprodon, J. A., Martínez-Raga, J., Alonso-Alonso, M., Shih, M. C. & Pascual-Leone, A. One session of high frequency repetitive transcranial magnetic stimulation (rTMS) to the right prefrontal cortex transiently reduces cocaine craving. Drug Alcohol Depend. 86, 91–94 (2007).
Politi, E., Fauci, E., Santoro, A. & Smeraldi, E. Daily sessions of transcranial magnetic stimulation to the left prefrontal cortex gradually reduce cocaine craving. Am. J. Addict. 17, 345–346 (2008).
Hanlon, C. A., Dowdle, L. T. & Scott, H. J. Modulating neural circuits with transcranial magnetic stimulation: Implications for addiction treatment development. Pharmacol. Rev. 70, 661–683 (2018).
Hanlon, C. A., Dowdle, L. T., Moss, H., Canterberry, M. & George, M. S. Mobilization of medial and lateral frontal-striatal circuits in cocaine users and controls: An interleaved TMS/BOLD functional connectivity study. Neuropsychopharmacology 41, 3032–3041 (2016).
Terraneo, A. et al. Transcranial magnetic stimulation of dorsolateral prefrontal cortex reduces cocaine use: A pilot study. Eur. Neuropsychopharmacol. 26, 37–44 (2016).
Hanlon, C. A. et al. What goes up, can come down: Novel brain stimulation paradigms may attenuate craving and craving-related neural circuitry in substance dependent individuals. Brain Res. 1628, 199–209 (2015).
Bolloni, C. et al. Bilateral transcranial magnetic stimulation of the prefrontal cortex reduces cocaine intake: A pilot study. Front. Psychiatry 7, 133 (2016).
Garza-Villarreal, E. A. et al. Clinical and functional connectivity outcomes of 5-Hz repetitive transcranial magnetic stimulation as an add-on treatment in cocaine use disorder: A double-blind randomized controlled trial. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 6, 745–757 (2021).
Steele, V. R., Maxwell, A. M., Ross, T. J., Stein, E. A. & Salmeron, B. J. Accelerated intermittent theta-burst stimulation as a treatment for cocaine use disorder: A proof-of-concept study. Front. Neurosci. 13, 1147 (2019).
Sanna, A. et al. Intermittent theta burst stimulation of the prefrontal cortex in cocaine use disorder: A pilot study. Front. Neurosci. 13, 765 (2019).
Young, J. R., Galla, J. T. & Appelbaum, L. G. Transcranial magnetic stimulation treatment for smoking cessation: An introduction for primary care clinicians. Am. J. Med. 134, 1339–1343 (2021).
Rosenberg, H. Clinical and laboratory assessment of the subjective experience of drug craving. Clin. Psychol. Rev. 29, 519–534 (2009).
Tiffany, S. T. & Wray, J. M. The clinical significance of drug craving. Ann. N. Y. Acad. Sci. 1248, 1–17 (2012).
Paliwal, P., Hyman, S. M. & Sinha, R. Craving predicts time to cocaine relapse: Further validation of the now and Brief versions of the cocaine craving questionnaire. Drug Alcohol Depend. 93, 252–259 (2008).
Sinha, R., Garcia, M., Paliwal, P., Kreek, M. J. & Rounsaville, B. J. Stress-induced cocaine craving and hypothalamic-pituitary-adrenal responses are predictive of cocaine relapse outcomes. Arch. Gen. Psychiatry 63, 324–331 (2006).
Heinonen, J. et al. Default mode and executive networks areas: Association with the serial order in divergent thinking. PLoS ONE 11, 1–16 (2016).
Rasgado-toledo, J., Issa-garcia, V., Alcalá-lozano, R. & Garza-villarreal, E. A. Cortical and subcortical connections change after repetitive transcranial magnetic stimulation therapy in cocaine use disorder and predict clinical outcome. (2023).
Hanlon, C. A., Dufault, D. L., Wesley, M. J. & Porrino, L. J. Elevated gray and white matter densities in cocaine abstainers compared to current users. Psychopharmacology 218, 681–692 (2011).
Ernst, M., Ph, D., London, E. D. & Ph, D. Neural Systems and Cue-Induced Cocaine Craving. (1999).
Martinez, D. et al. A pilot study of transcranial magnetic stimulation of the medial prefrontal and cingulate cortices and cocaine self-administration. Brain Stimul.: Basic, Transl. Clin. Res. Neuromodulation 12, 555 (2019).
Whitfield-Gabrieli, S. et al. Brain connectomics predict response to treatment in social anxiety disorder. Mol. Psychiatry 21, 680–685 (2016).
Fu, C. H. Y., Steiner, H. & Costafreda, S. G. Predictive neural biomarkers of clinical response in depression: A meta-analysis of functional and structural neuroimaging studies of pharmacological and psychological therapies. Neurobiol. Dis. 52, 75–83 (2013).
Siegle, G. J., Carter, C. S. & Thase, M. E. Use of FMRI to predict recovery from unipolar depression with cognitive behavior therapy. Am. J. Psychiatry 163, 735–738 (2006).
Kumari, V. et al. Dorsolateral prefrontal cortex activity predicts responsiveness to cognitive-behavioral therapy in schizophrenia. Biol. Psychiatry 66, 594–602 (2009).
Hoeft, F. et al. Neural systems predicting long-term outcome in dyslexia. Proc. Natl. Acad Sci. U S A 108, 361–366 (2011).
Whalen, P. J. et al. A functional magnetic resonance imaging predictor of treatment response to venlafaxine in generalized anxiety disorder. Biol. Psychiatry 63, 858–863 (2008).
Cash, R. F. H. et al. A multivariate neuroimaging biomarker of individual outcome to transcranial magnetic stimulation in depression. Hum. Brain Mapp. 40, 4618–4629 (2019).
Harika-Germaneau, G. et al. Baseline clinical and neuroimaging biomarkers of treatment response to high-frequency rTMS over the left DLPFC for resistant depression. Front. Psychiatry 13, 1–12 (2022).
Koutsouleris, N. et al. Predicting response to repetitive transcranial magnetic stimulation in patients with schizophrenia using structural magnetic resonance imaging: A multisite machine learning analysis. Schizophr. Bull. 44, 1021–1034 (2018).
Hobkirka, A. L., Bella, R. P., Utevskyb, A. V., Huettelb, S. & Meade, C. S. Reward and executive control network resting-state functional connectivity is associated with impulsivity during reward-based decision making for cocaine users. Physiol. Behav. 176, 139–148 (2016).
Hester, R. & Garavan, H. Executive dysfunction in cocaine addiction: Evidence for discordant frontal, cingulate, and cerebellar activity. J. Neurosci. 24, 11017–11022 (2004).
Whitfield-Gabrieli, S. & Nieto-Castanon, A. Conn: A functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. 2, 125–141 (2012).
Nieto-Castanon, A. & Whitfield-Gabrieli, S. CONN functional connectivity toolbox: RRID SCR_009550, release 20. Preprint at https://doi.org/10.56441/hilbertpress.2048.3738. (2020).
Chen, C. H. et al. Brain imaging correlates of depressive symptom severity and predictors of symptom improvement after antidepressant treatment. Biol. Psychiatry 62, 407–414 (2007).
Nieto-Castanon, A. Handbook of Functional Connectivity Magnetic Resonance Imaging Methods in CONN (Hilbert Press, 2020).
Morris, T. P. et al. Resting state functional connectivity provides mechanistic predictions of future changes in sedentary behavior. Sci Rep 12, 1–11 (2022).
Norman, K. A., Polyn, S. M., Detre, G. J. & Haxby, J. V. Beyond mind-reading: multi-voxel pattern analysis of fMRI data. Trends Cogn. Sci. 10, 424–430 (2006).
Nieto-Castanon, A. Brain-wide connectome inferences using functional connectivity MultiVariate Pattern Analyses (fc-MVPA). PLoS Comput. Biol. 18, 1–28 (2022).
Haslwanter, T. An Introduction to Statistics with Python (Springer Cham, 2016). https://doi.org/10.1007/978-3-319-28316-6.
Bonson, K. R. et al. Neural systems and cue-induced cocaine craving. Neuropsychopharmacology 26, 376–386 (2002).
Bellamoli, E. et al. rTMS in the treatment of drug addiction: An update about human studies. Behav. Neurol. 2014, 815215 (2014).
Breiter, H. C. et al. Acute effects of cocaine on human brain activity and emotion. Neuron 19, 591–611 (1997).
Prisciandaro, J. J., McRae-Clark, A. L., Myrick, H., Henderson, S. & Brady, K. T. Brain activation to cocaine cues and motivation/treatment status. Addict. Biol. 19, 240–249 (2014).
Camchong, J. et al. Frontal hyperconnectivity related to discounting and reversal learning in cocaine subjects. Biol. Psychiatry 69, 1117–1123 (2011).
Pando-Naude, V. et al. Gray and white matter morphology in substance use disorders: A neuroimaging systematic review and meta-analysis. Transl. Psychiatry 11, 29 (2021).
Zhao, K. et al. A generalizable functional connectivity signature characterizes brain dysfunction and links to rTMS treatment response in cocaine use disorder. Preprint at https://doi.org/10.1101/2023.04.21.23288948 (2023).
Müller, C. P., De Silva, M. A. S. & Huston, J. P. Double dissociating effects of sensory stimulation and cocaine on serotonin activity in the occipital and temporal cortices. Neuropharmacology 52, 854–862 (2007).
Anderson, C. M. et al. Cerebellar vermis involvement in cocaine-related behaviors. Neuropsychopharmacology 31, 1318–1326 (2006).
Miquel, M., Gil-Miravet, I. & Guarque-Chabrera, J. The Cerebellum on Cocaine. Front. Syst. Neurosci. 14 (2020).
Goldstein, R. Z. & Volkow, N. D. Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat. Rev. Neurosci. 12, 652–669 (2011).
Goldstein, R. Z. et al. Role of the anterior cingulate and medial orbitofrontal cortex in processing drug cues in cocaine addiction. Neuroscience 144, 1153–1159 (2007).
Knoch, D., Pascual-Leone, A., Meyer, K., Treyer, V. & Fehr, E. Diminishing reciprocal fairness by disrupting the right prefrontal cortex. Science 1979(314), 829–832 (2006).
Hanlon, C. A. et al. Probing the Frontostriatal Loops Involved in Executive and Limbic Processing via Interleaved TMS and Functional MRI at Two Prefrontal Locations: A Pilot Study. PLoS ONE 8, e67917 (2013).
Noël, X. et al. Contribution of frontal cerebral blood flow measured by (99m) Tc-Bicisate spect and executive function deficits to predicting treatment outcome in alcohol-dependent patients. Alcohol Alcohol. 37, 347–354 (2002).
Koutsouleris, N. et al. Predicting response to repetitive transcranial magnetic stimulation in patients with schizophrenia using structural magnetic resonance imaging: a multisite machine learning analysis. Schizophr. Bull. 44, 1021–1034 (2017).
Zhang, S. et al. Evaluating the treatment outcomes of repetitive transcranial magnetic stimulation in patients with moderate-to-severe Alzheimer’s disease. Front. Aging. Neurosci. 14, 1070535 (2022).
Ge, R. et al. Predictive value of acute neuroplastic response to rTMS in treatment outcome in depression: A concurrent TMS-fMRI trial. Am. J. Psychiatry 179, 500–508 (2022).
Berlow, Y. A. et al. Modeling the antidepressant treatment response to transcranial magnetic stimulation using an exponential decay function. Sci. Rep. 13, 1–9 (2023).
Morris, T. P. et al. Resting state functional connectivity provides mechanistic predictions of future changes in sedentary behavior. Sci. Rep. 12, 1–11 (2022).
Esterman, M., Tamber-Rosenau, B. J., Chiu, Y. C. & Yantis, S. Avoiding non-independence in fMRI data analysis: Leave one subject out. Neuroimage 50, 572–576 (2010).
Kriegeskorte, N., Simmons, W. K., Bellgowan, P. S. F. & Baker, C. I. Circular analysis in systems neuroscience: The dangers of double dipping. Nat. Neurosci. 12, 535–540 (2009).
Chekroud, A. M. et al. The promise of machine learning in predicting treatment outcomes in psychiatry. World Psychiatry 20, 154–170 (2021).
Acknowledgements
N. G. and H. S. Z gratefully appreciate the Iran National Science Foundation (INSF), Tehran, Iran, for financial support during this study.
Author information
Authors and Affiliations
Contributions
N.G. Conceptualization, design, analysis, interpretation of data, manuscript writing, E.A.G. Conceptualization, interpretation of data, substantial revision, H.S.Z. Conceptualization, interpretation of data, substantial revision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ghazi, N., Garza-Villarreal, E.A. & Soltanian-Zadeh, H. Brain connectomics markers for response prediction to transcranial magnetic stimulation in cocaine use disorder. Sci Rep 15, 15336 (2025). https://doi.org/10.1038/s41598-025-99113-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-99113-6
Keywords
- Resting state functional magnetic resonance imaging (rsfMRI)
- Brain connectomics
- Response prediction model
- Repetitive transcranial magnetic stimulation (rTMS)
- Craving
- Dorsolateral prefrontal cortex (DLPFC)
- Anterior cingulate cortex (ACC)
- Seed-based connectivity (SBC)
- Multi voxel pattern analysis (MVPA)
- Neuromarkers