Abstract
Stress urinary incontinence (SUI) is usually associated with aging and parity, but repetitive intra-abdominal pressure during physical activity can also trigger its onset in young women. Horseback riding, despite potential benefits for pelvic floor strengthening, may also contribute to pelvic floor strain and urinary symptoms. The study included 100 Polish women aged 21–54 engaged in horse riding, both recreational (85%) and professional (15%). Participants were divided into three age groups and screened for urinary incontinence symptoms. Quality of life was assessed with validated questionnaires (IIQ-7, UDI-6SF) and a custom 25-item survey covering equestrian practice, reproductive history, urinary symptoms, and general health. Statistical analyses were performed in R using non-parametric tests, correlation, and logistic regression. Stress urinary incontinence was reported by 36% of participants, significantly impairing quality of life. Women with higher BMI, older age, and childbirth history demonstrated increased symptom prevalence and higher IIQ-7 and UDI-6 scores (p < 0.05). Training type, frequency, and duration were not significantly associated with outcomes, except for shorter riding experience, which correlated with higher UDI-6 scores. Logistic regression identified age (OR = 1.07) and BMI (OR = 1.16) as risk factors, while greater height reduced SUI odds (OR = 0.92). Urinary incontinence is a relevant issue among female horse riders, negatively affecting physical, social, and emotional well-being. Age, BMI and childbirth history play crucial roles in the incidence of SUI symptoms. The longer the training experience, the lower is reported urinary incontinence distress. Preventive measures such as pelvic floor training, individualized physiotherapy, and interdisciplinary care may reduce symptom burden and support longer professional activity. Raising awareness and encouraging early intervention are essential to improving athletes’ quality of life.
Similar content being viewed by others
Introduction
Regular training is the basis of good physical condition and mental health. However, every sport, trained in a methodical way, brings about some consequences: sport-related injuries and pain, pelvic floor muscles (PFM) strain and its long-term sequelae.
Even though stress urinary incontinence (SUI) is more associated with age and parity, repetitive intra-abdominal pressure, such as strenuous physical activity might be a trigger factor for its onset1. SUI exacerbates with the number and the intensity of training, occurring in 8% of recreational and 21% competitive athletes, while in sedentary women it was reported in 11%2. In many cases, incontinence is transient and limited to sport activity. Occasional SUI while performing selected sport was reported by 28% of university nulliparous athletes3. Nevertheless, it is still under-admitted by patients who are not seeking help4.
Continence is provided by the proper role of the urethral support system and urethral sphincteric closure system. Urethral support system is based on stability of pelvic floor muscles and structures surrounding urethra: anterior vaginal wall, endopelvic fascia and tendinous arch of pelvic fascia5.
Studies on the impact of physical activity on pelvic floor function do not provide a clear answer as to whether it is predominantly beneficial or may also have concomitant detrimental effects on women’s health.
On one hand, regular contractions are strengthening muscles, which provide better stabilization of the structure6.
On the other hand, it was also found excessive intra-abdominal pressure during exercises contributes to muscle damage, especially periurethral striated muscles in overstretching mechanisms5,6,7. Another mechanism of urinary leakage is also connected to muscle fatigue after prolonged recruitment of type II fibers, which are responsible for rapid contraction and which maintain urethra close during increased intra-abdominal pressure7.
The objective of the work was to assess the prevalence and risks factors of urinary leakage associated with horseback riding, both in amateur and professional riders. Also, we aim to assess the correlation of symptoms and quality of life. The major question of the study is if the equestrianism alone as a sport is a predisposing factor for SUI.
Materials and methods
This research was designed as an observational cross-sectional study assessing urinary symptoms and related factors among female horseback riders. The transversal study included 100 women aged 21–54 years from across Poland, engaged in horse riding either recreationally (85%) or professionally (15%). Inclusion criteria were: female sex, age ≥ 18 years, horse riding activity, and voluntary consent to participate. Exclusion criteria were: male sex, age < 18 years, previous prolapse or urinary incontinence operations, current genitourinary infection and lack of consent. Data were collected between March and November 2023 using a self-administered electronic questionnaire in Polish. The survey link was distributed via equestrian clubs, riding schools, and social media groups dedicated to horseback riding in Poland. Participation was voluntary and anonymous, and all questionnaires were completed electronically without researcher assistance.
The research was approved by the Bioethics Committee of the Medical University in Wroclaw with a number of KB − 806/2018. Informed consent was obtained from all participants. The study was performed in accordance with the Declaration of Helsinki. The study sample size was determined based on preliminary power estimations performed prior to data collection. Power calculations indicated that approximately 85 participants would be sufficient to detect medium effect sizes (Cohen’s d ≈ 0.5) in non-parametric group comparisons with 80% power at α = 0.05. To allow for subgroup analyses and potential missing data, we aimed to recruit at least 100 women, and this target was achieved.
Participants were stratified into three age groups based on the potential risk of urinary incontinence (UI): 18–30 years (n = 46), 31–45 years (n = 30), and 46–55 years (n = 24). In total, 36 women reported UI symptoms, while 64 did not.
The impact of UI on quality of life was assessed using two validated questionnaires: the Incontinence Impact Questionnaire (IIQ-7) and the Urinary Distress Inventory (UDI-6SF). IIQ-7 consists of seven items addressing the impact of UI on daily activities, mental health, and social functioning, scored on a 0–3 Likert scale, with a maximum score of 288,9. UDI-6SF contains six items evaluating urinary symptoms and their distress level, also scored on a 0–3 scale, with a maximum score of 2410. UDI-6SF helps in differentiation of the type of urinary incontinence: urge, stress, mixed and neurogenic incontinence. Also, a question about pain or discomfort in lower abdominal, pelvic, or genital area might indicate another condition overlapping with incontinence. Higher scores in both tools indicate greater symptom burden and quality-of-life impairment. Participants without UI did not complete these questionnaires.
In addition, a custom-designed 25-item questionnaire was administered. It covered four domains: (I) horse riding characteristics (discipline, training frequency, duration, and years of practice), (II) childbirth history and postpartum care (including consultations with gynecologists or urogynecological physiotherapists), (III) UI-related aspects (occurrence, triggers, and psychosocial consequences), and (IV) general health indicators (other physical activity, smoking, medication use, urogenital infections and procedures - such as laser or radiofrequency and comorbidities such as diabetes or hypertension). The questionnaire was developed specifically for this study to capture horse-riding characteristics, reproductive history, urinary symptoms, and general health. The initial pool of questions was generated based on literature regarding urinary incontinence in physically active women and clinical experience of a urogynecological physiotherapist.
Patients who reported not having the incidence of urinary incontinence of any kind were asked to fulfill only a 25-item questionnaire, as the UDI-6SF and IIQ-7 are designed to assess the kind and severity of incontinence symptoms.
The statistical analysis was performed entirely in R11.
The answers given by the surveyed women were transformed according to the Likert scale - the more severe the respondent’s symptoms were, the higher the point value of individual answers. Then, separately for each questionnaire (IIQ7, UDI6), the sum of points for all questions was determined. The obtained sums were unitarized to zero, according to the following formula:
where \({x_i}\) is the sum of the points of the i-th respondent in the questionnaire, x is a vector of sums of points of all respondents in the questionnaire and \({z_i}\) is the point value of the i-th respondent.
The compliance of the distribution of variables with the normal distribution was verified with the Shapiro-Wilk test at the significance level α = 0.05.
The statistical significance of an impact of the considered factors on the point values of the IIQ7 (IIQ7 score) and UDI6 (UDI6 score) questionnaires, due to the lack of compliance of the variable distribution with the normal distribution, was verified with the Wilcoxon test for independent samples (in the case of two groups) or the Kruskal-Wallis test (in the case of 3 or more groups) at the significance level α = 0.05. Post hoc analysis after the Kruskal–Wallis test, whose p-value < 0.05, was performed using Dunn’s test at a significance level α = 0.05. The analysis described above was performed using the rstatix library12.
The correlation analysis of age, height, body weight, BMI, IIQ7 score and UDI6 score was performed using the psych library13. The statistical significance of correlation coefficients was verified at the significance level α = 0.05. The statistical significance of differences in the corresponding correlation coefficients (in healthy women and in women with diagnosed NTM) was verified on the basis of confidence intervals. Correlation coefficients whose confidence intervals had no common part were considered statistically significantly different14.
The statistical significance of the impact of age, height, body weight and BMI on the occurrence of NTM was verified on the basis of logistic regression analysis at the significance level α = 0.05.
The statistical significance of the influence of factors (e.g. number of deliveries) on the occurrence of NTM was verified by the χ2 test or Fisher’s exact test at the significance level α = 0.05 with a post hoc analysis in the case of 3 or more levels of the factor.
Results
Internal consistency was high for both instruments, with Cronbach’s alpha of 0.801 for IIQ-7 and 0.840 for UDI-6 (Table 1). Both coefficients were statistically significant and did not differ significantly between questionnaires.
IIQ-7 outcomes
Lifestyle and health factors were generally not associated with IIQ-7 scores, except for women reporting recurrent urogenital infections/urogynecological procedures and those with hypertension or diabetes, who presented significantly higher scores (p = 0.0318 and p = 0.0403, respectively; Table 2).
Women with stress urinary incontinence (SUI) reported markedly higher IIQ-7 scores compared with those without symptoms (p = 1.13 × 10−7, large effect size; Table 3). Longer symptom duration and difficulties in everyday life or travel were strongly associated with higher scores. UI-related behaviors, such as urination “in reserve,” problems with sexual activity, and feelings of embarrassment, were also linked to significantly higher IIQ-7 values.
Reproductive history was not significantly associated with IIQ-7 scores, including birth status, number or type of deliveries, and participation in uro-physiotherapy (Table 4). Similarly, equestrian training characteristics (type, frequency, duration, and intensity) showed no significant relationships with IIQ-7 results (Table 5).
UDI-6 outcomes
Lifestyle and health analyses indicated higher UDI-6 scores among women with a history of urogenital infections/procedures (p = 0.0437) and urinary bladder inflammation (p = 0.0098), while other factors were not significant (Table 6).
The presence of SUI was strongly associated with elevated UDI-6 scores (p = 1.28 × 10−11, large effect size; Table 7). Higher scores were also observed in women with longer symptom duration, difficulties in daily life or traveling, urination “in reserve,” and feelings of embarrassment. Problems with sexual activity were linked to particularly high UDI-6 values.
Reproductive history was related to UDI-6 outcomes. Women who had given birth demonstrated significantly higher scores than nulliparas (p = 0.0039; Table 8). Increasing number of births was associated with a progressive increase in UDI-6 scores, although type of delivery and uro-physiotherapy were not significant factors.
Equestrian training characteristics were generally not associated with UDI-6, with the exception of training duration. There was no significant difference in severity of symptoms between amateur, recreational and professional groups. Also, no correlation with the training duration or training frequency was found. Women training less than one year demonstrated higher scores compared to those with longer experience (p = 0.0105; Table 9).
Correlation analyses
Correlation analysis showed significant negative associations of height with both IIQ-7 and UDI-6 scores, while BMI was positively correlated with UDI-6 (Table 10). IIQ-7 and UDI-6 scores were strongly intercorrelated (r = 0.70, p < 0.05).
Subgroup analyses indicated that in women diagnosed with SUI, BMI was strongly correlated with body weight (r = 0.94) and with both IIQ-7 and UDI-6 scores. In healthy women, significant associations were observed between body height and weight (r = 0.50) (Table 11).
Logistic regression analysis
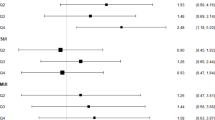
Logistic regression identified age (OR = 1.07, p = 0.0011) and BMI (OR = 1.16, p = 0.0284) as significant predictors of SUI (Table 12). Greater height was associated with reduced odds of SUI (OR = 0.92, p = 0.0316).
Reproductive history and SUI
The occurrence of SUI was significantly higher among women who had given birth compared to nulliparas (p = 0.0007). An increasing number of births was associated with a higher prevalence of SUI (p = 0.0013; Table 13).
Discussion
Our research indicates that stress urinary incontinence (SUI) is present among female horseback riders and is associated with established risk factors such as age, BMI, and parity. These results are consistent with previous studies indicating that increasing age and body weight contribute to weakening of pelvic floor structures, thereby increasing SUI risk15. In this context, cooperation with physiotherapists during equestrian training may be valuable, which is supported by Carboni’s observations showing improved pelvic floor function following guided training16. These associations highlight that demographic factors remain more influential than sport-specific factors in determining SUI risk among horseback riders in our study.
Several studies have proposed that horseback riding may strengthen pelvic musculature. Some research suggests that regular equestrian practice may even be associated with a lower risk of SUI and is not linked to sexual dysfunctions or lower urinary tract symptoms17. Although Battaglia et al. reported clitoral microcalcifications in riders, potentially indicating microtrauma, examined women remained asymptomatic18, which aligns with our observation that not all mechanical loads translate directly into symptoms in riders reporting SUI.
Schäfer indicated that as the muscle tension increases with the horse’s ride velocity, that sport may have different impacts depending on the kind of activity19. Elmeua González showed that different parts of muscles were involved in novice and advanced riders. Discrepancies in muscle involvement are due to different forms of horseback riding - it is performed in a different way during diverse horse gaits: walk, trot, canter. Differences were also shown between recreational riders and professionals20. These findings are consistent with our results showing that professional riders reported SUI less frequently, possibly due to refined technique and improved neuromuscular control acquired over years of training. Notably, despite these biomechanical differences, our study did not identify significant associations between specific training characteristics and SUI occurrence.
Our study also confirms the significance of widely recognized SUI risk factors BMI, parity and age, known to contribute to pelvic floor weakening. Interestingly, while earlier studies associated taller height with increased risk of incontinence21, we observed the opposite trend. As no anatomical or functional explanation has been identified, this finding requires further investigation.
The incidence of urogenital infections in our sample was relatively high, but aligns with a Polish study showing that infections such as candidiasis affect up to 24% of women aged 15–4422. Although no specific data exist regarding urinary tract infections in equestrians, prolonged riding time, delayed voiding, and exposure to cold conditions may contribute to irritation or infection, which could partially explain some urinary symptoms reported in our cohort.
Previous studies have shown that strenuous sports can temporarily increase SUI symptoms, although long-term effects may not persist beyond the period of intense activity1. This is relevant to our group, whose age distribution may not yet fully reflect the later-life consequences of repeated high-impact exposure. A possible mechanism of urinary incontinence across sports is the imbalance between forces exerted on perineal structures and the stabilizing capacity of pelvic tissues4.
A confounding factor, found also among our patients, might be a presence of comorbidities, such as hypertension and diabetes, leading to more frequent urination, nycturia or nocturia.
Limitation of the study is lack of objective gynecological test performed in the studied group: Bonney test and ultrasound confirmation, as anatomical changes in the urethral support system can be confirmed with urethral funneling. Major limitations of our study is a non-homogenous group of patients, especially concerning age, parity and professional level of equestrianism. Future studies incorporating objective pelvic floor assessment and gait-specific biomechanical analysis are needed to clarify the mechanisms underlying SUI in equestrians.
Preventive measures such as pelvic floor training, individualized physiotherapy, and interdisciplinary care may reduce symptom burden and support longer professional activity. Raising awareness and encouraging early intervention are essential to improving athletes’ quality of life4,16,23.
Speaking with athletes about the mechanism of SUI, using preventive measures and advising them to simultaneously exercise Kegel’s muscle may broaden the knowledge and help limit the distress associated with involuntary leakage.
Conclusions
The study confirmed that stress urinary incontinence and related symptoms may significantly impair quality of life among female horse riders, particularly in older women, those with higher BMI, and those with a history of childbirth. Our results suggest that longer riding experience is associated with lower incontinence-related distress, although the exact influence of horseback riding on SUI remains difficult to determine. There is an impact of the time of training and incidence of urinary leakage symptoms, suggesting that longer training predisposes to develop either preventing behaviors or is associated with better pelvic floor functioning. However, it is hard to verify the exact influence of horseback activity on urinary incontinence. Preventing or treating urinary incontinence may not only enhance athletes quality of life, but also prolong their professional activity lifespan.
Interdisciplinary team of professionals including sport doctors, orthopedics, physiotherapists, and, if recommended, gynecologist, should provide care and counselling on sport professionals.
Data availability
The datasets used and/or analyzed during the current study areavailable from the corresponding author on reasonable request.
References
Nygaard, I. E. Does prolonged high-impact activity contribute to later urinary incontinence? A retrospective cohort study of female olympians. Obstet. Gynecol. 90, 718–722 (1997).
Manaseer, T. S. & Al-Nassan, S. M. The point prevalence of stress incontinence among young adult Jordanian women based on activity levels. Physiother. Res. Int. 29, e70000 (2024).
Nygaard, I. E., Thompson, F. L., Svengalis, S. L. & Albright, J. P. Urinary incontinence in elite nulliparous athletes. Obstet. Gynecol. 84, 183–187 (1994).
Rzymski, P., Burzyński, B., Knapik, M., Kociszewski, J. & Wilczak, M. How to balance the treatment of stress urinary incontinence among female athletes? Arch. Med. Sci. 17, 314–322 (2021).
Bø, K. Urinary incontinence, pelvic floor dysfunction, exercise and sport. Sports Med. 34, 451–464 (2004).
Cygańska, A. K., Giżewska, B. & Zdunek, K. Evaluating pelvic floor dysfunction in female horse show jumpers using the Australian pelvic floor questionnaire. Med. Sci. Monit. 31, e946830 (2025).
Pires, T., Pires, P., Moreira, H. & Viana, R. Prevalence of urinary incontinence in high-impact sport athletes: A systematic review and meta-analysis. J. Hum. Kinet. 73, 279–288 (2020).
Chang, Y. W. et al. Evaluating the efficacy of the single-incision uphold system for pelvic organ prolapse repair. Taiwan. J. Obstet. Gynecol. 58, 94–98 (2019).
Lin, L. et al. Comparison between tension-free vaginal tape and transobturator tape in treating stress urinary incontinence after vaginal mesh surgery. Taiwan. J. Obstet. Gynecol. 57, 528–531 (2018).
Kieres, P., Rokita, W., Stanisławska, M., Rechberger, T. & Gałęzia, M. Diagnostic value of selected questionnaires (UDI-6 SF, Gaudenz, MESA, ICIQ-SF and King’s Health Questionnaire) in the diagnosis of various clinical forms of urinary incontinence in women. Ginekol. Pol. 79, 338–341 (2008).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2023).
Kassambara, A. Rstatix: Pipe-friendly framework for basic statistical tests. R Package Version 0.7.2. https://CRAN.R-project.org/package=rstatix (2023).
Revelle, W. psych: Procedures for personality and psychological research.. Northwestern University, Evanston, IL, R package version 1.8.12. https://CRAN.R-project.org/package=psych (2023).
Bender, R. & Lange, S. Adjusting for multiple testing—when and how? J. Clin. Epidemiol. 54, 343–349 (2001).
Batmani, S. et al. Prevalence and factors related to urinary incontinence in older adult women worldwide: A comprehensive systematic review and meta-analysis of observational studies. BMC Geriatr. 21, 212. https://doi.org/10.1186/s12877-021-02135-8 (2021).
Carboni, C., Blanquet, M. & Bouallalene Jaramillo, K. Effectiveness of horseback riding in the management of pelvic floor dysfunctions. Int. J. Health Rehabil. Sci. 3, 1–6 (2014).
Alanee, S., Heiner, J., Liu, N. & Monga, M. Horseback riding: Impact on sexual dysfunction and lower urinary tract symptoms in men and women. Urology 73, 109–114 (2009).
Battaglia, C. et al. Ultrasonographic and doppler findings of subclinical clitoral microtraumatisms in mountain bikers and horseback riders. J. Sex. Med. 6, 464–468 (2009).
Schäfer, D. & Pannek, J. Measurement of pelvic floor function during physical activity: A feasibility study. Scand. J. Urol. Nephrol. 43, 315–318 (2009).
Elmeua González, M. & Šarabon, N. Muscle modes of the equestrian rider at walk, rising trot and canter. PLoS ONE 15, e0237727 (2020).
Vahdatpour, B., Zargham, M., Chatraei, M., Bahrami, F. & Alizadeh, F. Potential risk factors associated with stress urinary incontinence among Iranian women. Adv. Biomed. Res. 4, 205 (2015).
Wójkowska-Mach, J. et al. Prevalence and antimicrobial susceptibility profiles of microorganisms associated with lower reproductive tract infections in women from Southern Poland: A retrospective laboratory-based study. Int. J. Environ. Res. Public Health 18, 335 (2021).
Sorrigueta-Hernández, A. et al. Benefits of physiotherapy on urinary incontinence in high-performance female athletes: A meta-analysis. J. Clin. Med. 9, 3240 (2020).
Author information
Authors and Affiliations
Contributions
Conceptualization, Gabriela Kołodyńska, Wiktoria Misztela; Data curation, Maciej Zalewski and Anna Mucha; Formal analysis, Maciej Zalewski, Anna Mucha, Aleksandra Piątek and Waldemar Andrzejewski; Funding acquisition, Maciej Zalewski; Investigation, Anna Mucha; Methodology, Waldemar Andrzejewski; Project administration and Supervision, Gabriela Kołodyńska; Writing – original draft, Gabriela Kołodyńska; Writing – review & editing, Waldemar Andrzejewski, Aleksandra Piątek.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
Informed consent was obtained from all subjects involved in the study.
Institutional review board statement
The study was approved by the Bioethics Committee of the Medical University of Wrocław number KB − 806/2018.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zalewski, M., Kołodyńska, G., Piątek, A. et al. Prevalence and risk factors of stress urinary incontinence among female horseback riders in Poland. Sci Rep 16, 5606 (2026). https://doi.org/10.1038/s41598-026-36444-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-36444-y