Abstract
Ti-6Al-4 V titanium alloy is widely utilized in orthodontic applications due to its favorable biocompatibility and mechanical properties. However, its long-term performance can be adversely affected by the dynamic and hostile oral environment, particularly under pathological conditions such as gastroesophageal reflux disease (GERD). Herein, In vitro corrosion behavior of Ti-6Al-4 V over a 240-hour immersion period at 37 °C in artificial saliva simulating GERD, with pepsin and Candida albicans, both individually and combined. Electrochemical impedance spectroscopy (EIS) and potentiodynamic polarization revealed that pepsin significantly improved corrosion resistance, achieving a maximum inhibition efficiency (IE) of 87.4%, while C. albicans showed a time-dependent decline in protection, with lower IE 71.8%. The combined presence of both agents further reduced IE to 55.6%, indicating a complex synergistic effect accelerating corrosion. Surface characterization by scanning electron microscopy (SEM) confirmed biofilm formation and surface degradation. Response Surface Methodology (RSM) modeling identified immersion time and component interactions as key factors influencing corrosion behavior. These findings offer novel insights into the interplay among enzymatic activity and microbial colonization, highlighting clinical implications for implant stability in GERD-affected oral environments.
Similar content being viewed by others
Introduction
In the past few decades, titanium alloys have been broadly used in dental prostheses for instance metal plates, orthodontic wires and orthodontic materials, metal-ceramic restorations, crowns, and bridges1. Human body susceptibility to hypoallergenic metal implants is a common adverse event, and the ability of FDA-approved titanium devices to tolerate the metal ions created or released during long-term implantation raises ethical considerations2. Titanium (Ti) alloys are indispensable in key applications, such as medical devices, owing to their outstanding specific strength and biocompatibility. The notable corrosion resistance of titanium is attributed not only to its inherent material properties and the formation of a stable, self-healing passive oxide layer, but also to its interactions with the surrounding environment3.
To ensure biocompatibility, it is essential to understand the corrosion mechanisms of titanium and its alloys within the oral cavity and to develop strategies that minimize the release of potentially harmful metal ions into the human body4.
The oral cavity represents a sensitive environment where a delicate equilibrium is maintained between the teeth and surrounding tissues. Saliva composition and pH vary among individuals, as saliva is a complex fluid containing inorganic salts, acids, enzymes, bacteria, and gastric secretions5. The oral microbiota comprises diverse microorganisms, including bacteria, fungi, protozoa, mycoplasma, and viruses6, which exist in a mutually regulated and interdependent relationship to sustain the homeostasis of the oral microbiome7.
Candida species are opportunistic fungi commonly present in the environment as well as on human skin and mucosal surfaces. While typically harmless, they can proliferate rapidly and form mycelial structures under specific conditions, leading to pathogenic infections8– 9.
Candida albicans is responsible for approximately 25–75% of oral candidiasis cases and affects up to 90% of the elderly population. Its pathogenicity is largely attributed to its remarkable ability to adhere to surfaces. This adhesion is further facilitated by the hydrophobic properties and electrostatic interactions of its cell surface10.
Gastroesophageal reflux disease (GERD) is a common chronic condition characterized by the reflux of gastric contents, including hydrochloric acid and bile, into the esophagus. This backflow leads to mucosal injury and symptoms such as heartburn, regurgitation, and dysphagia11– 12. Recently, salivary pepsin has gained attention as a potential biomarker for the diagnosis of GERD and other acid-related disorders13.
Pepsin is one of the primary proteolytic enzymes responsible for protein digestion within the digestive system14– 15. Gastric juice, characterized by a highly acidic pH ranging from 1 to 3, contains hydrochloric acid (HCl), chloride ions (Cl⁻), and pepsin16. In contrast, intestinal juice has a near-neutral pH between approximately 5.0 and 7.7, achieved through neutralization of gastric acid by pancreatic secretions and buffering agents to protect the intestinal mucosa from acidic damage17.
Saliva, the primary aqueous component of the oral cavity, becomes more acidic in the presence of GERD, with pH values dropping to around 4.9 compared to the near-neutral pH of 6.5 observed in healthy individuals18. The inclusion of Candida albicans in this study is justified by its prominent role as a pathogen in oropharyngeal candidiasis, particularly among patients with orthodontic devices. Investigating the influence of pepsin on corrosion and biofilm development on titanium alloys offers critical insights into the complex interplay between microbial infections, enzymatic activity, and the oral environment, and their combined impact on the durability of orthodontic materials.
This study (Fig. 1) evaluates the electrochemical behavior of the Ti–6Al–4 V alloy in artificial saliva, with a particular focus on the impact of Candida albicans as a biological contaminant. The influence of pepsin—a digestive enzyme frequently present in the oral environment under gastroesophageal reflux disease (GERD) conditions—was also assessed at 37 °C across different immersion periods. Corrosion inhibition efficiency was quantified as a function of immersion time. Additionally, Response Surface Methodology (RSM) was utilized to model and optimize the corrosion response, given its demonstrated effectiveness in simplifying and predicting complex corrosion phenomena19.
Experimental procedure
Solution preparation and electrode composition
Pepsin was sourced from Loba Chemie and prepared in a solution adjusted to pH 2.5. To clarify the final pH of the GERD-simulated solution, pepsin was first dissolved in distilled water and adjusted to pH 2.5 to ensure enzymatic activation. This pepsin stock solution was then mixed with artificial saliva (initial pH 6.5) in the predetermined volume ratio. Upon mixing, the solution equilibrated to a physiologically relevant GERD-simulated pH of approximately 4.9 as a result of the interaction between the acidity of pepsin and the buffering capacity of artificial saliva. The final pH (4.9 ± 0.1) was verified using a calibrated pH meter prior to initiating all corrosion measurements. The pH was carefully controlled using 0.1 M NaOH or 0.1 M HCl, measured with a HANNA 213 pH meter. The Ti-6Al-4 V alloy utilized in this study, supplied by Johnson and Matthey (England), has its elemental composition (wt%) presented in Table 1. Cylindrical specimens of the alloy, each with an exposed surface area of 0.196 cm², were prepared according to protocols outlined in our previous research.
Artificial saliva (AS) was formulated using high-purity (analytical grade) chemicals with the following concentrations (g/L)21: 0.72 KCl, 0.22 CaCl₂·2 H₂O, 0.60 NaCl, 0.68 KH₂PO₄, 0.866 Na₂HPO₄·12 H₂O, 1.50 KHCO₃, 0.06 KSCN, and 0.03 citric acid. The pH of the solution was adjusted to 6.5 using triply distilled water.
Fungal culture media
Candida albicans, obtained from the National Collection of Industrial Microorganisms, was maintained on an agar-based culture medium containing (g/L): glucose (10), peptone (10), yeast extract (5), and agar (10–20), and stored at 4 °C. A microbial smear was subsequently introduced into the artificial saliva solution for testing. The solution without microbial inoculation served as the control. The Candida albicans inoculum was prepared and standardized using the same methodology reported in our previous work16, ensuring consistency and reproducibility.
The sterilized orthodontic Ti–6Al–4 V alloy, serving as the working electrode, was immersed in artificial saliva at 37 °C under anaerobic conditions for varying durations (1 h, 24 h, 48 h, 96 h, 168 h, and 240 h). Following each immersion period, electrochemical impedance spectroscopy (EIS) and potentiodynamic polarization measurements were performed from − 1.2 to 0 V at a scan rate of 1 mV·s⁻¹. Electrochemical analyses were conducted using an SP-150 potentiostat (EC-Lab® software, BioLogic), employing its integrated software for data acquisition and curve fitting. A conventional three-electrode setup was used under conditions consistent with those described by El-Kamel et al.15,22. All electrochemical experiments were repeated in triplicate to ensure reproducibility and statistical robustness, with results reported as mean values and standard deviations where applicable.
To optimize and model the inhibition efficiency, Response Surface Methodology (RSM) was applied using a Central Composite Design (CCD) for experimental planning. Data analysis was performed with Minitab 19 software, and Analysis of Variance (ANOVA) was used to evaluate the statistical significance of the independent variables (immersion time and component presence) and their interaction effects. A second-order polynomial (quadratic) model was developed to predict the inhibition efficiency, with model adequacy assessed via the coefficient of determination (R²) and adjusted R-squared (R²_adj) values.
Surface characterization
Surface characterization of the Ti–6Al–4 V alloy was performed using a Quanta 250 FEG Scanning Electron Microscope (Field Emission Gun; FEI Company, Netherlands). The instrument operated at an accelerating voltage of 30 kV, offering magnifications ranging from 14X to 1,000,000X and a resolution of 1 nm.
Results and discussion
Characterization of Ti alloy electrode
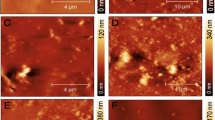
Figure 2. of SEM characterization reveals the surface morphology of Ti-6Al-4 V alloy as bare in Fig.2A with the absence of pepsin in Fig. 2B in the presence of pepsin and microbial group after immersion time. A compact biofilm formed due to the presence of pepsin as a protein in artificial saliva solution (Fig. 2B). Non-uniform C. albicans colony adsorption is observed at the alloy surface (Fig. 2C and D), with Fig. 2D showing a higher magnification (2000x) of C. albicans presence.
Key morphological features such as scratches, biofilm clusters, and corrosion-affected zones were visually annotated in Fig. 2 to facilitate interpretation of surface changes under different exposure conditions.
Electrochemical impedance spectroscopy (EIS) measurements
Figures 3 and 4 display the Nyquist and Bode plots derived from electrochemical impedance spectroscopy (EIS) of orthodontic Ti-6Al-4 V alloy samples immersed in artificial saliva, both with and without the presence of pepsin and Candida albicans, individually and combined. The impedance spectra consistently demonstrate pronounced capacitive behavior, with phase angles approaching a maximum near 80°23.
The Nyquist plots (Fig. 3) consistently exhibit incomplete semicircles, where a larger semicircle diameter corresponds to enhanced corrosion resistance. The data reveal that samples exposed to pepsin demonstrated superior corrosion resistance and inhibition compared to those with Candida albicans, which is characterized by its vigorous metabolic activity. Complementary Bode plots (Fig. 4) indicate that pepsin, as a protein, plays a significant chelating and barrier role, influencing the corrosion kinetics of the titanium alloy. This corrosion inhibition is attributed to the physical adsorption of pepsin molecules onto the alloy surface24]– [25. Furthermore, the protective blocking effect of pepsin intensifies with prolonged immersion time, effectively reducing the corrosion rate of the Ti-6Al-4 V alloy26.
Accumulation of biofilm formed by Candida albicans metabolites—including polysaccharides, proteins, and organic acids—enhances cellular adhesion and cohesion on both inert and active surfaces, facilitating stable hyphal attachment to the metal substrate.
A minor disruption was observed when Candida albicans coexisted with pepsin in the saliva solution, potentially reflecting conditions within the oral cavity where various microbial species can influence titanium-containing materials27,28,29. Orthodontic appliances provide an ideal surface for C. albicans adhesion and biofilm development, involving complex interactions. Pepsin actively inhibits the attachment of pathogenic microorganisms, alters local pH and microbial habitat, secretes antimicrobial agents, and modulates the host’s immune response. Additionally, fungal metabolic byproducts in the presence of pepsin may modify the microenvironment, potentially contributing to the corrosion of titanium alloys.
Previous studies30 have demonstrated that solutions with pH values below 2.5 exhibit significant antimicrobial activity against fungi, bacteria, and various microorganisms, with this effect diminishing at pH 3.0 and becoming negligible above pH 3.5. Additionally, research indicates no bactericidal effect within the pH range of 4.0 to 7.031. However, in the oral cavity affected by gastroesophageal reflux disease, the buffering action of pepsin elevates the pH to approximately 4.5, considerably higher than the stomach environment. Moreover, bacteria may be protected from acidic conditions by binding to pepsin, suggesting that the stomach’s acidic milieu does not effectively eradicate these microorganisms under such circumstances.
The experimental EIS data were analyzed by fitting to the equivalent electrical circuit depicted in Fig. 5. This circuit models the oxide film on the Ti-6Al-4 V alloy as a bilayer structure comprising an outer and an inner layer. In the model, Rs denotes the solution resistance, R1 represents the resistance of the outer oxide layer, and R2 corresponds to the charge transfer resistance at the inner layer. The elements Q1 and Q2 are constant phase elements (CPEs) associated with the double-layer capacitance and the passivation film, respectively. Notably, the charge transfer resistance R2 serves as a critical parameter directly related to the corrosion resistance of the alloy. The double-layer equivalent circuit model shown in Fig. 5 (R₁/Q₁ for the outer layer and R₂/Q₂ for the inner layer) was selected because titanium alloys typically form a duplex passive film consisting of an outer porous/hydrated oxide layer and an inner compact TiO2 barrier layer32-33.
The constant phase element (CPE) was employed instead of an ideal capacitor (C) to account for the surface heterogeneity and roughness of the alloy. The fitting parameters derived from this model, as detailed in Table 2, accurately represent the electrochemical behavior observed during the measurements34:
where Q is the magnitude of the CPE, j is the imaginary unit, ω is the angular frequency, and α is the CPE exponent. The fitting data is given in Table 2.
The alloys immersed in the simulated oral environment exhibited a passive state throughout the study. The increase in impedance values indicates an enhancement in the electrochemical resistance of the passive film under the combined action of pepsin and Candida albicans, suggesting improved protective behavior. The solution resistance (Rs) remained relatively low, attributable to the high ionic content of the saline medium. The increased resistance values indicate that biofilm formation contributed to the overall enhancement of the impedance response.
The inhibition efficiency is estimated from35– 36:
Where \(\:{R}_{T}^{o}\) and\(\:\:{R}_{T}\:\)represnt the total resistances of unexposed Ti and exposed Ti alloys to pepsin, Candida albicans; and both, respectively.
The inhibition efficiency of the titanium alloy progressively increased during immersion in artificial saliva containing pepsin, reaching a maximum of 87.4% after 240 h. In contrast, the combined presence of Candida albicans and pepsin in saliva resulted in a reduced inhibition efficiency of 55.6% at the same duration. Samples exposed to C. albicans alone in saliva maintained effective inhibition up to 96 h, after which a decline was observed, suggesting that while C. albicans initially contributes to surface protection, prolonged exposure may promote degradation. These findings imply a complex interplay between pepsin and C. albicans, where the protective influence of pepsin is attenuated by microbial activity over time. Moreover, C. albicans may evade low pH stress by binding to pepsin, thereby modulating its role in the corrosion behavior of the titanium alloy.
In the absence of protective agents, such as in saliva alone, the titanium alloy is directly exposed to the corrosive electrolyte, resulting in the highest corrosion rate due to the lack of any protective barrier film. Conversely, when pepsin or Candida albicans is present individually, the titanium surface experiences enhanced protection compared to their combined presence or saliva alone.
Pepsin, when present alone in artificial saliva, likely adsorbs onto the alloy surface forming a proteinaceous film that acts as a barrier, limiting the diffusion of corrosive ions toward the metal interface. Similarly, C. albicans alone forms a microbial biofilm on the titanium surface, serving as a physical shield that impedes the transport of aggressive species and restricts anodic dissolution. The reduction in anodic corrosion current observed in these cases indicates that both films effectively inhibit the anodic metal oxidation reaction, thereby retarding the corrosion rate.
However, the simultaneous presence of pepsin and C. albicans diminishes this protective effect, as evidenced by a more negative corrosion potential and elevated corrosion current density. This decline in inhibition may result from biochemical interactions in which pepsin’s proteolytic activity degrades components of the microbial biofilm matrix or compromises microbial cell integrity, leading to fragmentation or destabilization of the biofilm barrier. Additionally, metabolic byproducts of C. albicans in the presence of pepsin, such as organic acids, could locally acidify the environment, further accelerating the corrosion process by intensifying the aggressive conditions at the metal surface.
Potentiodynamic polarization measurements
To validate the EIS findings, Tafel polarization curves were recorded for bare Ti-6Al-4 V alloy immersed in artificial saliva containing pepsin, Candida albicans, and their combination after 10 days at 37 °C. Measurements were conducted within a potential range of − 1.2 to 0 V at a scan rate of 1 mV·s⁻¹. The resulting Tafel plots are presented in Fig. 6, while the derived polarization parameters—including corrosion potential (Ecorr) and corrosion current density (Icorr), obtained by extrapolating the linear regions of the curves—are summarized in Table 337. A trend of increasingly positive corrosion potentials was observed in the following order: pepsin + saliva > C. albicans + saliva > pepsin + C. albicans + saliva > saliva alone. Notably, the anodic corrosion currents were lower in the presence of either C. albicans or pepsin alone compared to the bare alloy and the combined presence of both agents, indicating superior corrosion inhibition under single-component exposure. This enhanced protection is likely attributable to the formation of a microbial or proteinaceous biofilm on the titanium surface, effectively reducing corrosion when either C. albicans or pepsin is present individually. These observations align well with the results obtained via EIS analysis. Furthermore, the most positive corrosion potential and minimal cathodic hydrogen evolution were recorded for the pepsin-only condition, confirming its significant protective effect as also evidenced by the EIS data.
In this study, artificial saliva containing Candida albicans exhibited the lowest corrosion current density of 79 ± 16 nA cm⁻² after 10 days of immersion, indicating a notably strong biological corrosion inhibition.
The corrosion characteristics of Ti-6Al-4 V alloy immersed in artificial saliva are significantly affected by the presence of pepsin and Candida albicans. These entities influence corrosion through intertwined biochemical and electrochemical mechanisms. Pepsin molecules adhere to the titanium alloy surface, creating a protective proteinaceous layer. This film serves as a physical barrier that impedes the ingress of corrosive ions such as chloride (Cl⁻) and protons (H⁺), thereby limiting their interaction with the metal substrate. This adsorption process can be generalized as follows:
The formation of this adsorbed layer obstructs anodic sites on the titanium surface, reducing the rate of metal dissolution and subsequently decreasing the corrosion current density.
Separately, C. albicans cells attach to the alloy surface and secrete extracellular polymeric substances (EPS), which mainly consist of polysaccharides, proteins, and organic acids. This secretion leads to the establishment of a biofilm:
The biofilm acts as a physical shield and alters the immediate microenvironment at the metal interface, potentially lowering corrosion by restricting the diffusion of aggressive ions.
The coexistence of pepsin and C. albicans results in an antagonistic synergy by turning the protective agents against one another. The synergistic acceleration of corrosion by pepsin and Candida albicans is driven by interfacial enzymatic degradation. Pepsin’s proteolytic activity compromises the protective fungal biofilm barrier, simultaneously releasing smaller peptides and acidic degradation products that act as corrosion promoters. This combined action creates a highly electrochemically active interface, which intensifies degradation and results in significantly higher overall corrosion compared to independent exposure conditions. The proteolytic function of pepsin at the simulated GERD pH actively and enzymatically degrades the structural polymers (proteins and polysaccharides) within the biofilm matrix:
This biochemical attack severely destroys the physical barrier function of the biofilm, immediately exposing the underlying titanium passive film to corrosive agents and driving the accelerated corrosion kinetics observed in the combined system.
Multivariate optimization
Analysis and optimization based on RSM methodology
The influence of pepsin and Candida albicans on inhibition efficiency was evaluated using Response Surface Methodology (RSM), a statistical and mathematical approach designed to model and analyze systems in which multiple variables influence a given response. Prior to applying RSM, a structured experimental design is required. In this study, a Central Composite Design (CCD) was employed, offering an efficient means of assessing potential interactions between variables and identifying optimal conditions, while minimizing the number of experimental trials and resource consumption38.
The experimental data obtained were analyzed using Minitab 19 software to perform an analysis of variance (ANOVA). Various predictive models, expressed in terms of coded factors, were developed to estimate the response values at specific levels of each variable. A three-level Central Composite Design (CCD) with two independent factors coded as − 1, 0, and + 1 was employed in this study. This design efficiently reduced the total number of experimental runs to 13, encompassing different point types for accurate model generation and response prediction.
Cube points: | 4 |
Center points in cube: | 5 |
Axial points: | 4 |
Center points in axial: | 0 |
The factor levels along with their corresponding actual values are summarized in Table 4, while the design of experiments (DOE), including both actual and coded values, is detailed in Table 5.
In the present study, Response Surface Methodology (RSM) was employed to investigate the interactive effects between the experimental variables, namely immersion time and component presence.
Analysis of Variance (ANOVA) was applied to identify the design parameters that significantly affect the inhibition efficiency. ANOVA also served to determine which model terms had a statistically meaningful influence on the experimental outcomes. Based on the Central Composite Design (CCD), the ANOVA results for the response surface model indicated that a quadratic model was well-suited for analyzing the experimental data, as summarized in Table 6.
The F-value (Fisher ratio) and the p-value are key indicators of the overall statistical significance of the model. A high F-value relative to a low p-value suggests that the model reliably explains the variability in the response and that the null hypothesis can be rejected. In this study, the model exhibited an F-value of 10.19 and a p-value of 0.004, which is below the significance threshold of 0.05. This confirms that the model is statistically significant and capable of predicting the optimal conditions for achieving maximum inhibition efficiency. Moreover, the quadratic term for factor B² displayed a p-value below 0.05 and an F-value of 29.83, indicating that this parameter has a substantial effect on the inhibition efficiency.
The adequacy of the model was assessed using the coefficient of determination (R²) alongside the adjusted R² value, as recommended in previous studies39. The obtained R² values of 79.30% and 87.92% for the inhibition efficiency response indicate a strong correlation and a satisfactory agreement between the experimental results and the values predicted by the model. These results confirm the model’s reliability and its suitability for accurately representing the system’s behavior.
The final regression equation, expressed in terms of coded factors, for predicting the inhibition efficiency is given as follows:
The final regression equation, expressed in terms of coded factors, for predicting the inhibition efficiency is presented below, where A represents the immersion time (hours) and B denotes the presence of components (pepsin and/or Candida albicans).
Surface response plots for Inhibition efficiency
The influence of the two factors—component presence and immersion time—on the response variable (IE) is illustrated through the three-dimensional surface plots shown in Figure 7a40.
When temperature is preserved persistent, IE increases with accumulative time and in pepsin addition components more than candida albicans and IE% decreasing with existence pepsin with candida at the same time. This results also confirmed with Contour plots of the combined effect of time and existence components variable on inhibition efficiency (Fig. 7b).
Factorial Plots (pareto chart of the standardization effect, interaction plot and main plots) are publicized in Figs. 8a), b) and c). Figure 8a) demonstrates the normal probability plot of the standardized effects for inhibition efficiency (IE%). Figure 8b) the interaction effect plot for the mean values of (IE%), which gained by DOE, which shows the main effect plot of IE% for Ti alloy in pepsin, c.albicans and both components at immersion time. Figure 8c) For immersion time, the IE% decreases till 120 h then increases when reaches 240 h. As for the presence of candida albicans only the IE% is high then decreases in the existence of both candida albicans and pepsin, the IE% decreased to the minimum. In the presence of pepsin, the IE% increased to the maximum.
Conclusions
This study demonstrates that the stability of orthodontic Ti-6Al-4 V alloys in pathological oral environments is critically dependent on component interactions. Pepsin alone provides significant, time-enhanced corrosion protection (IE = 87.4%) by forming a protective protein film. However, when co-present with C. albicans, pepsin’s proteolytic activity antagonizes the protective microbial biofilm, resulting in a synergistic corrosive effect that drastically lowers protection (IE = 55.6%). The RSM model confirmed that the component interaction is the most significant factor, offering a powerful predictive tool. These findings underscore the clinical importance of managing GERD in orthodontic patients to prevent premature implant failure.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Ohkubo, C., Hanatani, S. & Hosoi, T. Present status of titanium removable dentures—A review of the literature. J. Oral Rehabil. 35, 706–714 (2008).
Qu, Q. et al. Corrosion behavior of titanium in artificial saliva by lactic acid. Materials 7, 5528–5542. https://doi.org/10.3390/ma7085528 (2014).
Li, Y. et al. Improved strength and corrosion resistance of Ti–50Zr alloy through heat treatment. Adv. Eng. Mater. 27 (21), e202501828 (2025).
Pathak, T. K. et al. Influence of Ag, Au and Pd noble metals doping on structural, optical and antimicrobial properties of zinc oxide and titanium dioxide nanomaterials. Heliyon, 5 (3). (2019).
Trolić, I. M. et al. Corrosion of nickel-titanium orthodontic archwires in saliva and oral probiotic supplements. Acta Stomatologica Croatica, 51(4), p.316. (2017).
Aas, J. A., Paster, B. J., Stokes, L. N., Olsen, I. & Dewhirst, F. E. Defining the normal bacterial flora of the oral cavity. J. Clin. Microbiol. 43 (11), 5721–5732 (2005).
Gao, L. et al. Oral microbiomes: more and more importance in oral cavity and whole body. Protein cell. 9 (5), 488–500 (2018).
Talapko, J. et al. Candida albicans-The Virulence Factors and Clinical Manifestations of Infection. J. Fungi (Basel). 7 (2), 79. https://doi.org/10.3390/jof7020079 (2021).
Morad, H. O. J. et al. Thornton C.R. Pre-clinical imaging of invasive candidiasis using ImmunoPET/MR. Front. Microbiol. 9, 1996. https://doi.org/10.3389/fmicb.2018.01996 (2018).
Chen, H., Zhou, X., Ren, B. & Cheng, L. The regulation of hyphae growth in Candida albicans. Virulence 11, 337–348. https://doi.org/10.1080/21505594.2020.1748930 (2020).
Fass, R. & Frazier, R. The role of Dexlansoprazole modified-release in the management of gastroesophageal reflux disease. Th. Adv. Gastroenterol. 10 (2), 243–251 (2017).
El-Serag, H. B., Sweet, S., Winchester, C. C. & Dent, J. Update on the epidemiology of gastro-oesophageal reflux disease: a systematic review. Gut 63 (6), 871–880 (2014).
Wang, Y. J. et al. Salivary Pepsin as an intrinsic marker for diagnosis of Sub-types of gastroesophageal reflux disease and gastroesophageal reflux disease-related disorders. J. Neurogastroenterol Motil. 26 (1), 74–84. https://doi.org/10.5056/jnm19032 (2020).
Fruton, J. S. A history of Pepsin and related enzymes. Q. Rev. Biol. 77 (2), 127–147 (2002).
El-Kamel, R. S. & Fekry, A. M. Enhanced modified poly-tyrosine voltammetric sensor for the quantification detection of salivary Pepsin. International J. Biol. Macromolecules, 134178. (2024).
Mamdouh, S., El-Kamel, R. S., Fekry, A. M. & Ameer, M. A. Electrochemical evaluation of stainless steel alloy as a ureteral stent in synthetic urine solution in-vitro with Candida urinary tract infection. BMC Chem. 19, 224. https://doi.org/10.1186/s13065-025-01586-z (2025).
Ville, E., Carrière, F., Renou, C. & Laugier, R. Physiological study of pH stability and sensitivity to Pepsin of human gastric lipase. Digestion 65 (2), 73–81 (2002).
Bechir, F., Pacurar, M., Tohati, A. & Bataga, S. M. Comparative study of salivary pH, buffer Capacity, and flow in patients with and without gastroesophageal reflux disease. Int. J. Environ. Res. Public. Health. 19 (1), 201. https://doi.org/10.3390/ijerph19010201 (2021).
Asmara, Y. P., Siregar, J. P., Kurniawan, T. & Bachtiar, D. October. Application of response surface methodology method in designing corrosion inhibitor. In: IOP Conference Series: Materials Science and Engineering 257, 1, 012090. (IOP Publishing, 2017).
Medany, S. S., Elkamel, R. S., Abdel-Gawad, S. A. & Fekry, A. M. A novel Nano-Composite CSNPs/ PVP/CoONPs coating for improving corrosion resistance of Ti-6Al-4V alloy as a dental implant. Metals 12, 1784. https://doi.org/10.3390/met12111784 (2022).
Hussein, M. S. & Fekry, A. M. Effect of fumed silica/chitosan/poly (vinylpyrrolidone) composite coating on the electrochemical corrosion resistance of Ti–6Al–4V alloy in artificial saliva solution. ACS Omega. 4, 73–78 (2019).
El-Kamel, R. S., Ghoneim, A. A. & Fekry, A. M. Electrochemical, biodegradation and cytotoxicity of graphene oxide nanoparticles/polythreonine as a novel nano-coating on AZ91E Mg alloy staple in gastrectomy surgery. Materials Sci. Eng. C, 103, 109780. (2019).
Zhang, Y. et al. The effect of enzymes on the in vitro degradation behavior of Mg alloy wires in simulated gastric fluid and intestinal fluid. Bioactive materials, 7, 217–226. (2022).
Advincula, M., Fan, X. W., Lemons, J. & Advincula, R. Surface modification of surface sol–gel derived titanium oxide films by self-assembled monolayers (SAMs) and non-specific protein adsorption studies. Colloid Surf. B. 42, 29–43. https://doi.org/10.1016/j.colsurfb.2004.12.009 (2005).
Zeng, F. et al. Adsorption of Candida albicans on Ti-6Al-4V surface and its corrosion effects in artificial saliva. Bioelectrochemistry, 148, p.108248. (2022).
Cui, Y. W., Wang, L. & Zhang, L. C. Towards load-bearing biomedical titanium-based alloys: From essential requirements to future developments. Progress Materials Sci., 144, 101277. (2024).
Zhang, S. M. et al. Corrosion behavior of pure titanium in the presence of actinomyces Naeslundii. J. Mater. Sci. Mater. Med. 24 (5), 1229–1237 (2013).
Souza, J. C. et al. Corrosion behaviour of titanium in the presence of Streptococcus mutans. J. Dent. 41 (6), 528–534 (2013).
Fukushima, A., Mayanagi, G., Nakajo, K., Sasaki, K. & Takahashi, N. Microbiologically induced corrosive properties of the titanium surface. J. Dent. Res. 93 (5), 525–529 (2014).
Zhu, H., Hart, C. A., Sales, D. & Roberts, N. B. Bacterial killing in gastric juice – effect of pH and Pepsin on Escherichia coli and Helicobacter pylori. J. Med. Microbiol. 55, 1265–1270 (2006).
Waterman, S. R. & Small, P. L. C. Acid-sensitive enteric pathogens are protected from killing under extremely acidic conditions of pH 2.5 when they are inoculated onto certain solid food sources. Appl. Environ. Microbiol. 64 (10), 3882–3886 (1998).
Chu, Y. H. et al. Unveiling the contribution of lactic acid to the passivation behavior of Ti–6Al–4V fabricated by laser powder bed fusion in hank’s solution. Acta Metall. Sinica (English Letters). 37 (1), 102–118 (2024).
Pan, W. et al. Coupling AFCCX topology with volume fraction control to engineer electron beam melted high-performance lattice structures for potential orthopedic application. J. Materials Sci. Technol.. (2025).
Ghoneim, A. A., El-Kamel, R. S. & Fekry, A. M. Hydrogen evolution and quantum calculations for potassium sorbate as an efficient green inhibitor for biodegradable magnesium alloy staples used for sleeve gastrectomy surgery. Int. J. Hydrog. Energy. 45 (46), 24370–24382 (2020).
Fekry, A. M. Electrochemical behavior of a novel nano-composite coat on Ti alloy in phosphate buffer solution for biomedical applications. RSC Adv. 6, 20276–20285 (2016).
Fekry, A. M., El-Kamel, R. S. & Ghoneim, A. A. Electrochemical behavior of surgical 316L stainless steel eye glaucoma shunt (Ex-PRESS) in artificial aqueous humor. J. Mater. Chem. B. 4 (26), 4542–4548 (2016).
El-Gamel, N. E. & Fekry, A. M. Antimicrobial ruthenium complex coating on the surface of titanium alloy. High efficiency anticorrosion protection of ruthenium complex. Bioelectrochemistry 104, 35–43 (2015).
Chahid, A., Chafi, M., Essahli, M., Alrashdi, A. A. & Lgaz, H. Exploring the efficacy of Congo Red dye as a corrosion inhibitor for aluminum in HCl solution: An interdisciplinary study with RSM modeling and theoretical simulations. Arabian J. Chem., 17 (7), 105810. (2024).
Saigaa, N. et al. Optimization of the Inhibition corrosion of carbon steel in an acidic medium by a novel eco-friendly inhibitor asphodelus ramosus using response surface methodology. J. Electrochem. Sci. Eng. 13 (3), 469–490 (2023).
Khoobi, A., Soltani, N. & Aghaei, M. Computational design and multivariate statistical analysis for electrochemical sensing platform of iron oxide nanoparticles in sensitive detection of anti-inflammatory drug diclofenac in biological fluids. J. Alloys Compounds, 831,154715. (2020).
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
R.S. El-Kamel (RSE) and A.M. Fekry (AMF) jointly conceived and designed the study. RSE performed the electrochemical and surface characterization experiments. Both authors contributed to data analysis and manuscript preparation, with AMF providing critical revisions. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El-Kamel, R.S., Fekry, A.M. Pepsin-driven corrosion of orthodontic titanium alloys in candidiasis-simulated saliva: electrochemical and statistical insights. Sci Rep 16, 5937 (2026). https://doi.org/10.1038/s41598-026-36707-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-36707-8