Abstract
The objective of this paper is to analyze the species diversity change following the invasion of L. camara and grass coverage in the woodland in the Somaline Region, Ethiopia. To achieve the objectives, two adjusted sites were selected (invaded and non-invaded); at each site, a five-transect line with a distance of 500 m was established. In this transect line, 30 plots with a size of 20 m * 20 m and a distance of 200 m were established for collecting tree and shrub data, while seedlings, saplings, and herbaceous data were collected in a 1 m * 1 m plot. Diversity indices were used for the data analysis. The study result indicates that 23 species with 12 families of herb, grass, shrub, and tree species were identified in the study area. The comparison of the Shannon diversity index and evenness showed that the non-invaded site demonstrated more diversity as compared to the invaded site. Additionally, the correlation result indicates that the rate of L. camara invasion negatively correlates with the Shannon diversity index and evenness, with a correlation coefficient of -0.78 (p = 0.00) and − 0.73 (p = 0.00), respectively. Furthermore, grass and herb coverage: the non-invaded area has high grass and herb coverage with 77.7 ± 12.3, while the invaded area has 29.9 ± 17. In addition, the correlation results indicated that the rate of L. camara invasion had an adverse effect on grass and herb coverage. The invasion of L. camara leads to a decline in species diversity, the number of seedlings and saplings, and grass and herb cover in the study area. Consequently, future research explores the specific interactions between L. camara and native species to better understand the impact of this invasion on local ecosystems, as well as its effects on soil nutrients, microbial communities, and overall composition.
Similar content being viewed by others
Introduction
Invasive alien species (IAS) pose a significant threat to global ecosystems by altering community composition, biotic interactions, and other ecosystem processes1,2,3,4. These species jeopardize not only biodiversity but also food security, human and animal health, and economic development5. Additionally, they found that 17% of the global land area6. In Ethiopia, the spread of IAS has intensified over the past decade, with reports identifying over 35 such species7,8. Furthermore, anthropogenic factors such as population pressure, over-stocking, overgrazing, and deforestation have facilitated the IAS, exacerbating their impact on biodiversity9. Among the most problematic IAS in Ethiopia, Lantana camara L. is the major one10,11.
L. camara is a member of the Verbenaceae family, and is a pantropical weed affecting pastures and native forests in over 60 countries12. It thrives in diverse habitats and soil types, growing best in open, unshaded areas like degraded lands and tropical forests. In its native range in tropical America, it forms small clumps, whereas in its naturalized range, it can develop into dense, monospecific thickets up to 15 m tall13. It is recognized as one of the most aggressive invasive plant species globally, particularly in tropical and subtropical regions. Its introduction as an ornamental plant has led to significant ecological consequences, including the displacement of native flora and fauna, alteration of ecosystem dynamics, and negative impacts on agricultural productivity and biodiversity14,15,16. It is listed among the world’s 100 worst invasive species due to its extensive impact on native biodiversity and ecosystem functions17. Additionally, its invasion leads to the loss of native species through competition, recruitment limitations, and ecosystem alteration13,18,19,20,21,22.
In Ethiopia, L. camara was initially introduced as an ornamental plant but has since spread prolifically due to its prolific seed production and dispersal (Belayneh et al., 2016). Its invasion negatively affected the composition and richness of local plant communities14,23. Its dense thickets suppress native grasses and forages, causing adverse effects on livestock health, including gastrointestinal issues and reproductive problems24. The invasion of L. camara has led to noticeable changes in community composition and diversity, often decreasing native species richness as its cover increases25. Despite these documented impacts, research specific to the Somali region of Ethiopia is limited. Therefore, the present study aims to assess species diversity change following the invasion of L. camara and grass coverage in the woodland ecosystem.
Materials and methods
Description of the study area
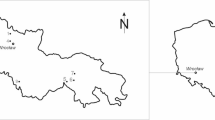
The study was undertaken in the Somali Regional State, Fafan Zone, in Golajo Kebele. Located at 9° 18’ 00” N latitude and 42° 40’ 12” E longitude, it is 650 km from the country’s capital city (Fig. 1). The average elevation of the zone is 1075 m above sea level26, and the study area covers 450 hectares. Thirty years of meteorological data from the Jigjiga center indicate that the mean minimum and maximum temperatures in the study area are 13.82 °C and 27.71 °C, respectively. The maximum temperature occurs in March, while the minimum temperature was recorded in January. The mean annual rainfall is 618.11 mm (Fig. 2). The rainfall conditions in the district are generally low, unreliable, and unevenly distributed. The soil is characterized by a mix of loam, sandy loam, and clay types, with loam being the most widespread (EPA, 2024). The vegetation of the study area is characterized by dry forests and woodlands, with Acacia-Commiphora woodland being the most widespread, with the dominant tree species such as Vachellia tortilis, Vachellia nilotica, Vachellia seyal, Balanites aegyptiaca, Vachellia albida, and Ziziphus spina-christi (EPA, 2024). The zone’s landscape is largely semi-arid, with a climate that supports a pastoralist lifestyle, which is the predominant way of life for the Somali population living there. In the study area, 90.9% of the population lived a lifestyle centered around pastoralism and semi-pastoralism.
Map of the study area(The map was developed by the first author using QGIS software version 3.4 (https://qgis.org/).
Vegetation data collection
In this study, systematic sampling method were used and the number of plots in the study site was determined based on the formula adopted27 as in the following.
Where, n= number of sample plot determined, AE= allowable error that is between 10 and 20%, CV= coefficient of variation, t = statistical sample of the t distribution for 95% level of confidence.
At each site (invaded and non-invaded), a five-transect line with a distance of 500 m was established. In this transect line, 30 plots with a size of 20 m * 20 m* and a distance of 200 m were established for collecting tree and shrub data, as described by Kent and Coker (1993). Additionally, within this main plot, five 1 m *1 m subplots (four corners and one center) were placed to collect data on seedlings and saplings (Fig. 3). Trees were considered to have a dbh ≥ 2.5 cm and a height exceeding 150 cm. In contrast, seedlings are considered regenerations with a height of less than 50 cm, while saplings are those with a height ranging from 50 to 150 cm (Manaye et al. 2019). The measurements were taken using a caliper and a Suunto hypsometer for dbh and height, respectively. The heights of seedlings and saplings were measured using a meter or a ruler. Woody species were identified both in the field and using available literature data, such as Bekele-Tesemma et al. (2007)28 and Vivero et al. (2005)29.
The percentage cover of herbaceous plants and grass coverage in each sample plot was estimated using wooden frames sized at 1 m x 1 m, which were subdivided into grids of 10 cm x 10 cm (Fig. 3). To assess herbaceous plant and grass coverage, the percentage of each grid section was recorded. The overall grass coverage area was calculated by averaging the coverage at each corner of the frame. For L. camara, its coverage was determined by measuring the average width and length of each patch within the plot. The total area covered by L. camara was then calculated by summing the areas of all patches and converting this sum into a percentage.
Data analysis
Diversity analysis
The data analysis was undertaken by R-software version 4.2.1 (Team, 2021), and Excel was used. Additionally, the woody plant diversity in the invaded and non-invaded areas was calculated using the Shannon–Wiener diversity index (H′) (Eq. 1), and a t-test was carried out to see the variation between the two sites. The species evenness was also calculated using the following formula (Eq. 2). The diversity of woody plants was calculated for each sample plot, and a correlation analysis (Pearson correlation) with the density of the invasive species (L. camara) was performed:
Analysis of vegetation structure
The vegetation structure in both invaded and non-invaded sites was analyzed based on the abundance, density, frequency, basal area, and ecologically significant species (the sum of relative densities, frequencies, and dominance) for each species. Basal area (BA), density (D), frequency (F), dominance (Do), and importance value index (IVI) data were calculated using the formula developed (Curtis and McIntosh (1951) to describe woody species structure in the forest.
Where BA is the basal area in square meters per hectare, d is the diameter at breast height in meters.
The importance value index (IVI) for each woody species was calculated as: importance value index (IVI) (%) = % relative dominance (RDo) + % relative density (RD) + % relative frequency (RF)
.
Result and discussion
Result
Diversity and species composition in the study area
Twenty-three species representing 12 distinct families of herbs, shrubs, and trees were recorded at the study site, and the list of identified species in the study area is presented in Table 1. Of these, 11 were trees, followed by five shrubs and herbs, with 5 and 2 grass species. Fabaceae is the most common family species; 35% of the species belong to this family, followed by Euphorbiaceae, Zygophyllaceae, Poaceae, and Solanaceae with 8%. The mean distribution of L. camara invasion within the study plots was approximately 51.4% (± 25.54), with coverage ranging from a minimum of 10% to a maximum of 90%.
Density of tree species and distribution
In comparing the number of tree species per hectare and the number of seedlings between two sites, the non-invaded area contains 127 trees and 14 seedlings per hectare. In contrast, the invaded area has 101 trees and 10 seedlings per hectare, respectively. Tree species such as V. tortilis and V. seyal tree species have the highest density of woody stems/ha in the invaded and non-invaded areas (Table 2). Important Value Index (IVI) analysis of the non-invaded area reveals that the vegetation structure is strongly dominated by V. seyal showed the highest IVI, indicating its ecological dominance through high relative basal area, frequency, and density. Other species such as V. tortilis, V. albida, and Z.spina-christi also show high IVI values, reflecting their strong contribution to the overall stand structure and distribution. These species form the core ecological framework of the ecosystem, supporting its stability and functioning. In contrast, species such as Sesbania sesban and Vachellia abyssinica recorded very low IVI values, indicating limited presence, low competitive ability, or restriction to specific microhabitats. In contrast, the IVI invaded area reveals that V. tortilis is overwhelmingly dominant, with an IVI of 200.16, far surpassing all other tree species. This high value in IVI reflects its substantial presence in terms of abundance, frequency, and basal area, indicating that it plays a major role in structuring the plant community. Other species, such as V. albida, V. seyal, and B.aegyptiaca, exhibit moderate IVI values ranging from 27.39 to 31.31, indicating that they are present but have a limited ecological influence compared to V. tortilis.
Diameter distribution regeneration status in invaded and non-invaded sites
The distribution of tree species by diameter class showed distinct patterns between the invaded and non-invaded sites (Fig. 4). At the invaded site, more trees occupied the higher diameter class. Conversely, at the non-invaded site, higher concentration of trees with diameters between 5 and 15 cm. Specifically, at the invaded site, trees within the smallest dbh class of 2–15 cm make up 29.6% of the total, while 71.4% of trees have a dbh exceeding 15 cm. Conversely, at the non-invaded site, 73.7% of trees belong to the smallest dbh class of 2–15 cm, and 36.3% of trees are found in the dbh class greater than 15 cm. The composition of seedling, sapling and shrub coverage indicates that, in non-invaded sites, saplings dominate with the highest coverage at 41.7%, followed by seedlings at 31.5% and shrubs at 26.9% (Fig. 5). On the other hand, invaded sites, shrubs are dominant, accounting for 51.5%, with saplings and seedlings following at 33.8% and 14.7%, respectively.
Species diversity within invaded and non-invaded sites
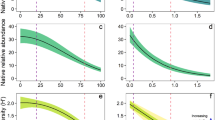
The diversity index measures like Evenness (E) and Richness (r), the non-invaded area has a higher value as compared to the invaded area with L. camara (Table 3). Whereas, in the Shannon (H) index value, the non-invaded area has a higher value than the invaded area, but this difference is not statistically significant. The Pearson correlation analysis indicated a negative relationship between the invasion of L. camara and both the Shannon diversity index and evenness, with correlation coefficients of −0.79 and − 0.73, respectively, at a significance level of p = 0.00) (Fig. 6). This implies that an increase in invasion corresponds to a decrease in the Shannon diversity index and evenness.
Grass coverage in the invaded and non-invaded area
The herbaceous species identified in the area are described in Table 1. A total of 7 herbaceous plants were identified. The grass and herb coverage of both areas is compared; accordingly, the mean grass coverage percentage in the non-invaded 77.7 ± 12.3, whereas in the invaded area it is 29.9 ± 17, and this difference is statistically significant (Fig. 7). Additionally, the result from Pearson correlation showed that the rate of L. camara coverage and grass and herb coverage have a negative correlation with the correlation coefficient of −0.88 (p = 0.00) (Fig. 6).
Discussion
Impact of L. camara on species composition and diversity
In the comparison of IVI, the non-invaded area is composed of tree species with 12 species, including V. abyssinica, E. tirucalli, P. aculeata, S. sesban, and Z. spina-christi, which only existed in the invaded area. In contrast, the invaded area is composed of six species. Additionally, the highest value IVI was taken by three species: V. seyal, Z. spina-christi, and V. tortilis, while the invaded area, V. tortilis, becomes extremely dominant with an IVI. Tree species like V. tortilis can cope with the aggression caused by L. camara. A recent investigation discovered that leaf extracts from V. tortilis impeded the germination and seedling development of the invasive species31. This trait may provide V. tortilis with a competitive advantage in ecosystems it has occupied by invasive species.
The tree diameter distribution showed that in the invaded area, the smaller dbh classes (2.5–5 cm and 5–10 cm) had relatively fewer trees, indicating a more mature tree population with a significant presence of larger trees. Whereas, in non-invaded sites, the larger dbh classes (20–25 cm, 25–30 cm, and > 30 cm) contain significantly fewer trees, suggesting a younger or regenerating forest with many small trees and fewer mature ones. This indicates that regeneration is better in the non-invaded site than in the invaded site. This result aligns with the findings of Barahukwa et al., 202332, and Vivian-Smith and Panetta, (2009)33, indicating that the invasion of L. camara hinders the regeneration of tree species. The dense proliferation of L. camara in invaded areas suppresses native tree growth by reducing their access to essential light and nutrients, which are critical for the survival of seedlings and saplings34. Additionally, the growth of seedlings and saplings is impeded by both the allelopathic effects of L. camara and the competition for resources (Barahukwa et al., 202332; Omeja et al., 201135; Wilson et al., 2014)36. Yet, the invaded area has a higher number of shrubs than the non-invaded area, as compared with the seedlings and saplings. Shrubs have the ability to adapt to light stress and can flourish even when they are given insufficient moisture levels37. However, their establishment is negatively impacted when they compete with invasive species like L. camara.
The diversity index showed that in all diversity measure indices, the non-invaded area has a higher diversity index than the invaded area. The finding is consistent with Bezabih et al., (2021)38, Qureshi et al., (2021)39, and Ruwanza (2020)40, indicating that areas invaded by L. camara show reduced diversity index values, including evenness (E) and the Shannon diversity index (H). Additionally, the presence of L. camara significantly diminishes ecological indices such as species richness, evenness, and overall diversity across a range of habitats41. Moreover, the increased percentage of L. camara invasion is correlated with a significant decline in ecological diversity, as evidenced by decreased values in key diversity indices such as the Shannon diversity index and Evenness. This indicates the expansion of L. camara disrupts the structural composition of native forest ecosystems, resulting in a reduction in tree species richness and evenness a shift toward community dominance by a limited number of tolerant species. The invasive nature of L. camara is further exacerbated by its ability to thrive in disturbed environments, where it often outcompetes native species for resources42,43. The implications of such invasions extend beyond biodiversity loss; they also affect ecosystem services and agricultural productivity, posing challenges to land management and conservation efforts44,45. Invasive species like L. camara can profoundly affect species diversity and ecosystem health. Its ability to dominate landscapes, coupled with its allelopathic effects and resource competition, underscores the need for effective management strategies to mitigate its impacts on native biodiversity.
Impact of L. camara in grass coverage
The invaded area has lower grass and herb coverage as compared to the non-invaded site. The presence of L. camara has been correlated with reduced grass and herb overage. Similar finding were observed by Mandiporeva et al., (2023)46; and Wilson et al., (2014)47, where the invasion of L. camara led to reduced grass and herb coverage. Furthermore, it was identified as one of the most troublesome invasive species, particularly in areas where it outcompeted native grasses, leading to significant reductions in grassland habitats48. Additionally, by outcompeting native grasses for resources such as light, water, and nutrients, leading to a marked decline in grass coverage in invaded areas21,34. The allelopathic properties of L. camara further exacerbate this issue, as it releases chemicals that inhibit the growth of surrounding plant species, including grasses (Sharma, 2011; Sharma et al., 2005)25. Changes in habitat structure due to dense L. camara thickets can lead to changes in microclimates, affecting soil moisture and temperature, which are critical for the survival of native grass species (Sengupta Dash, 202434; Sharma, 2011). Grasslands are vital for soil conservation, carbon sequestration, and providing habitat for various wildlife species. The decline in grass coverage can disrupt these functions, leading to broader ecological consequences49. In the study area, the community practices semi-pastoralism, needs rangeland for cattle grazing, and thus, the invasion will affect the grazing land.
Conclusions
The study’s findings indicated a difference in the diversity index between sites that were invaded and non-invaded by L. camara. The non-invaded site demonstrated more diversity as compared to the invaded site. Moreover, the non-invaded area contained a greater number of seedlings and saplings than the invaded area. Additionally, the invaded area has less grass coverage than the non-invaded one. These observations suggest that the invasion of L. camara leads to a decline in species diversity, the number of seedlings and saplings, and grass and herb cover in the study area. Consequently, we recommend that future research explore the specific interactions between L. camara and native species to better understand the impact of this invasion on local ecosystems, as well as its effects on soil nutrients, microbial communities, and overall composition.
Data availability
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
References
David, P. et al. Elsevier,. Impacts of invasive species on food webs. in Advances in Ecological Research vol. 56 1–60 (2017).
Gallardo, B., Clavero, M., Sánchez, M. I. & Vilà, M. Global ecological impacts of invasive species in aquatic ecosystems. Glob. Change Biol. 22, 151–163 (2016).
Schirmel, J., Bundschuh, M., Entling, M. H., Kowarik, I. & Buchholz, S. Impacts of invasive plants on resident animals across ecosystems, taxa, and feeding types: a global assessment. Glob. Change Biol. 22, 594–603 (2016).
Impact of Biological Invasions on Ecosystem Services (Springer International Publishing, 2017). https://doi.org/10.1007/978-3-319-45121-3.
McNeely, J. A. A Global Strategy on Invasive Alien Species. Gland and Cambridge (2001).
Early, R. et al. Global threats from invasive alien species in the twenty-first century and National response capacities. Nat. Commun. 7, 12485 (2016).
Shetie, G. The ecological distribution and socio-economic impacts of prosopis juliflora (Sw.) DC. the AmibaraWoreda, Afar National Region State. Addis Ababa, Ethiopia (2008).
Tamiru, G. Invasive alien weed species distribution, impacts on agriculture, challenge and reaction in ethiopia: A review. Journal Biology 7, 136-146 (2017).
EBI. Threats Caused by IAS – Ethiopian Biodiversity Institute – EBI. (2025). https://ebi.gov.et/biodiversity/conservation/invasive-species/threats-caused-by-ias/
Seifu, A. Impact of invasive alien plant, xanthium strumarium, on species diversity and composition of invaded plant communities in borena zone, Ethiopia. BIJ 1, (2017).
Tessema, T., Ulrichs, C. & Buettner, C. Invasive Alien Plant Species in Ethiopia: Impacts, Challenges and Responses. (2009).
Parsons, W. T. & Cuthbertson, E. G. Noxious Weeds of Australia (CSIRO publishing, 2001).
Swarbrick, J. T., Willson, B. W. & Hannan-Jones, M. A. The biology of Australian weeds 25. Lantana Camara L. Plant Prot. Q. 10, 82–82 (1995).
Assefa, A. S. & Molla, E. L. Impact of invasive alien plant species, Lantana camara, on the species composition, richness and evenness of invaded plant communities in Sidama, Gamogofa and Gedio Zones, Ethiopia. Preprint at (2021). https://doi.org/10.21203/rs.3.rs-943114/v1
Singh, A., Khudsar, F., Nag, S. & Adhikari, B. Ecological and socioeconomic impacts of Lantana Camara invasion: A case study in central India. Indian J. For. 46, 187–199 (2024).
Verma, S. Medicinal potential of Lantana camara: verbenaceae. J. Drug Delivery Ther. 8, 62–64 (2018).
Lowe, S., Browne, M., Boudjelas, S. & De Poorter, M. 100 of the World’s Worst Invasive Alien Species: A Selection from the Global Invasive Species Database Vol. 12 (Invasive Species Specialist Group Auckland, 2000).
Bhatt, Y. D., Rawat, Y. S. & Singh, S. P. Changes in ecosystem functioning after replacement of forest by Lantana shrubland in Kumaun himalaya. J. Veg. Sci. 5, 67–70 (1994).
Dobhal, P. K., Kohli, R. K. & Batish, D. R. Evaluation of the impact of Lantana camara L. invasion, on four major woody shrubs, along Nayar river of Pauri Garhwal, in Uttarakhand Himalaya. (2010).
Fensham, R. J., Fairfax, R. J. & Cannell, R. J. The invasion of Lantana Camara L. in Forty Mile scrub National Park, North Queensland. Aust. J. Ecol. 19, 297–305 (1994).
Kohli, R. K., Batish, D. R., Singh, H. P. & Dogra, K. S. Status, invasiveness and environmental threats of three tropical American invasive weeds (Parthenium hysterophorus L., ageratum conyzoides L., Lantana Camara L.) in India. Biol. Invasions. 8, 1501–1510 (2006).
Sharma, G. P., Raghubanshi, A. S. & Singh, J. S. Lantana invasion: an overview. Weed Biology Manage. 5, 157–165 (2005).
Solomon, C. A. & Adane, A. T. Impact of invasion: A case study on the ecological and socioeconomic impact of Lantana Camara (L.) in Abay millennium park (AMP), Bahir Dar, Ethiopia. J. Ecol. Nat. Environ. 7, 132–145 (2015).
Bufebo, B. & Elias, E. Impact of Lantana Camara L. on plant diversity and soil physicochemical characteristics in Wollo floristic region. Ethiopia 6, 22-31 (2018).
D’Antonio, C. M. & Vitousek, P. M. Biological invasions by exotic grasses, the grass/fire cycle, and global change. Annu. Rev. Ecol. Syst. 23, 63–87 (1992).
Hussein, H. A., Ahmed, J. M., Musse, A. H. & Gizaw, Y. Prevalence and risk factors of bovine tuberculosis in cattle in selected districts of Fafan pastoral settings, Eastern Ethiopia. Heliyon 10, e24998 (2024).
Baidoo, A., Abugre, S. & Sarfo, D. A. Forest Measurement and Inventory. (2018). https://doi.org/10.13140/RG.2.2.35794.66240 doi:10.13140/RG.2.2.35794.66240.
Bekele-Tesemma, A., Birnie, A. & Tengnas, B. Useful trees and shrubs for Ethiopia. Reg. Soil. Conserv. Unit. (RSCU) Swed. Int. Dev. Auth. (SIDA). 2, 123–127 (2007).
Vivero, J. L., Kelbessa, E. & Demissew, S. The red list of endemic trees and shrubs of Ethiopia and Eritrea. (2005).
Curtis, J. T. & McIntosh, R. P. An upland forest continuum in the prairie-forest border region of Wisconsin. Ecology 32, 476–496 (1951).
Lolimo, F. N., Ondeng’i, B. O. & Odhiambo, K. O. Allelopathic effects of vachellia nubica, vachellia tortilis and hyphaene compressa on germination and seedling growth of prosopis Juliflora. Asian J. Environ. Ecol. 23, 26–40 (2024).
Barahukwa, A. et al. The effects of the invasive species, Lantana camara, on regeneration of an African rainforest. Afr. J. Ecol. 61, 451–460 (2023).
Vivian-Smith, G. & Panetta, F. D. Lantana (Lantana camara) seed bank dynamics: seedling emergence and seed survival. Invasive plant. sci. Manag. 2, 141–150 (2009).
Sengupta, R. & Dash, S. S. Impacts and status of invasion by Lantana Camara in natural forests of an Indo-Burma biodiversity hotspot region- A case study in Mizoram, India. Res. Ecol. 1–10. https://doi.org/10.30564/re.v6i3.6343 (2024).
Omeja, P. A. et al. Intensive tree planting facilitates tropical forest biodiversity and biomass accumulation in Kibale National Park, Uganda. For. Ecol. Manag. 261, 703–709 (2011).
Wilson, G., Desai, A. A., Sim, D. A., Gruber, M. A. M. & Lester, P. J. The association between invasive Lantana Camara and seedlings/saplings of a plant community in Mudumalai tiger Reserve, India. J. Trop. Ecol. 30, 551–563 (2014).
Kumar, A. et al. Investigating the phenology and interactions of competitive plant species co-occurring with invasive Lantana Camara in Indian Himalayan region. Sci. Rep. 14, 400 (2024).
Bezabih, B., Gobezie, T. & Hassen, S. Impact of Lantana Camara L. on plant diversity and soil physicochemical characteristics in Wollo floristic region. Ethiopia 6, 22-31 (2021).
Qureshi, H. et al. Multiple comparisons of diversity indices invaded by Lantana Camara. Braz J. Biol. 81, 83–91 (2021).
Ruwanza, S. Effects of Lantana Camara invasion on vegetation diversity and composition in the Vhembe biosphere Reserve, Limpopo Province of South Africa. Sci. Afr. 10, e00610 (2020).
Bhatta, S., Hejda, M. & Pyšek, P. Impact of invasive plants on vegetation in protected areas of Nepal. Biol. Invasions. 26, 3745–3759 (2024).
Dalu, T., Stam, E. M., Ligege, M. O. & Cuthbert, R. N. Highways to invasion: powerline servitudes as corridors for alien plant invasions. Afr. J. Ecol. 61, 379–388 (2023).
Shackleton, R. T., Witt, A. B., Aool, W. & Pratt, C. F. Distribution of the invasive alien weed, Lantana camara, and its ecological and livelihood impacts in Eastern Africa. Afr. J. Range Forage Sci. 34, 1–11 (2017).
Gautam, S. & Sharma, A. Invasive weed Lantana utilization for textile finishes. Indian J. Weed Sci. 54, 192–196 (2022).
Goncalves, E. et al. Global invasion of Lantana camara: has the Climatic niche been conserved across continents? PLoS ONE. 9, e111468 (2014).
Mandiporeva, J., Gamundani, T. P., Gurajena, B., Mpakairi, K. & Magqina, T. Spatial distribution and effects of Lantana Camara invasion on soil properties and native vegetation community composition in Tugwi-Mukosi recreational Park, Masvingo, Zimbabwe. Afr. J. Ecol. 61, 185–203 (2023).
Wilson, G., Gruber, M. A. M. & Lester, P. J. Foraging relationships between elephants and Lantana Camara invasion in Mudumalai tiger Reserve, India. Biotropica 46, 194–201 (2014).
Panedy, K., Poudel, G., Neupane, A., Acharya, K. R. & Adhikari, S. Status of invasive alien plant species in urban forest of Hetauda, Nepal. Forestry: J. Inst. Fores Nep. 18, 107–118 (2021).
Sampson, C. et al. Effects of illegal grazing and invasive Lantana Camara on Asian elephant habitat use. Biol. Conserv. 220, 50–59 (2018).
Acknowledgements
We are indebted to the financial support of Ethiopia Forestry development Dire Dawa Center. We are grateful for the support provided for all individuals who participate during the data collection.
Funding
This work was financially supported by Ethiopia Forestry development Dire Dawa Center.
Author information
Authors and Affiliations
Contributions
Abu Mulatu : Methodology, data collection Investigation, analysis, Writing - original draft, Kebad Edmealem and Habtamu Achenif : data collection, and writing Shiferaw Alem and Abeje Eshete : Reviewing & editing, analysis reviewing & editing, Supervision and Conceptualization.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The collection of plant material and the performance of experimental research on such plants complied with the national guidelines of Ethiopia.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mulatu, A., Edmealem, K., Tesema, H.A. et al. Species diversity and grass cover change following the invasion of Lantana camara in a woodland ecosystem. Sci Rep 16, 7677 (2026). https://doi.org/10.1038/s41598-026-39006-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-39006-4