Abstract
This paper focuses on how regulatory bodies respond to artificial intelligence (AI)-enabled medical devices. To achieve this, we present a comparative overview of the United States (USA), European Union (EU), and China. Our search in the governmental database identified 59 AI medical devices approved in China as of July 2023. In comparison to the rules-based regulatory approach in China, the approaches in the USA and EU are more standards-oriented.
Similar content being viewed by others
Introduction
The resurgence of interest in deep learning (DL), coupled with advancements in training techniques and hardware, has significantly contributed to the widespread adoption of artificial intelligence (AI). This use of AI is also gradually changing medical device related practice. The main factors contributing to this situation are the ongoing explosion in the availability of digital health data, including image data, sensor time series, text, medical records, financial transactions, etc. Moreover, there is a broad access to increasingly powerful and cost-effective computing infrastructure. Data are the key ingredient of all machine learning systems. However, even with massive datasets, often referred to as “Big Data”, the data alone are of little use until meaningful knowledge and insights are extracted from them1. Therefore, the era of ‘big data’ provides opportunities for AI, and more powerful and cheaper processing ability enables these opportunities to become reality.
The healthcare sector has adopted AI technologies in developing medical products, software, workflows, solutions, and decision-making processes to relieve doctors’ ever-expanding working burdens and improve patient outcomes. Academic progress in this area has paralleled the significant investments and activities within the industry. It is apparent the traditional way of working in the medical field will inevitably be transformed by AI and will keep evolving in the coming years at a rapid pace. However, the real-world clinical implementation of AI-based technologies in medicine has not yet become a reality by 20192. Even as of now, interest and research from various stakeholders in the use of AI in medicine have not fully translated to widespread adoption in practice. To understand the barriers on the path to real implementation, this paper seeks to delve deep into the landscape of approved AIMDs, which are commercial AI solutions available for sale and ready for use in clinical practice. The analysis aims to explore why the number of approved AIMDs does not proportionally align with the abundance of research papers. It also investigates how these devices are evaluated before regulatory approvals, the response actions taken by regulatory agencies, and additional considerations beyond the regular experiments reported in research papers.
The approval of medical devices, along with associated regulations and legislation, is handled differently in various regions worldwide. In the United States (USA), the US Food and Drug Administration (FDA) is responsible for approving commercially marketed medical devices. In China, the corresponding department responsible for approval is the National Medical Products Administration (NMPA). Unlike the USA and China, medical devices in the EU are not approved in a centralized way. Instead, there are many Notified Bodies eligible to conduct conformity assessments and issue a Conformité Européenne (CE) mark. To cope with the submission and approval request of AI-enabled medical devices, competent authorities have announced their acts on how to regulate medical devices with the AI algorithms involved, followed by the publication of specific regulatory policies and registration review guidelines. While existing studies3,4,5,6,7 predominantly discuss the regulatory environment in the USA and EU, analyzing FDA-approved and CE-marked AIMDs, the issues surrounding AI are naturally universal. China is also a major player in global AI due to its significant research base and large population, high-demand for improving clinical work efficiency, and vastly available systematically collected data that can be utilized by AI training algorithms. However, there are few studies on the regulatory compliance requirements in China, potentially due to language barriers. To bridge this gap, we propose including AIMDs approved in China in our study and conducting a comparative analysis systematically. Furthermore, we provide insights into Chinese legislation and regulations. It is expected that this paper is able to contribute to a clearer global map of AIMDs regulations and legislation, especially in the context of Chinese practices and products, and serves as a valuable reference. Table 1 presents a comparative overview of medical device regulation in the USA, Europe, and China. Tables 2 and 3 summarize the definitions and classifications of AIMD in various countries.
From AI to AIMDs
Although AI dates back to 1956 at Dartmouth, it has developed into a very broad concept and has not yet converged to a well-accepted unified definition. It continues to evolve with the vision of achieving super-human performance. However, from a long-term perspective, we are still at an early stage. When it comes to AIMDs, AI-empowered medical devices are relatively new to researcher’s investigation. The explosive success in many application domains including computer vision and natural language processing, has triggered various research motivations to equip the traditional clinical practice with AI tools. This open up an area of inquiry where it is possible to improve the workflow efficiency and facilitate the early diagnosis by replacing several procedures with AI assistants. As an umbrella word, “AI” is also sometimes being studied under the name of machine learning (ML). Therefore, AI and ML will be interchangeably used in this paper. In addition, NMPA has specifically pointed out that they would not make a strict distinguishment between AI and ML to medical devices.
In general, AI can be classified into model-based and data-based types, and the AI used in AIMDs normally belongs to the latter. Remarkably, data-based AI methods present an additional benefit thanks to the availability of medical data. According to the presence of data label, AI can be also grouped into four types, namely supervised-learning-based, unsupervised-learning-based, semi-supervised-learning-based and reinforcement-learning-based methods. When an AI algorithm is intended to be developed into an AIMD, acting as a special assistant in clinical practice, its intrinsic characteristics associated with AI have to be considered carefully, including ethics, explainability, reliability, privacy, bias, fairness, adaptability, reproducibility and so on.
Methods
To gain a deeper understanding of approved AIMDs, we conduct a mini-review of current research analyzing approved AIMDs. While most existing research works focus on interpreting and analyzing the regulatory approaches for AI medical device approvals in the USA and Europe3,4,5,6,7, this paper aims to expand this analysis by including China, thereby providing a more global perspective on AIMDs development. In 2019, the Center for Medical Device Evaluation (CMDE) of NMPA, issued a key document on the topic: “Key Review Points for Deep Learning Assisted Decision-Making Medical Software”. This document provided comprehensive review requirements covering the entire AI product lifecycle, from requirement analysis and algorithm development to verification and validation stages. In 2022, CMDE released a revised version titled “Artificial Intelligence Medical Device Technical Review Guideline”. Additionally, CMDE has been developing disease-specific review guidelines, detailing more specific requirements for devices with particular functions. Concurrently, the National Institutes for Food and Drug Control (NIFDC), another division of the NMPA, is formulating standards to support these guidelines by specifying how to meet the requirements. Although currently recommended, these standards could potentially become mandatory in the future. Figure 1 demonstrates a non-exhaustive list of the progress made by the China regulatory body in regulating AIMDs. In order to determine the approved AIMDs across the global market, we aim in this section to identify and analyze the AIMDs approved in China as of July 2023. A common challenge in assessing AIMDs is the lack of structured, publicly available AIMD databases. Therefore, we applied the following specific search strategies and selection criteria to conduct analysis. There are no human or animal participants in this study and informed consent or ethical approval is not required.
Data source, search strategy and selection criteria
We included AIMDs approved in China as of July 25, 2023.
Although there is not a specific database for AIMDs, the official NMPA website hosts a comprehensive database of all registered medical devices8. The challenge, however, is that this database’s search engine only allows queries based on product code, name, and company name, none of which directly indicate AI involvement. This limitation makes it impossible to search specifically for terms like “Artificial Intelligence” or “Machine Learning” to filter for AIMDs. Thus, on one hand, directly acquiring the official AIMD product list is not feasible. On the other hand, the database only supports search entry using a keyword, making it impossible to pull the entire medical device list. Manually reviewing the entire database of medical devices is also impractical.
To address this challenge, we leveraged NMPA guidelines on medical device naming, which require AIMDs to include specific components in their names, such as the type of input data (e.g., CT scan, fundus photograph), target disease, intended use (e.g., computer-assisted detection/triage/diagnosis/assessment), etc. Utilizing this naming convention, we performed keyword searches in the NMPA database with terms like “computer assisted detection,” “computer assisted triage,” “computer assisted diagnosis,” and “computer assisted assessment,” followed by a manual review of the results.
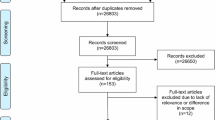
To ensure our list of AIMDs was as complete as possible, we used every target disease identified in the initial search results as keywords for subsequent searches. Additional online sources, such as AIMDs analytical reports posted on the international medical society websites, manufacturer advertisements, and news articles, were also reviewed. We further verified our findings by searching for the manufacturer name or device name on the NMPA website, followed by a manual review to assess AI relevance. However, the manual review and identification process might be hampered by the brevity of the attached summary tables on the NMPA website, which are typically less than a page and provide insufficient detail to determine AI relevance. While the NMPA does publish more comprehensive multi-page review reports for certain devices, only a few review reports are selected for publication rather than reports for all registered devices. These reports, when available, contain detailed information, including safety summary, clinical performance, algorithms used, validation methods, and the number of sites involved. Insufficient public information makes identification more difficult. In cases where it was unclear, we investigated the manufacturer’s website for more clarification. Devices that lacked sufficient information, even after consulting both NMPA and manufacturer websites, and those with vague or scarce information, were excluded from our selection (see Fig. 2).
During the review process, both two rounds of manual review were conducted independently by authors Y.L. and W. Y. to try to obtain a more consistent result. For any identifications in doubt, the authors would review again and discuss them together in a face-to-face manner until they reach a consensus.
Through our selection process, we collected a total of 59 approved devices that met our selection criteria
Data extraction
For all selected AIMDs, the following characteristics were collected: device name, manufacturer name, approval date, intended use, origin city of the manufacture, and device classification. We also extracted information regarding the medical specialty each device is associated with, the specific type of AI algorithm employed, and the body area targeted by the device. Supplementary Table 3 lists all identified NMPA approved AIMDs and their characteristics.
Results
Our search identified a total of 59 AIMDs, which formed the final cohort for our study. The number of AIMDs approved by the NMPA has steadily increased: from 9 devices in 2020, marking the year of the first AI-enabled medical device approval, to 13 in 2021, and further to 25 in 2022. In the first half of 2023 alone, up to July 27, 12 additional AIMDs had been approved (shown in Fig. 3). It is important to note that these 59 devices are all classified as Class III devices by the NMPA. Eight (13·56%) out of 59 devices did not mention AI or any other terms that could imply the presence of an AI algorithm. Among the rest of devices, the majority, 50 (84·74%), specified the use of deep learning, and only one device employed the region growing technique, which is one of the most popular AI algorithms for segmentation tasks, as described in the review report published by the NMPA. For eight such unclear cases, as illustrated in Fig. 2, another round of manual review was conducted by consulting their official websites to determine whether they belong to AIMDs. All 59 devices were designed for professional use, with none intended for patient use. In terms of medical specialties, 22 (37%) of the 59 AIMDs were approved for radiology, followed by 10 devices for cardiovascular applications, eight devices for ophthalmology, six devices for neurology, five devices for orthopedics, three devices for internal medicine, two devices for hematology, two devices for endocrinology and one device for therapeutic radiology (see Fig. 3).
Features shown in the figure include approval years, descriptions of AI-enabled medical technologies, and related medical specialties. NMPA National Medical Products Administration, AIMD artificial intelligence medical device, AI/ML artificial intelligence and machine learning, CT Computed Tomography, FFR Fractional Flow Reserve, ECG Electrocardiogram.
Figure 4 demonstrates the geographic distribution of cities where manufacturers have developed approved devices. It can be seen that they are mainly concentrated in Beijing (23 devices), Shanghai (nine devices), Shenzhen (eight devices) and Hangzhou (eight devices). Through the figure, it can be observed that among the top four cities with large number of approved AIMDs, three out of them happen to be first-tier cities in China, of which there are only four in total. In comparison to other cities, the first-tier cities are recognized for their high level of development and substantial economic activities. They typically have a relatively high concentration of academic institutions and healthcare facilities with research units. As for the fourth city, Hangzhou in Zhejiang, it does not belong to first-tier cities. However, it is known for its large number of internet companies and robust tech industry, largely due to the presence of major companies like Alibaba.
Figure 5 provides a visualization of main manufacturers leading the development of AIMDs and their numbers of approved AIMDs for each type. Out of 59 devices, 40 (67·79%) were developed by AI companies that have had more than one device approved. For instance, Shukun has nine devices approved by the NMPA, covering lung nodule detection, pneumonia triage, rib fracture detection, Fractional Flow Reserve (FFR) computation, intracranial hemorrhage triage, coronary artery stenosis triage, ischemic stroke assessment, head and neck assessment. DeepWise follows with six approved devices, then InferVision, United Imaging, and Carewell, each with four devices. The most common applications of these 59 devices include computer-aided software for detection, triage, assessment, and diagnosis. Other applications involve FFR computation (two devices), contouring of the region of interest, and surgery planning (one device each). Supplementary Table 4 details the target disease distribution of the approved AIMDs, covering approximately 19 medical scenarios, including lung nodule detection with 11 medical devices (18·6%), pneumonia triage with six medical devices (10·1%), eye disease diagnosis with eight (13·5%) medical devices (six devices on Diabetic retinopathy, one device on glaucoma and one device on both), fracture detection with six (10·1%) medical devices, ECG with four (6·7%) medical devices from the same company Lepu Medical (Carewell), etc. Of these medical devices, the majority is based on CT scan, with others utilizing fundus photograph, X-ray, ECG, endoscopic image, pathology image, etc., as shown in Fig. 6.
The types are classified by the hardware and the fill color gradation in each tile represents different numbers of AIMDs which are added to each tile. The top plot shows the total number of approved AIMDs for each manufacturer. AIMD artificial intelligence medical device. CT Computed Tomography, ECG Electrocardiogram, X-ray X-radiation.
Comparison between the USA and China
As indicated in Table 1, the absence of an official database for AIMDs approved in Europe presents significant challenges in collecting and comparing associated information. Therefore, this paper focuses on a comparative analysis between the USA and China. For a detailed analysis of Europe, interested readers are referred to previous studies7, with a comprehensive review available in the supplementary Tables 1 and 2. According to the list published by the FDA on October 5, 2022, 521 devices have been included, with the first approval dating back to 1995. This number substantially exceeds the 59 AIMDs approved by the NMPA. Despite potential underestimation of the number of AIMDs in China, there is still a large gap between the total number of devices at the FDA and NMPA. Another contributing factor is the different interpretations and definitions of AI; a broader definition by the FDA has led to more AI-enabled medical devices being approved since 1995. Besides, the start year of AI adoption in healthcare in China is not as early as that in the USA. In China, the first AIMD was not proposed until 2020, known for its FFR computation using deep learning algorithms. From the information provided by the NMPA, it is observed that 50 out of the 59 devices explicitly mention the use of deep learning, which indicates that the scope of AIMDs in China is largely confined to deep learning. Whereas, the FDA approved AIMDs might not only cover the third-generation AI (DL-based), but also the first-generation (rule-based) and second-generation AIs (ML-based).
The analysis of AIMD approvals
A common trend observed is the significant approval of AIMDs for radiology use, with 23 devices (39%) in China and 392 devices (75·2%) in the USA. This trend can be attributed to the continuously growing volume of radiological imaging data, which disproportionately exceeds the number of available trained radiologists9. The primary motivation behind the development and integration of AI-enabled tools in radiology has been to alleviate the increasing workload on radiologists and to enhance clinical care efficiency. The remarkable success of deep learning algorithms in image detection, segmentation, and classification tasks has led to the wider application of AI in computer-assisted diagnostics to meet clinical needs. Compared to other medical specialties, radiology data are more readily accessible and provide a rich resource for scientific and medical exploration.
During our study period, seven devices received approval from both the FDA and the NMPA. Notably, six of these were first approved by the FDA in the USA before gaining approval in China, as shown in Fig. 7. The exception is Keya’s DeepVessel FFR, which received NMPA approval first. A previous study indicated that most AI-based medical devices commonly receive CE marking in Europe and subsequent approval in the USA, initially gaining approval in Europe10. It was suggested that the FDA’s regulatory approach for medical devices may be more rigorous than Europe’s. Following this logic, companies might prefer to first seek FDA approval before pursuing approval in China. However, it should be noted that the approval time is neither reflecting strictness nor efficiency of a country’s regulation. In addition, we also identified the home-based countries of the companies, which ideally might be a related factor to their first approval county. It turned out that all of their home-based countries were China, and there was not much correlation with the approval time.
Among the 521 AI-based medical devices with FDA approval, 500 (96%) were approved through the 510(k) regulatory pathway, 18 through the De-novo pathway, and only three via the Premarket approval pathway. However, it is noteworthy that all devices were classified as Class II in the USA. In contrast, all the identified AIMDs approved in China were reviewed as Class III devices. Such a difference in the class distribution might be due to the difference in AIMDs classification between the USA and China. Specifically, the determination of AIMD categories relies on a combination of the intended purposes of the product and the maturity of the algorithm used. If the algorithm has low maturity (i.e., not approved before or its safety and effectiveness have not been proved before) and is used for decision-support purposes, then the associated AIMDs should be classified as class III types. Since the first AIMD approved by NMPA was in 2020, most algorithms are new in the field of medical devices, thus being classified as class III.
China’s regulatory framework includes point-by-point requirements during the review process, as outlined in regulations and standards, aiming to establish a comprehensive life-cycle supervision model. While all regulatory bodies require manufacturers to provide evidence of safety and performance, the NMPA has published a review guideline detailing how manufacturers should provide such evidence. This guideline includes explicit checklists for manufacturers to follow, encompassing the entire algorithm development process from algorithm specification, data collection, algorithm selection and training, to algorithm testing, and validation. It essentially requires developing a ‘digital twin’ of the algorithm to ensure traceability, enabling digital oversight of the entire process and the potential to reproduce the algorithm model. This can be interpreted as using “rules” approach, while FDA uses “standards” approach. In rules approach, rules or formulas are well established to simplify and speed up the review process, and it can be less noisy, i.e. more consistent for easier comparison. Whereas for US, standards approach may provide more room for manufacturer for innovations. Such difference is in line with the fact that most devices try to obtain FDA approval first.
The analysis of regulatory requirements
The NMPA review guideline places special emphasis on an evidence-based approach relying on data, highlighting the significance of data sufficiency, diversity and representativeness, and addressing data bias in AI algorithm development. Most AI algorithms, particularly those based on deep learning, are heavily data-driven. With the same model structure, the use of different training data can lead to vastly different model parameters. Therefore, training a model on an “inappropriate” dataset could result in poor performance. The guidelines strongly recommend using training data from multiple sources, such as various hospitals, and ensuring balance across disease subtypes and demographic subgroups. This is consistent with the findings from a previous study6, highlighting deep learning models at a single site alone can mask weakness in the models and lead to worse performance across multiple sites. In that case study, significant performance drop-offs were observed when comparing within-site tests to evaluations on other sites. To prove data sufficiency, the guideline recommends depicting a curve of model performance over the amount of training data used to demonstrate the model performance across increasing number of training size. As the size of the training data increases, the model performance is expected to improve and eventually stabilize, which implies that enough data has been provided. Regarding algorithm testing, while balance in the testing data distribution is not required, it should reflect real-world scenarios by ensuring high prevalence similarity in testing data to the target population.
In addition, stratified performance evaluations across disease subtypes, device types, different geographical regions, and demographic subgroups are specified for analysis. By quantifying how AI performs across diverse populations, it helps to explore the factors (e.g., device types, disease types, demographic groups) that might impact the model performance, thereby improving the explainability of the devices and gaining a more comprehensive understanding. Moreover, Unlike the general performance evaluation on the testing data, which is the most essential part and mandatory for each AIMD, adversarial and stress tests are advisable if applicable, to explore the potential of AI models. Besides the above functional performance evaluation (e.g. sensitivity and specificity for classification tasks, DICE metric for segmentation tasks), non-functional performance evaluation such as timeliness is also important to report and submit. With the increasing complexity of AI algorithms, running time can vary significantly, while waiting time is always an important indicator for user satisfactory.
Noted that before data is ready for AI model development, the data preparation stage that includes data collection, data organization, data preprocessing and data annotation, is also an indispensable part of the AI product lifecycle. All these steps are related to data quality, and data annotation should be given special attention since the performance of the developed AI models directly depends on the quality of data annotation. Thus, the details of how the dataset is curated and annotated are required to be specified as well.
We believe that the regulatory strategy, which requires recording every possible useful piece of information generated during the algorithm development process, is crucial for advancing transparency and enhancing understanding of these medical devices among various stakeholders (e.g., users, buyers, researchers). This rich information, encompassing data aspects, safety, and clinical performance, not only enables learning from the strengths and weaknesses of the medical devices but also facilitates more intuitive comparisons between devices with similar applications. The NMPA has even developed regulation documents for specific diseases, pre-defining the information that manufacturers need to provide and the performance indicators that should be evaluated. As illustrated in Fig. 1, the review guideline outlines general principles, while the standards serve as complementary instructions, and the disease/application-specific regulations detail the variations in requirements across different device types. By adhering to consistent guidelines for similar devices, the information provided by manufacturers becomes uniform, simplifying comparative analysis. However, a common issue in both the USA and China is that this information is currently accessible only to regulators and reviewers, with the public receiving vague and limited details. More transparency from the FDA and NMPA would be beneficial, offering companies and researchers a clearer understanding of the current landscape, potential gaps for clinical improvement, and enabling stakeholders to compare and assess these tools more effectively for informed decision-making regarding AI commercial solutions for clinical use. The recent surge in academic progress has been matched by a flurry of activity in companies, with a multitude of commercial AI-based solutions now available (as shown in Fig. 3). For instance, 11 devices have been approved for lung nodule detection and 8 devices for diagnosing diabetic retinopathy or chronic glaucoma. Without sufficient information from the NMPA, it is challenging for radiologists to choose the most suitable commercial AI solution for their specific needs.
Even though there is a growing trend of AIMDs approvals, the number of approved AIMDs is still far smaller than the AI models reported in research. In research, there are also guidelines followed by researchers for reporting and assessing the risk of bias in AI models, such as TRIPOD-AI11, SPIRIT-AI12, CONSORT-AI10, DECIDE-AI13 and so on. These reporting guidelines are being developed in close collaboration with key stakeholders consisting of clinicians, computer scientists, journal editors, researchers, industry leaders, regulators, funders, policy makers, patient groups and so on, to generate a consensus applicable across the community. Furthermore, groups from typically underrepresented regions are also engaged to ensure the guidelines are applicable and international across a global scale14. By contrast, different regulatory agencies use highly variable methods for definition, production, approval, marketing and postmarketing surveillance. In addition, guidelines typically focus on a part of development pathway for AI systems with an emphasis on scientific evaluation and reporting, whereas government regulations cover the whole lifecycle supervision, paying attention to practical implementation and ethic aspects. A recent review also placed the legal and regulatory aspects in the group of key elements of ethics15. Moreover, government regulations must be complied with through a series of systematic review before AIMDs approval, and the approved AIMDs are held under the sole accountability of the manufacturer. There was a study finding out that, among their study cases, journal publications did not acknowledge the key limitations of the available evidence identified in regulatory documents16. Given the somewhat different focuses of research guidelines and regulatory assessment, as well as the differences across regulatory jurisdictions35, there are gaps between the guidelines and government regulations. These gaps are part of the reason why the number of approved AI models is far smaller than the number published in research. It is also important to note that, besides regulations, which serve as a downstream gatekeeper for the AI industry, upstream factors in the AI supply chain, such as AI base model development and dataset quality, could possibly be another part of the reason that need to be aware of.
Discussion
In our study in China, 59 AIMDs were identified in the NMPA search, all classified as class III. The number of AIMDs has been significantly increasing since 2020, the year when the first AIMD was approved by the NMPA. The most common application for these AIMDs is in radiology. Although NMPA-approved AIMDs share similarities with CE-marked devices in Europe and FDA-approved devices in the USA, such as a growing trend in approvals and a focus on radiology, there are still many differences between them. One major difference is the smaller number of AIMDs approved in China compared to Europe and the USA. This disparity is partly due to the broader definition of AI and the earlier start year of AIMDs in the USA, which leads to more AI-enabled medical devices. For instance, the first AI-enabled device was approved by the FDA in 1995, whereas in China, it was not until 2020. Another key difference is that all NMPA-included AIMDs are class III, implying a higher risk level than the AI-enabled medical devices approved by the FDA, which are mostly class II. This reflects NMPA’s more cautious approach towards AI-empowered medical devices. The NMPA uses “rules” approach by imposing specific requirements and pays close attention to aspects like data quality, annotation, representativeness, algorithm description, and performance evaluation, including both functional and non-functional aspects like generalization ability, robustness, and timeliness. The functional performance evaluation encompasses overall average performance in corresponding metrics (e.g. sensitivity and specificity for classification and DICE for segmentation), stratified performance, and, if applicable, adversarial and stress tests. By contrast, FDA uses “standards” approach, and such difference may also contribute to another observed trend where six out of seven commonly approved AIMDs by both the FDA and NMPA first received approval in the USA before China, since FDA tends to provide manufacturers more diversity room for review innovations. Despite the NMPA’s emphasis on transparency and the importance of submitting comprehensive information, they have not opted to publish more detailed information. Consequently, we are unable to fully leverage these rich resources for AI development in clinical practice. More transparency in the review comments is anticipated to help researchers to determine the directions and methods for developing AI algorithms to improve upon the existing ones. We hope that with the constant improvement of the AIMD review framework and process, more information will become accessible to researchers of interest. In addition, a more complete AIMD review ecosystem could not only facilitate an increase in the number of AIMDs but also expand new AI applications in the medical field and push the boundaries of AIMDs in the near future.
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
Code availability
No code was used to compute the results. The underlying code for drawing the figures is available on request from the corresponding author.
References
Ghahramani, Z. Probabilistic machine learning and artificial intelligence. Nature 521, 452–459 (2015).
He, J. et al. The practical implementation of artificial intelligence technologies in medicine. Nat. Med. 25, 30–36 (2019).
Pesapane, F., Volont´e, C., Codari, M. & Sardanelli, F. Artificial intelligence as a medical device in radiology: ethical and regulatory issues in Europe and the United States. Insights imaging 9, 745–753 (2018).
Zhu, S., Gilbert, M., Chetty, I. & Siddiqui, F. The 2021 landscape of FDA-approved artificial intelligence/machine learning-enabled medical devices: an analysis of the characteristics and intended use. Int. J. Med. Inform. 165, 104828 (2022).
Benjamens, S., Dhunnoo, P. & Mesk´o, B. The state of artificial intelligence-based FDA-approved medical devices and algorithms: an online database. NPJ Digit. Med. 3, 118 (2020).
Wu, E. et al. How medical AI devices are evaluated: limitations and recommendations from an analysis of FDA approvals. Nat. Med. 27, 582–584 (2021).
Muehlematter, U. J., Daniore, P. & Vokinger, K. N. Approval of artificial intelligence and machine learning-based medical devices in the USA and Europe (2015–20): a comparative analysis. Lancet Digit. Health 3, e195–e203 (2021).
NMPA. Medical Devices Database. https://www.nmpa.gov.cn/datasearch/search-result.html (2024).
Hosny, A., Parmar, C., Quackenbush, J., Schwartz, L. H. & Aerts, H. J. Artificial intelligence in radiology. Nat. Rev. Cancer 18, 500–510 (2018).
Liu, X. et al. Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension. Lancet Digit. Health 2, e537–e548 (2020).
Collins, G. S. et al. TRIPOD + AI statement: updated guidance for reporting clinical prediction models that use regression or machine learning methods. BMJ 385, e078378 (2024).
Rivera, S. C. et al. Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension. Lancet Digit. Health 2, e549–e560 (2020).
Vasey, B. et al. Reporting guideline for the early stage clinical evaluation of decision support systems driven by artificial intelligence: DECIDE-AI. BMJ 377, 924–933 (2022).
Sounderajah, V. et al. Developing specific reporting guidelines for diagnostic accuracy studies assessing AI interventions: The STARD-AI Steering Group. Nat. Med. 26, 807–808 (2020).
Kolbinger, F. R., Veldhuizen, G. P., Zhu, J., Truhn, D. & Kather, J. N. Reporting guidelines in medical artificial intelligence: a systematic review and meta-analysis. Commun. Med. 4, 71 (2024).
Naci, H. et al. Design characteristics, risk of bias, and reporting of randomised controlled trials supporting approvals of cancer drugs by European Medicines Agency, 2014-16: cross sectional analysis. BMJ 366, l5221 (2019).
Premarket Approval (PMA). https://www.fda.gov/medical-devices/premarket-submissions-selecting-and-preparing-correctsubmission/premarket-approval-pma (2022).
FDA. Premarket Notification 510k. https://www.fda.gov/medical-devices/premarket-submissions-selecting-and-preparing-correct-submission/premarket-notification-510k (2023).
FDA. De Novo Classification Request. https://www.fda.gov/medical-devices/premarket-submissions-selecting-and-preparing-correct-submission/de-novo-classification-request (2022).
FDA. US FDA Artificial Intelligence and Machine Learning Discussion Paper. https://www.fda.gov/media/122535/download (2019).
FDA. Artificial Intelligence and Machine Learning in Software as a Medical Device. https://www.fda.gov/medical-devices/software-medical-device-samd/artificial-intelligence-and-machine-learning-software-medical-device (2024).
FDA. Good Machine Learning Practice for Medical Device Development: Guiding Principles. https://www.fda.gov/medical-devices/software-medical-device-samd/good-machine-learning-practice-medical-device-development-guiding-principles (2021).
FDA. Computer-Assisted Detection Devices Applied to Radiology Images and RadiologyDevice Data - Premarket Notification [510(k)] Submissions. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/computer-assisted-detection-devices-applied-radiology-images-and-radiology-device-data-premarket (2022).
FDA. Clinical Performance Assessment: Considerations for Computer-Assisted Detection Devices Applied to Radiology Images and Radiology Device Data in Premarket Notification (510(k)) Submissions. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/clinical-performance-assessment-considerations-computer-assisted-detection-devices-applied-radiology (2022).
FDA. Developing a Software Precertification Program: A Working Model. https://www.fda.gov/media/119722/download (2019).
EU. Medical Devices. https://health.ec.europa.eu/medical-devices-sector_en (2024).
EU. Types of legislation. https://european-union.europa.eu/institutions-law-budget/law/types-legislation_en (2024).
EU. New Regulations. https://health.ec.europa.eu/medical-devices-sector/new-regulations_en (2024).
EU. New EU Rules to Ensure Safety of Medical Devices. https://health.ec.europa.eu/system/files/2020-07/md_generic_fs_en_0.pdf (2020).
EU, Guidance-MDCG endorsed documents and other guidance. https://health.ec.europa.eu/medical-devices-sector/new-regulations/guidance-mdcg-endorsed-documents-and-other-guidance_en (2024).
EU. Proposal for a Regulation laying down harmonised rules on artificial intelligence. https://digital-strategy.ec.europa.eu/en/library/proposal-regulation-laying-down-harmonised-rules-artificial-intelligence (2021).
FDA. Artificial Intelligence and Machine Learning (AI/ML)-Enabled Medical Devices. https://www.fda.gov/medical-devices/software-medical-device-samd/artificial-intelligence-and-machine-learning-aiml-enabled-medical-devices (2024).
NMPA. A Notice for NMPA releasing an Artificial Intelligence Medical Devices Review Guideline. https://www.cmde.org.cn/xwdt/shpgzgg/gztg/20220309090800158.html (2022).
EU. Medical Device Regulation 2017/745, Article 51, classification of devices. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:02017R0745-20230320 (2017).
Author information
Authors and Affiliations
Contributions
Y.L. contributed to study design, data collection and analysis, and drafted the manuscript. W.Y. contributed to study design, data interpretation of the manuscript, supervision, manuscript review & editing. T.D. provided revisions. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, Y., Yu, W. & Dillon, T. Regulatory responses and approval status of artificial intelligence medical devices with a focus on China. npj Digit. Med. 7, 255 (2024). https://doi.org/10.1038/s41746-024-01254-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41746-024-01254-x
This article is cited by
-
Current challenges and the way forwards for regulatory databases of artificial intelligence as a medical device
npj Digital Medicine (2026)
-
Artificial Intelligence-Based Medical Devices for Diabetic Retinopathy Screening in the European Union
Ophthalmology and Therapy (2026)
-
Radiology AI and sustainability paradox: environmental, economic, and social dimensions
Insights into Imaging (2025)
-
SEM model analysis of diabetic patients’ acceptance of artificial intelligence for diabetic retinopathy
BMC Medical Informatics and Decision Making (2025)
-
An artificial intelligence cloud platform for OCT-based retinal anomalies screening system in real clinical environments
npj Digital Medicine (2025)