Abstract
Sleep supports memory consolidation, but the specific roles of different sleep stages in this process remain unclear. While rapid eye movement sleep (REM) has traditionally been linked to the processing of emotionally charged material, recent evidence suggests that slow wave sleep (SWS) also plays a role in strengthening emotional memories. Here, we use targeted memory reactivation (TMR) during REM and SWS in a daytime nap to directly examine which sleep stage is primarily involved in consolidating emotional declarative memories. Contrary to our hypothesis, reactivating emotional stimuli during REM impairs memory. Meanwhile, TMR benefit in SWS is strongly correlated with the product of time spent in REM and SWS. The emotional valence of cued items modulates both delta/theta power and sleep spindles. Furthermore, emotional memories benefit more from TMR than neutral ones. Our findings suggest that SWS and REM have complementary roles in consolidating emotional memories, with REM potentially involved in forgetting them. These results also expand on recent evidence highlighting a connection between sleep spindles and emotional processing.
Similar content being viewed by others
Introduction
Sleep supports memory consolidation, which allows the stabilization and integration of recently formed labile memories, thus enhancing their recall1. However, it is unclear how different sleep stages, with their distinct physiology, contribute to this process. For declarative memories, the predominant contemporary models implicate primarily non-REM sleep (NREM) and particularly slow wave sleep (SWS)2,3. However, there is strong evidence indicating a role for rapid eye movement sleep (REM) as well4. Instead of a NREM-REM dichotomy, alternative but compatible accounts, such as the sequential hypothesis5 and others6, ascribe the memory benefit of sleep to successive NREM-REM episodes.
Emotional charge of memories allows better remembering, including the associated contextual information7. Processing of emotional material during sleep has been traditionally attributed to REM8. Activation of the amygdala and other relevant emotion-processing brain regions during REM, increased cortisol activity in the second half of the night, and alterations in REM measures in psychiatric disorders have been taken as indirect evidence of this REM dependency9. Consistent with this view, studies have shown higher retention of emotional explicit memory following REM-rich late-night sleep but not after early sleep10,11,12,13 as well as in SWS-deprived participants but not in those deprived of REM14. In addition, REM measures correlated positively with increased retention of emotional declarative memories12,15,16,17,18. Based on these findings, it was hypothesized that REM provides a unique milieu that facilitates the consolidation of memory for emotional experiences19. However, this hypothesis is challenged by studies that found no association of REM with emotional declarative memory performance20,21,22,23,24,25,26. More importantly, increasing evidence suggests that NREM is involved in emotional memory consolidation. Two studies found NREM, with or without REM, to support emotional declarative memory consolidation27,28. Furthermore, several studies showed correlations between the benefit of sleep for emotional memories with time spent in SWS23,29,30,31 and spectral power in the delta band30. Beyond correlations, pharmacologically increasing slow wave activity32 and sleep spindle density20 enhanced memory for emotional items.
Another line of evidence for the role of NREM in emotional declarative memory consolidation comes from targeted memory reactivation (TMR) studies. TMR, which relies on replaying cues associated with recently encoded memories during sleep, has been shown to enhance declarative memory retention33,34. In one TMR study, emotional and neutral memories were cued during SWS35. While there were no overall memory benefits, SWS duration and number of spindles were both associated with better subsequent performance (shorter reaction times) for emotional items. In another study that cued emotional and neutral items in NREM or REM, memory for emotional items was improved after cueing in NREM but not REM36. Furthermore, successful cueing was associated with increases in spindle and theta power. In contrast, another study found no effect of TMR in NREM on emotional memory37.
In this study, we used TMR to examine which sleep stage is primarily involved in the consolidation of emotional declarative memories. Given the prevailing view in the literature on the role of REM in emotional memory consolidation, we hypothesized that replay of cues for emotional items during REM would lead to enhanced retention compared to replay in SWS. We also explored whether emotional memories are retained better than neutral memories overall. We hypothesized that replay of cues for emotional items would lead to greater memory improvements than the cueing of neutral items.
Results
Here, we present the results from the immediate retest (T2; see Methods). Results from the delayed retest (T3) are included in the Supplementary Materials.
Memory performance
Effect of TMR on memory within groups
We examined the effect of reactivation on percentage change in error from T1 (%ΔError)within each group using linear mixed-effects models (LMMs). In these models, %ΔError was included as the dependent variable, and ‘Reactivation’ (reactivated vs. non-reactivated) was included as a fixed factor. Compound symmetry covariance structure was used for the repeated measures (‘Reactivation’), with ‘Subject’ included as a random effect.
In the group in which emotional items were reactivated during REM (E-REM), there was a significant main effect for ‘Reactivation’ (β[SE] = –0.11 [0.05], t(24) = 2.13, p = 0.04), but contrary to our expectations, reactivated items displayed a larger increase in error (22% for reactivated items vs. 11% for non-reactivated items), indicating that REM cueing was associated with memory impairment (Fig. 1A, Supplementary Table 2). There was no significant reactivation effect on %ΔError in the group in which emotional items were reactivated during SWS (E-SWS; β[SE] = 0.017[0.07], t(23) = 0.24, p = 0.81) or the group in which neutral items were reactivated during SWS (N-SWS;β[SE] = 0.03[0.05], t(30) = 0.57, p = 0.57) (Fig. 1A, Supplementary Table 2).
Comparison of TMR effects on memory between groups
In the LMMs that compared the effect of reactivation on %ΔError between groups (E-REM vs. E-SWS, and E-SWS vs. N-SWS), ‘Reactivation,’ ‘Group’ and ‘Group × Reactivation’ interaction were included as fixed effects. The product of time spent in SWS and REM (%SWS × %REM; ‘SWS×REM product’) was included as a covariate, and its interactions with ‘Reactivation’ and ‘Group’ were included as fixed factors because this measure was correlated with cueing benefit (CB; see “Correlations with Sleep Measures”). Compound symmetry covariance structure was used for the repeated measures (‘Reactivation’), and ‘Subject’ was included as a random effect. In the analysis that included E-SWS vs. E-REM, there was no ‘Group × Reactivation’ interaction (β [SE] = 0.16 [0.14], t(45) = 1.12, p = 0.27), but a significant ‘Reactivation × Group × SWS×REM product’ interaction (Supplementary Table 3). On the other hand, in the analysis that included E-SWS with N-SWS, ‘Group × Reactivation’ interaction was significant (in addition to a significant three-way interaction), with reactivation of emotional items showing a larger memory benefit compared to neutral items (β [SE] = 0.39[0.12], t(49) = 3.36, p = 0.002) (Fig. 1B, Supplementary Table 3).
In summary, emotional memories were impaired when reactivated in REM. In addition, emotional memory reactivation in SWS produced a larger memory benefit than the reactivation of neutral memories.
Correlations with sleep measures
To examine the associations with measures of sleep architecture and cue-evoked EEG activity, the memory benefit of reactivation was indexed as CB38, calculated as: %ΔError for the non-reactivated - %ΔError for the reactivated items. Higher CB indicated better remembering for reactivated items compared to non-reactivated items. The correlation of time spent in SWS and REM (%SWS and %REM) was examined with CB in the reactivation groups (E-SWS, E-REM and N-SWS) and %ΔError in E-Nap. Because there is evidence suggesting that NREM and REM may have complementary roles in memory consolidation, we also examined the correlations with SWS×REM product39,40,41.
In the E-SWS group, CB showed a strong correlation with SWS×REM product (spearman’s rs = 0.66, p = 4 × 10-4; Fig. 2A). No such significant correlation was found in E-REM (r = –0.04, p = 0.87) or N-SWS (r = –0.29, p = 0.14). In E-SWS also, CB was also correlated with %REM (r = .43, p = 0.03), although this correlation did not survive Bonferroni correction. In the E-Nap group, %ΔError was positively correlated with %REM (spearman’s rs = 0.46, p = 0.04; Fig. 2B), suggesting that REM was associated with impairment of memory for emotional items. However, this correlation also did not survive correction for multiple comparisons. No other significant correlations were observed.
A In the E-SWS group, SWS×REM product was strongly correlated with CB. No such significant correlation was found in E-REM or N-SWS. B In E-Nap, there was a trend for a significant correlation of %REM with increased %ΔError, suggesting that REM was associated with forgetting. This result is consistent with the impairment in memory for items that were reactivated during REM. Dotted lines indicate 95% confidence interval.
Baseline memory, sleep architecture, and recall in nap and wake groups
By design, recall at baseline (T1) for items that would subsequently be reactivated did not differ significantly from the recall of those that would not be reactivated in any of the groups (all p values >0.50) (Supplementary Table 4). There was also no difference in recall at baseline between groups (F = 1.91, p = 0.15). Finally, we compared the sleep architecture between reactivation groups and found a higher %REM in E-REM than in E-SWS (p < 0.001) and N-SWS (p = 0.01) (Supplementary Table 5). The higher %REM in the E-REM group may be due to the fact that only participants with REM that were stable and long enough to allow a full round of reactivation were included in this category. It is also possible that the reactivation of emotional memories during REM may have further increased the time spent in this sleep stage42,43. There was no difference in %ΔError between E-Nap and E-Wake (t(40) = 1.44, p = 0.16) (Fig. S1).
Cue-evoked EEG activity
To investigate cue-related modulation of EEG activity during sleep, we first identified clusters of cue-induced activity that were significantly different from zero (see Methods). In the E-SWS group, we identified two time-frequency clusters in which the cues induced increased activity (p < 0.05, FDR adjusted; Fig. 3A). The first cluster (2.0–8.5 Hz), extending from 235–975 ms after cue onset (ending just before the end of the 1-s auditory cue), comprises activity in the canonical delta and theta bands. The second cluster (11.6–19.1 Hz), occurring 638–1475 ms following cue onset, corresponds broadly with the sigma (spindle) band. This pattern of increased delta/theta and spindle band activity following the replay of emotional sounds during SWS replicates findings in other studies of cueing during SWS36,44,45.
Time-frequency response to cue presentation during SWS at electrode Cz, in E-SWS (A) and N-SWS (B). Time zero represents the initiation of sound presentation during sleep. Black contour lines highlight significant clusters of activation (p < 0.05, FDR adjusted). In E-SWS, the average valence of the emotional sounds cued during sleep was correlated with the cluster-averaged time frequency activity in the 2–8 Hz response (delta-theta; C). Delta-theta power also showed a positive correlation with CB, which did not survive correction for multiple comparisons (D). The valence of emotional sounds was also significantly correlated with the spindle band (12–19 Hz) response (E), which suggested that emotional valence modified spindle activity. This was further supported by the higher power in the same cluster in response to emotional sounds compared to neutral sounds (E-SWS vs. N-SWS; F; means and standard errors are displayed). **p < 0.01.
The valence of the emotional sounds (as determined from our pilot study; see Supplementary Materials) played during sleep was positively correlated with the delta-theta power (r = 0.56, p = 0.006; Fig. 3C). Delta-theta power also showed a positive correlation with CB, which did not survive Bonferroni correction (r = 0.47, p = 0.03; Fig. 3D). Interestingly, the valence of the emotional sounds played during sleep was also significantly correlated with the magnitude of the spindle band response (r = 0.55, p = 0.008; Fig. 3E), indicating that the emotional charge of the stimuli modified spindles. This was further supported by the significantly higher power in the spindle cluster in the E-SWS group compared to the N-SWS group (t(41.9) = 2.99, p = 0.005; Fig. 3F). Spindle power was not significantly correlated with CB in any of the groups. When cueing during REM, we observed an increase in power across a broad frequency range throughout the entire post-cue period, but the increase was only significant in alpha-beta frequencies (9.4–26.5 Hz) from 1375 to 1725 ms following cue onset (Fig. 4). Power in the identified cluster did not correlate with CB (r = –0.11, p = 0.64). Similarly, EEG activity following REM cueing was not related to stimulus valence (r = 0.12, p = 0.61).
No significant clusters emerged at the <0.05 (FDR corrected) level in N-SWS (Fig. 3B).
Association of sleep fragmentation and arousal with memory
A recent study showed that participants who were awakened by reactivations showed memory impairment for cued items46. To see whether the memory impairment in the E-REM group was associated with sleep disruptions, we examined the correlation of CB with arousal index (AI; number of arousals/minute47;), sleep fragmentation index (SFI; number of sleep stage transitions/minute48;) and the duration of wake time after sleep onset (WASO). None of these revealed a significant association (AI: spearman’s rs =–0.29, p = 0.18; SFI: spearman’s rs = 0.12, p = 0.58; WASO: spearman’s rs = 0.26, p = 0.21).
Another recent TMR study found an inverse correlation of cueing benefit with cue-evoked power in the beta band and a strong trend for a similar correlation with power in the alpha band49. To examine if a similar mechanism might explain the memory impairment for the reactivated items in E-REM, we took the absolute difference in spectral power between the pre-and post-cue periods in the alpha (8–11 Hz) and beta (17–21 Hz) bands as an EEG-measure of cue-related arousal49. There were no correlations between these measures and the CB in the E-REM group (alpha: r = –0.15, p = 0.52; beta: r = –0.23, p = 0.34).
Discussion
We applied TMR in SWS and REM to test the hypothesis that consolidation of emotional memories occurs primarily in REM. Contrary to our expectations, reactivation of emotional stimuli during REM led to poorer recall. In addition, the memory benefit of emotional memory reactivation during SWS was strongly correlated with the SWS×REM product. We also found that reactivation in SWS led to a larger memory benefit for emotional memories than for neutral ones. Finally, delta/theta activity and sleep spindles were modulated by the emotional charge of memories.
Memory impairment following cuing in REM was unexpected, yet it was consistent with the trend for more REM to be associated with impaired recall in the nap-only group (E-Nap). To the best of our knowledge, this is the first TMR study to show such an effect associated with REM for declarative memories. Our findings seemingly contradict the notion that REM sleep supports emotional memory consolidation. However, as reviewed recently8, evidence so far does not unequivocally support this postulation. A role for REM in eliminating memories was first proposed by Crick and Mitchison50. Later, different lines of research provided evidence suggesting that REM may be involved in the selective removal of memory representations51,52,53, although a direct link with forgetting in humans has been missing. While theta peak activity is associated with long-term potentiation in the hippocampus, several studies found that activity in the troughs causes depotentiation54,55. In one study, hippocampal place cells reversed their discharge phase to theta troughs in REM once the exposed environment became familiar, suggesting that REM may serve to “refresh” synapses for future use when encoding new memories56. Another study showed that firing rates of hippocampal neurons were decreased during REM, and the decrease was correlated with REM theta57. Furthermore, two recent studies showed direct evidence of synaptic pruning during REM after learning58,59. Finally, direct behavioral evidence for REM-related memory decay was found in a recent study60, which showed that a subset of melanin-concentrating-hormone-producing neurons were active specifically in REM and their activation was associated with impairment of memory. In light of this evidence, we can speculate that REM may have led to the weakening of synapses that represented emotional memories in our study.
Memory transformation might also have contributed to the association of REM with memory impairment (possibly with the contribution of mechanisms summarized above). REM is particularly conducive to forming new associations and might have facilitated the extraction of common elements across stimuli61, such as bodily harm, at the expense of information specific to individual stimuli (spatial location)52. Another possibility is that we are seeing the effects of an “emotional memory trade-off,” characterized by sleep selectively enhancing emotional aspects of memory (picture content) while memory for less salient elements (spatial location) weakens62. Finally, decline in memory might be related to “arousal”46,49, as TMR in REM increased alpha/beta activity. However, this increase was not correlated with changes in memory, and we observed a trend for REM to be associated with worse recall in a group with no reactivations, which makes it unlikely that results can be explained entirely by alpha/beta increase. Furthermore, additional mechanisms, such as activation in regions involved in emotional processing63, may explain the cue-evoked activity we observed in the REM group.
The strong correlation of cueing benefit with SWS × REM product in the E-SWS group suggests a complementary role for these sleep stages. This finding is in agreement with accounts that propose that both sleep stages are required for sleep-dependent memory consolidation64,65,66,67 and the expanding literature that provides supporting evidence in humans. Studies using a visual discrimination task found that improvement was correlated with the product of times spent in SWS and REM39, largest after a full night of sleep compared to early or late night half sleep68, and present after a nap only if it contained both NREM and REM40. In studies that used verbal memory tasks, recall was positively correlated with the duration of NREM/REM cycles69 and was impaired when sleep cycles were interrupted, but not after sleep fragmentation with intact sleep cycles70. Finally, similar to our study, several TMR studies that carried out reactivations in SWS found that REM or both REM and SWS were associated with outcomes41,71,72,73,74. In summary, there is now a substantial body of evidence that suggests that memory consolidation is attained via processes that take place across NREM and REM sleep. Further studies are needed to examine the dynamics of this coordination, such as how brain oscillations interact across sleep stages.
Direct cueing in REM sleep impaired memory, but SWS × REM time enhanced SWS cueing may at first appear contradictory. Work in rodents and humans suggests that SWS cueing promotes a direct reinstatement of the cued memory, leading to memory strengthening75 (see Denis & Cairney 202376 for a review). Another (though not necessarily mutually exclusive) possibility is that SWS reactivation promotes a transient destabilization of memory, allowing for the integration of new memories into existing knowledge networks. Subsequent REM-based consolidation processes then serve to strengthen the new, integrated memory 71,77. Such a process may be especially important for emotional memories, where it would be adaptive to integrate salience cues into existing knowledge networks to help us avoid similar threatening situations in the future. A related possibility is that the initial reactivation of emotional memories in SWS acted as a tagging mechanism for later REM strengthening. This is consistent with our finding that a larger spindle response following emotional SWS cues was found for more negatively valenced stimuli.
Whilst these accounts offer potential explanations for why SWS × REM time enhanced SWS cueing, when cues were presented in isolation during REM sleep, forgetting occurred. This could imply that cueing during REM sleep does not engage REM consolidation processes as directly as SWS cueing does. Interestingly, a recent study of REM cueing of a procedural memory task found that the ability to reinstate memory content during REM sleep was closely tied to the emergence of theta oscillations in the post-cue period 78. In the present study, we did not observe a post-cue increase in theta power, suggesting that our REM cueing procedure did not engage the necessary oscillatory mechanisms to facilitate REM-based memory strengthening. Instead, we saw cue-evoked increases in arousal-related alpha and beta bands, consistent with the idea that REM cueing may have been disruptive. However, we note that alpha and beta activity did not correlate with a lower cueing benefit at an individual level.
Cueing emotional stimuli during SWS generated EEG responses first in the delta/theta band and then at spindle frequencies. Notably, the valence of the sound cues was correlated with activity increase in both frequency ranges, suggesting that they contribute to emotional processing during sleep. There was also a trend for the delta/theta response to be positively correlated with cueing benefit, suggesting that delta-theta activity may have mediated the memory strengthening effect of emotional valence79. That spindle activity was modulated by emotional valence was supported by the additional finding that power in the spindle band was the only difference in cue-evoked activity between E-SWS and N-SWS groups, with significantly higher spindle power in E-SWS. A role for spindles in emotional processing has been suggested in recent studies that showed an association of spindle activity with enhanced consolidation of emotional memories21,23,36 and with a reduction in reactivity to negative memories after sleep80. In addition to correlations, another study20 found that increasing spindle activity pharmacologically significantly improved memory for negative and high-arousal items. Spindles have been found to be associated with psychiatric disorders81,82,83,84,85 and specific emotional symptoms86,87,88. Our results lend further support to the burgeoning evidence suggesting that spindles may be involved in emotional processing. The correlation of cue-evoked spindle activity with average valence ratings for the replayed stimulus -and specifically with valence but not arousal ratings- argues against the possibility that the difference in spindle power may be due to any systematic differences in sound characteristics between the stimulus sets.
Our study had several limitations. First, a neutral REM group, i.e. an additional group in which neutral stimuli were reactivated during REM, was not included due to constraints in time and resources. Including such a group would give us the opportunity to compare the memory benefit of reactivating neutral sounds in REM compared to emotional sounds and could thus be further informative in teasing out the role of REM in emotional memory consolidation. Second, we did not observe a memory benefit for the nap-only condition compared to wakefulness, and this may be related to the sleep characteristics of the E-Nap group. Several participants for which reactivations were planned ended up in this group because they did not achieve sufficient SWS or REM. This led to a “superficial” sleep in the E-Nap group compared to the reactivation groups (lower total sleep time, %REM, SWS×REM product, and higher wake after sleep onset, %N1, and %N2). This may also be the reason for the lack of correlation between the SWS×REM product and the change in memory in this group. Third, we found that emotional memories are retained better than neutral ones. However, given the lack of cue-evoked EEG activity in the N-SWS group, this difference might be simply due to neutral sounds not being sufficiently discernible or failure to establish strong sound-picture associations in this group. Finally, the primarily between-subjects analyses and relatively small sample size of each group may have meant we were underpowered to detect more subtle effects.
In sum, our results demonstrate important differences when reactivating emotional memories in either SWS or REM sleep. Whilst direct reactivation of memories in REM sleep induced forgetting, reactivation in SWS was dependent on the combination of SWS and REM sleep. These findings add further evidence to theories of SWS-REM cycling as being critical for memory consolidation, and the further unpacking of the interrelated role of SWS and REM sleep should be a priority for future research.
Methods
Participants
Healthy young participants were recruited from colleges in the greater Boston area. A total of 185 participants consented to the experiment, of which 123 (age: 21.6 ± 2.7; 63.9% female; see below for details) provided usable datasets for analysis. Participants reported no abnormal sleep patterns, history of psychiatric or neurological disorders, or current medication use except birth control. They were instructed to keep a regular sleep schedule for the three nights preceding the experiment and were asked to refrain from recreational drugs and alcohol for 48 h and caffeine in the morning before their visits. All participants provided written consent approved by the Institutional Review Board of Beth Israel Deaconess Medical Center. All ethical regulations relevant to human research participants were followed.
Participants were assigned to one of 5 groups. Emotional SWS (E-SWS) and emotional REM (E-REM) groups learned emotional items and were then exposed to reactivations during SWS or REM, respectively. Similarly, a neutral SWS (N-SWS) group learned neutral items with reactivations in SWS. We also included emotional nap (E-Nap) and emotional wake (E-Wake) groups, which learned emotional items and took a nap without any reactivations or did not take a nap but rested quietly for a similar period before testing. Although a neutral REM group was also planned, this was abandoned due to the time constraint imposed by an upcoming halt in laboratory operations. Groups were matched in age (F = .54, p = 0.71) and sex (X2 = 2.4, p = 0.66).
Design and procedures
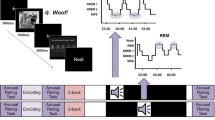
The protocol included two visits (Fig. 5A). On the first visit, participants arrived at the laboratory at around 11:00 AM. After consenting, they filled out the Epworth Sleepiness Scale (Johns, 1991) and questionnaires about their sleep patterns and quality for the preceding three nights. They were then wired for EEG (detailed below), and a brief (7-minute) resting state EEG was recorded with eyes closed. We do not report on the EEG data from the rest periods in this paper.
A Timeline of the study procedures for different groups are depicted. In all groups, the first visit started at 11:00 AM. After training and practice, a baseline test (T1) was carried out immediately before the nap. The first retest (T2) was approximately 45 min after waking up. The second retest (T3) was approximately 1 week after the first visit. B During training, participants passively viewed emotional or neutral pictures appearing at different locations on a grid, while a paired sound was played. During practice and test, pictures appeared in the center of the screen, and participants were asked to move them to their correct location.
We used a modified version of the TMR task described in previous publications (Rudoy et al., 2009; Creery et al., 2015). Learning included two successive phases: training and practice. During training, participants viewed 50 neutral or negative pictures appearing in different locations on a grid, in random order, while simultaneously hearing a 1-second sound that was naturally linked with that object (Fig. 5B, left). During practice, pictures appeared in the center of the screen while their corresponding sound played (Fig. 5B, right). Participants were instructed to move the objects to their original location and press the mouse button. After the mouse was clicked, the picture was moved to its correct location, providing feedback. In the first two runs of the practice phase, all 50 objects were tested. In subsequent rounds, items that had been placed within 150 pixels of their correct locations in two successive rounds no longer appeared. The practice phase continued until all objects had been removed from the testing pool. The baseline test (T1) followed immediately after learning. This phase was similar to practice, except participants placed each picture only once, and no feedback was provided. Following Test 1, a second 7-min resting state EEG was collected.
For participants in the sleep groups, lights were turned off shortly after the baseline test, usually around 2 PM, and a 2-h nap opportunity was provided. For all sleep participants, white noise was played through bedside speakers, starting with lights off and continuing until lights on. In the reactivation groups, half of the sounds (n = 25) were presented in a random order, with 5-second interstimulus intervals, with sound intensity of ~39 dB at the pillow. Cue presentations were started manually ~1.5 min after SWS or REM onset. Cues were selected by a computer algorithm so that memory accuracy at T1 for the cued and non-cued items was similar. Sounds were presented until the specific sleep stage (SWS or REM) ended. If the participant entered the target sleep stage again, reactivation was resumed. Data from a participant were included in analyses only if all 25 cues were played at least once. After lights on, a final resting state EEG was collected. Wake participants were also wired for EEG and spent an equal amount of time in the bedroom, doing relaxing activities such as reading, while being observed to ensure continued wakefulness.
Retests were identical to T1. The immediate retest (T2) took place approximately 45 min after the lights on or at the end of the rest period for the wake group. Before they left the laboratory, participants were instructed that their memory would be tested in the same way at their second visit. The second visit was approximately one week later and included only the delayed retest (T3), which took place at 4:45 PM, to match the timing of Test 2. EEG was not monitored at this visit.
Before training and each retest, participants filled out the Stanford Sleepiness Scale89 (Supplementary Table 1) and a two-item questionnaire about their ability to concentrate and their level of “feeling refreshed.”
We focused on results from T2, as the majority of the TMR studies indicated memory benefits evident shortly after sleep. Assessment at T3 was included as exploratory, given that some studies showed that TMR benefits might be longer lasting90.
Stimuli
50 emotional and 50 neutral images from online picture databases, the International Affective Pictures System91 and, the Geneva Affective Picture Database92, and Google image searches were used. Images were 150 × 150 pixels and were displayed on a 67.5 cm × 57.25 cm monitor with a viewing resolution of 1440 × 900 pixels. Emotional images were all negative. 1-second sounds that were naturally linked to each of these images were then taken from the database Pond5 (www.pond5.com) and paired with each image.
Prior to the main study, a pilot study was carried out to confirm that the emotional and neutral stimulus sets were significantly different in emotion ratings. This study confirmed that both emotional sounds and emotional sound-picture pairs were significantly more negative than their neutral counterparts (see Supplementary Materials).
EEG acquisition and preprocessing
EEG data was acquired from 57 channels (positioned according to the 10–20 system). Additional electrodes were placed on the left and right mastoids (for EEG references), above the right eye and below the left eye (for EOG), and on the chin (two; for EMG), forehead (recording reference), and collarbone (ground). Data were collected at 400 Hz with an Aura-LTM64 amplifier and TWin software (Grass Technologies). All impedances were kept to <25 kOhm.
Sleep scoring was performed using TWin software and MATLAB (The MathWorks, Natick, MA) according to AASM criteria93. Subsequent EEG analyses were performed in MATLAB using custom scripts. First, all EEG channels were re-referenced to the average of the two mastoids, high-pass filtered at 0.3 Hz and notch filtered at 60 Hz. Data were then artifact-rejected based on visual inspection, with bad segments of data being marked and removed from subsequent analyses. Bad channels were identified by visual inspection and interpolated using a spherical spline algorithm. Artifact-free data were then subjected to further analysis.
EEG data analysis
Filtered, artifact-free data were segmented into epochs that extended from 1 s before stimulus-onset to 3 s after and baseline adjusted to the mean voltage during the 1 second before cue onset. Complex Morlet wavelets were used to decompose the epoched and baseline-adjusted time series data into time-frequency representations94, with spectral power being extracted at 30 logarithmically spaced frequencies from 2 to 40 Hz and the number of wavelet cycles increasing from 3 to 10 in 30 logarithmically spaced steps to match frequency bins. For analysis, power was decibel-normalized within-subject (10× log10(power/baseline)), where the baseline was mean power in the 200–500 ms prior to cue-onset. This baseline period was chosen to mitigate contamination of the baseline period by post-stimulus activity. As such, positive values reflect relative increases in power following sound cues compared to the pre-cue baseline, whereas negative values reflect decreases. To reduce EEG data complexity, we focused time-frequency analyses on electrode Cz as this site has previously shown to be sensitive to TMR-related effects95,96.
Data reduction
A participant’s data was excluded if (i) less than one full round of reactivation was achieved (n = 32; 17%), (ii) a full round of reactivation was carried out in the wrong sleep stage (n = 4; 2%), (iii) sleep was too short or fragmented (sleep <45 min or WASO > 30 min; n = 18; 10%), (iv) an equipment failure occurred (n = 5; 3%), (v) the protocol was not completed (n = 2; 1%), or healthy control criteria were not met (n = 1; <1%). In addition, 1 participant from the E-REM group was removed because their error at T1, as well as at T2, was more than three standard deviations from the mean. The final sample included 24 participants in E-SWS, 25 in E-REM, 31 in N-SWS, 22 in E-Nap, and 20 in E-Wake. Finally, the EEG of 1 participant in N-SWS could not be used in spectral analyses due to a recording error.
Statistics and reproducibility
Recall accuracy was measured as the distance between where participants placed the pictures and their correct locations, measured in pixels, with smaller errors in placement indicating better recall accuracy. Because error was not normally distributed within subjects, we used subject medians for each test in all analyses. The effect of reactivation on recall accuracy was examined using LMMs. In these models, the % change in error from T1 (%ΔError) was included as the dependent variable. Larger %ΔError values indicated more forgetting. ‘Reactivation’ (reactivated vs. non-reactivated) was included as a fixed effect. In the analyses that compared the effect of reactivation between groups, ‘Group’ and ‘Group × Reactivation’ interaction were included as additional fixed effects. Our primary interest was in the two-way interaction between ‘Group’ and ‘Reactivation’; however, we used full-factorial models, which also included the three-way interaction (‘Reactivation × Group × SWS×REM product’) for completeness. In all models, compound symmetry covariance structure was used for the repeated measures, with ‘Subject’ included as a random effect.
A one-way MANOVA was used to compare the sleep variables (total sleep time [TST], wake after sleep onset [WASO], %N1, %N2, %SWS, %REM, and SWS×REM product) between the reactivation groups, with Tukey’s test for post-hoc analyses. Pearson’s correlation was used for correlations between normally distributed variables. Spearman’s correlation was used if variables were not normally distributed.
To analyze cue-evoked EEG data, we first identified clusters of time-frequency points for which post-cue activity was significantly different from zero. For each group, we conducted one-sample t tests across participants to detect points in the time-frequency space at which spectral power was systematically different from zero (p < 0.05), false discovery rate (FDR) adjusted; see ref. 44 for a similar approach. Clusters of significant activity were identified using the bwlabeln function in MATLAB94. Within each cluster, spectral power was averaged across all time-frequency points in that cluster, producing for each participant a single spectral power value for each cluster.
All hypothesis testing was carried out using two-sided tests. The significance threshold was set at p < 0.05. For correlations, Bonferroni correction was used to control family wise error rate.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Demographic, behavioral, and EEG time-frequency data can be found at: https://osf.io/myhcf/?view_only=635c9a9ad20b4ff5952f24faa2a7258f.
Code availability
Codes necessary to reproduce the results can be found at: https://osf.io/myhcf/?view_only=635c9a9ad20b4ff5952f24faa2a7258f.
References
Stickgold, R. Sleep-dependent memory consolidation. Nature 437, 1272–1278 (2005).
Klinzing, J. G., Niethard, N. & Born, J. Mechanisms of systems memory consolidation during sleep. Nat. Neurosci. 22, 1598–1610 (2019).
Tononi, G. & Cirelli, C. Sleep and the price of plasticity: From synaptic and cellular homeostasis to memory consolidation and integration. Neuron 81, 12–34 (2014).
Boyce, R., Williams, S. & Adamantidis, A. REM sleep and memory. Curr. Opin. Neurobiol. 44, 167–177 (2017).
Giuditta, A. Sleep memory processing: the sequential hypothesis. Front Syst. Neurosci. 8, 219 (2014).
Singh, D., Norman, K. A. & Schapiro, A. C. A model of autonomous interactions between hippocampus and neocortex driving sleep-dependent memory consolidation. Proc. Natl. Acad. Sci. USA 119, e2123432119 (2022).
Schmidt, K., Patnaik, P. & Kensinger, E. A. Emotion’s influence on memory for spatial and temporal context. Cogn. Emot. 25, 229–243 (2011).
Davidson, P., Jonsson, P., Carlsson, I. & Pace-Schott, E. Does sleep selectively strengthen certain memories over others based on emotion and perceived future relevance? Nat. Sci. Sleep. 13, 1257–1306 (2021).
Genzel, L., Spoormaker, V. I., Konrad, B. N. & Dresler, M. The role of rapid eye movement sleep for amygdala-related memory processing. Neurobiol. Learn Mem. 122, 110–121 (2015).
Wagner, U., Gais, S. & Born, J. Emotional memory formation is enhanced across sleep intervals with high amounts of rapid eye movement sleep. Learn Mem. 8, 112–119 (2001).
Groch, S., Wilhelm, I., Diekelmann, S. & Born, J. The role of REM sleep in the processing of emotional memories: evidence from behavior and event-related potentials. Neurobiol. Learn Mem. 99, 1–9 (2013).
Sopp, M. R., Michael, T., Weess, H. G. & Mecklinger, A. Remembering specific features of emotional events across time: The role of REM sleep and prefrontal theta oscillations. Cogn. Affect Behav. Neurosci. 17, 1186–1209 (2017).
Groch, S., Zinke, K., Wilhelm, I. & Born, J. Dissociating the contributions of slow-wave sleep and rapid eye movement sleep to emotional item and source memory. Neurobiol. Learn Mem. 122, 122–130 (2015).
Wiesner, C. D. et al. The effect of selective REM-sleep deprivation on the consolidation and affective evaluation of emotional memories. Neurobiol. Learn Mem. 122, 131–141 (2015).
Nishida, M., Pearsall, J., Buckner, R. L. & Walker, M. P. REM sleep, prefrontal theta, and the consolidation of human emotional memory. Cereb. Cortex 19, 1158–1166 (2009).
Payne, J. D., Chambers, A. M. & Kensinger, E. A. Sleep promotes lasting changes in selective memory for emotional scenes. Front Integr. Neurosci. 6, 108 (2012).
Gilson M., et al. REM-enriched naps are associated with memory consolidation for sad stories and enhance mood-related reactivity. Brain Sci. 6, (2015).
Harrington, M. O., Nedberge, K. M. & Durrant, S. J. The effect of sleep deprivation on emotional memory consolidation in participants reporting depressive symptoms. Neurobiol. Learn Mem. 152, 10–19 (2018).
Goldstein, A. N. & Walker, M. P. The role of sleep in emotional brain function. Annu Rev. Clin. Psychol. 10, 679–708 (2014).
Kaestner, E. J., Wixted, J. T. & Mednick, S. C. Pharmacologically increasing sleep spindles enhances recognition for negative and high-arousal memories. J. Cogn. Neurosci. 25, 1597–1610 (2013).
Cairney, S. A., Durrant, S. J., Jackson, R. & Lewis, P. A. Sleep spindles provide indirect support to the consolidation of emotional encoding contexts. Neuropsychologia 63, 285–292 (2014).
Ackermann, S., Hartmann, F., Papassotiropoulos, A., de Quervain, D. J. & Rasch, B. No Associations between Interindividual Differences in Sleep Parameters and Episodic Memory Consolidation. Sleep 38, 951–959 (2015).
Alger, S. E., Kensinger, E. A. & Payne, J. D. Preferential consolidation of emotionally salient information during a nap is preserved in middle age. Neurobiol. Aging 68, 34–47 (2018).
Baran, B., Pace-Schott, E. F., Ericson, C. & Spencer, R. M. Processing of emotional reactivity and emotional memory over sleep. J. Neurosci. 32, 1035–1042 (2012).
Sopp, M. R., Michael, T. & Mecklinger, A. Effects of early morning nap sleep on associative memory for neutral and emotional stimuli. Brain Res. 1698, 29–42 (2018).
Hutchison, I. C. et al. Targeted memory reactivation in REM but not SWS selectively reduces arousal responses. Commun. Biol. 4, 404 (2021).
Morgenthaler, J. et al. Selective REM-sleep deprivation does not diminish emotional memory consolidation in young healthy subjects. PLoS One 9, e89849 (2014).
Cellini, N., Torre, J., Stegagno, L. & Sarlo, M. Sleep before and after learning promotes the consolidation of both neutral and emotional information regardless of REM presence. Neurobiol. Learn Mem. 133, 136–144 (2016).
Cairney, S. A., Durrant, S. J., Power, R. & Lewis, P. A. Complementary roles of slow-wave sleep and rapid eye movement sleep in emotional memory consolidation. Cereb. Cortex 25, 1565–1575 (2015).
Payne, J. D. et al. Napping and the selective consolidation of negative aspects of scenes. Emotion 15, 176–186 (2015).
Wagner, U., Kashyap, N., Diekelmann, S. & Born, J. The impact of post-learning sleep vs. wakefulness on recognition memory for faces with different facial expressions. Neurobiol. Learn Mem. 87, 679–687 (2007).
Benedict, C., Scheller, J., Rose-John, S., Born, J. & Marshall, L. Enhancing influence of intranasal interleukin-6 on slow-wave activity and memory consolidation during sleep. FASEB J. 23, 3629–3636 (2009).
Hu, X., Cheng, L. Y., Chiu, M. H. & Paller, K. A. Promoting memory consolidation during sleep: a meta-analysis of targeted memory reactivation. Psychol. Bull. 146, 218–244 (2020).
Carbone, J. & Diekelmann, S. An update on recent advances in targeted memory reactivation during sleep. NPJ Sci. Learn 9, 31 (2024).
Cairney, S. A., Durrant, S. J., Hulleman, J. & Lewis, P. A. Targeted memory reactivation during slow wave sleep facilitates emotional memory consolidation. Sleep 37, 701–707 (2014).
Lehmann, M., Schreiner, T., Seifritz, E. & Rasch, B. Emotional arousal modulates oscillatory correlates of targeted memory reactivation during NREM, but not REM sleep. Sci. Rep. 6, 39229 (2016).
Ashton, J. E., Cairney, S. A. & Gaskell, M. G. No effect of targeted memory reactivation during slow-wave sleep on emotional recognition memory. J. Sleep. Res. 27, 129–137 (2018).
Creery, J. D., Oudiette, D., Antony, J. W. & Paller, K. A. Targeted memory reactivation during sleep depends on prior learning. Sleep 38, 755–763 (2015).
Stickgold, R., Whidbee, D., Schirmer, B., Patel, V. & Hobson, J. A. Visual discrimination task improvement: A multi-step process occurring during sleep. J. Cogn. Neurosci. 12, 246–254 (2000).
Mednick, S., Nakayama, K. & Stickgold, R. Sleep-dependent learning: a nap is as good as a night. Nat. Neurosci. 6, 697–698 (2003).
Hu, X. et al. Cognitive neuroscience. Unlearning implicit social biases during sleep. Science 348, 1013–1015 (2015).
Kim, E. J. & Dimsdale, J. E. The effect of psychosocial stress on sleep: a review of polysomnographic evidence. Behav. Sleep. Med. 5, 256–278 (2007).
Suchecki, D., Tiba, P. A. & Machado, R. B. REM Sleep Rebound as an Adaptive Response to Stressful Situations. Front Neurol. 3, 41 (2012).
Schechtman, E. et al. Multiple memories can be simultaneously reactivated during sleep as effectively as a single memory. Commun. Biol. 4, 25 (2021).
Forcato, C. et al. Reactivation during sleep with incomplete reminder cues rather than complete ones stabilizes long-term memory in humans. Commun. Biol. 3, 733 (2020).
Goldi, M. & Rasch, B. Effects of targeted memory reactivation during sleep at home depend on sleep disturbances and habituation. NPJ Sci. Learn 4, 5 (2019).
Berry, R. B. et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J. Clin. Sleep. Med. 8, 597–619 (2012).
Haba-Rubio, J., Ibanez, V. & Sforza, E. An alternative measure of sleep fragmentation in clinical practice: the sleep fragmentation index. Sleep. Med. 5, 577–581 (2004).
Whitmore, N. W., Bassard, A. M. & Paller, K. A. Targeted memory reactivation of face-name learning depends on ample and undisturbed slow-wave sleep. NPJ Sci. Learn 7, 1 (2022).
Crick, F. & Mitchison, G. The function of dream sleep. Nature 304, 111–114 (1983).
Poe, G. R. Sleep is for forgetting. J. Neurosci. 37, 464–473 (2017).
Langille, J. J. Remembering to forget: A dual role for sleep oscillations in memory consolidation and forgetting. Front Cell Neurosci. 13, 71 (2019).
Feld, G. B. & Born, J. Sculpting memory during sleep: Concurrent consolidation and forgetting. Curr. Opin. Neurobiol. 44, 20–27 (2017).
Huerta, P. T. & Lisman, J. E. Bidirectional synaptic plasticity induced by a single burst during cholinergic theta oscillation in CA1 in vitro. Neuron 15, 1053–1063 (1995).
Holscher, C., Anwyl, R. & Rowan, M. J. Stimulation on the positive phase of hippocampal theta rhythm induces long-term potentiation that can Be depotentiated by stimulation on the negative phase in area CA1 in vivo. J. Neurosci. 17, 6470–6477 (1997).
Poe, G. R., Nitz, D. A., McNaughton, B. L. & Barnes, C. A. Experience-dependent phase-reversal of hippocampal neuron firing during REM sleep. Brain Res. 855, 176–180 (2000).
Grosmark, A. D., Mizuseki, K., Pastalkova, E., Diba, K. & Buzsaki, G. REM sleep reorganizes hippocampal excitability. Neuron 75, 1001–1007 (2012).
Li, W., Ma, L., Yang, G. & Gan, W. B. REM sleep selectively prunes and maintains new synapses in development and learning. Nat. Neurosci. 20, 427–437 (2017).
Zhou, Y. et al. REM sleep promotes experience-dependent dendritic spine elimination in the mouse cortex. Nat. Commun. 11, 4819 (2020).
Izawa, S. et al. REM sleep-active MCH neurons are involved in forgetting hippocampus-dependent memories. Science 365, 1308–1313 (2019).
Lewis, P. A., Knoblich, G. & Poe, G. How Memory Replay in Sleep Boosts Creative Problem-Solving. Trends Cogn. Sci. 22, 491–503 (2018).
Denis, D., Sanders, K. E. G., Kensinger, E. A. & Payne, J. D. Sleep preferentially consolidates negative aspects of human memory: Well-powered evidence from two large online experiments. Proc. Natl. Acad. Sci. USA 119, e2202657119 (2022).
Vijayan S., Lepage K. Q., Kopell N. J., Cash S. S. Frontal beta-theta network during REM sleep. Elife 6, (2017).
Walker, M. P. & Stickgold, R. Overnight alchemy: sleep-dependent memory evolution. Nat. Rev. Neurosci. 11, 218 (2010). author reply 218.
Diekelmann, S. & Born, J. The memory function of sleep. Nat. Rev. Neurosci. 11, 114–126 (2010).
Giuditta, A. et al. The sequential hypothesis of the function of sleep. Behav. Brain Res. 69, 157–166 (1995).
Ficca, G. Salzarulo P. What in sleep is for memory. Sleep. Med 5, 225–230 (2004).
Gais, S., Plihal, W., Wagner, U. & Born, J. Early sleep triggers memory for early visual discrimination skills. Nat. Neurosci. 3, 1335–1339 (2000).
Mazzoni, G. et al. Word recall correlates with sleep cycles in elderly subjects. J. Sleep. Res. 8, 185–188 (1999).
Ficca, G., Lombardo, P., Rossi, L. & Salzarulo, P. Morning recall of verbal material depends on prior sleep organization. Behav. Brain Res 112, 159–163 (2000).
Batterink, L. J., Westerberg, C. E. & Paller, K. A. Vocabulary learning benefits from REM after slow-wave sleep. Neurobiol. Learn Mem. 144, 102–113 (2017).
Tamminen, J., Lambon Ralph, M. A. & Lewis, P. A. Targeted memory reactivation of newly learned words during sleep triggers REM-mediated integration of new memories and existing knowledge. Neurobiol. Learn Mem. 137, 77–82 (2017).
Goldi, M., van Poppel, E. A. M., Rasch, B. & Schreiner, T. Increased neuronal signatures of targeted memory reactivation during slow-wave up states. Sci. Rep. 9, 2715 (2019).
Wang, J. Y., Heck, K. L., Born, J., Ngo, H. V. & Diekelmann, S. No difference between slow oscillation up- and down-state cueing for memory consolidation during sleep. J. Sleep. Res. 31, e13562 (2022).
Bendor, D. & Wilson, M. A. Biasing the content of hippocampal replay during sleep. Nat Neurosci. 15, 1439–1444 (2012).
Denis, D. & Cairney, S. A. Neural reactivation during human sleep. Emerg. Top Life Sci. 7, 487–498 (2023).
Rasch, B. & Born, J. Maintaining memories by reactivation. Curr. Opin. Neurobiol. 17, 698–703 (2007)
Abdellahi, M. E. A., Koopman, A. C. M., Treder, M. S. & Lewis, P. A. Targeted memory reactivation in human REM sleep elicits detectable reactivation. Elife 12 https://doi.org/10.7554/eLife.84324 (2023).
Hayes A. F. Introduction to mediation, moderation, and conditional process analysis: A regression-based approach, Second edn. Guilford (2018).
Azza, Y. et al. Sleep’s role in updating aversive autobiographical memories. Transl. Psychiatry 12, 117 (2022).
Lopez, J., Hoffmann, R. & Armitage, R. Reduced sleep spindle activity in early-onset and elevated risk for depression. J. Am. Acad. Child Adolesc. Psychiatry 49, 934–943 (2010).
Sesso, G. et al. Electrophysiological and microstructural features of sleep in children at high risk for depression: a preliminary study. Sleep. Med. 36, 95–103 (2017).
Nishida, M., Nakashima, Y. & Nishikawa, T. Topographical distribution of fast and slow sleep spindles in medicated depressive patients. J. Clin. Neurophysiol. 31, 402–408 (2014).
Plante, D. T. et al. Topographic and sex-related differences in sleep spindles in major depressive disorder: a high-density EEG investigation. J. Affect Disord. 146, 120–125 (2013).
Wilhelm, I., Groch, S., Preiss, A., Walitza, S. & Huber, R. Widespread reduction in sleep spindle activity in socially anxious children and adolescents. J. Psychiatr. Res. 88, 47–55 (2017).
Hamann, C., Rusterholz, T., Studer, M., Kaess, M. & Tarokh, L. Association between depressive symptoms and sleep neurophysiology in early adolescence. J. Child Psychol. Psychiatry 60, 1334–1342 (2019).
Kathrin, B., Michael, A. H., Ines, W. & Kerstin, H. The relation between sigma power and internalizing problems across development. J. Psychiatr. Res. 135, 302–310 (2021).
van der Heijden A. C. et al. Sleep spindle dynamics suggest over-consolidation in post-traumatic stress disorder. Sleep 45, (2022).
Hoddes, E., Dement, W. & Zarcone, V. The development and use of the Stanford Sleepiness Scale (SSS). Psychophysiology 9, 150 (1972).
Cairney, S. A., Guttesen, A. A. V., El Marj, N. & Staresina, B. P. Memory Consolidation Is Linked to Spindle-Mediated Information Processing during Sleep. Curr. Biol. 28, 948–954.e944 (2018).
Lang, P. J., Bradley, M. M. & Cuthbert, B. N. International Affective Picture System (IAPS): Instruction manual and affective ratings, Technical Report A-8., (2008).
Dan-Glauser, E. S. & Scherer, K. R. The Geneva affective picture database (GAPED): a new 730-picture database focusing on valence and normative significance. Behav. Res Methods 43, 468–477 (2011).
Berry Q. S., et al.; for the American Academy of Sleep Medicine. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications, Version 2.6. (2020).
Cohen X. Analyzing Neural Time Series Data Theory and Practice. Massachusetts Institute of Technology (MIT) Press. (2014).
Denis D., Payne J. D. Targeted memory reactivation during nonrapid eye movement sleep enhances neutral, but not negative, components of memory. eNeuro 11, (2024).
Schechtman, E., Heilberg, J. & Paller, K. A. Memory consolidation during sleep involves context reinstatement in humans. Cell Rep. 42, 112331 (2023).
Acknowledgements
This work was funded by NIH grant MH048832 to Dr. Stickgold.
Author information
Authors and Affiliations
Contributions
C.Y and R.S conceptualized the study. C.Y, A.O, J.C, E.S collected data. C.Y, D.D, R.C, A.M and R.S analyzed the data. B.R provided guidance on the statistical approach. C.Y, D.D, R.C, and R.S wrote the manuscript draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: Christina Karlsson Rosenthal. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yuksel, C., Denis, D., Coleman, J. et al. Both slow wave and rapid eye movement sleep contribute to emotional memory consolidation. Commun Biol 8, 485 (2025). https://doi.org/10.1038/s42003-025-07868-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-025-07868-5
This article is cited by
-
Effect of horse sleep behavior on performance in a field-side spatial reversal learning test
Scientific Reports (2026)