Abstract
Many animals and humans use scream calls to signal imminent threats, which typically evoke alarm and escape responses in recipients. Compared to animals, human scream calls are more diversified with varying levels of alarm signaling. Certain low-alarm human screams thus might not elicit full neural alarm cascades and threat avoidance actions. Here we investigated the neural circuits for decoding alarm and avoid-approach information from positive and negative scream calls in humans. Alarm and avoid-approach decisions showed minimal neural overlap, with alarm decisions recruiting neural cascades of sensory-affective decoding for action preparation, whereas avoid-approach judgments recruited neural systems for spatio-affective decoding for social decision-making. Furthermore, decision patterns revealed both an alarm quotient and an avoid quotient for risk arbitration, which linked the likelihood of an urgent response (alarm, avoid) to a slowing of choosing a potential safe option in response to scream calls. While the avoid quotient positively predicted neural activity in a broad cortico-limbic network, the alarm quotient predicted neural suppression with increasing alarm levels, especially in the amygdala as part of a presumed limbic alarm system. This points to a critical involvement of the amygdala at neural levels of choice arbitrations rather than in threat evaluations signaled by screams.
Similar content being viewed by others
Introduction
Alarm signaling is critical for the survival of individual organisms and conspecifics. Many vertebrate species have developed various visual and auditory alarm signals to indicate danger to in-group members and to scare off predators. Among these various alarm signals, vocal signals are one of the most important signals of threat, especially in long-range communication with limited visual contact. The most alarming and intense vocal signals of threat and danger are scream calls1,2, and scream calls are part of the vocal repertoire of many mammalian and vertebrate species. Scream calls are alarm signals for environmental predatory threats or for in-group agonistic and aggressive encounters. Screams are short, high-pitched, and intense calls with a rough acoustic quality1,2. Registering scream calls in listeners often induces startling responses3 and further triggers immediate and urgent response tendencies for adaptive behavior4. The detection and recognition of screams as alarm signals, which critically depends on recognizing the alarm quality and alarm distinctiveness of the scream, thus seem vital to many species including humans.
Accurately registering the alarm signal in screams shapes adaptive response preparation and overt actions in listeners, which often serve as distal proxies that signify the actual alarm quality of the scream produced by the caller. Although the concept of screams as alarm signals thus seems well established in biological5 and psychological literature1,2, it seems less well defined in terms of the neurocognitive process that leads to the perceptual decision of whether a scream call is alarming or not. Furthermore, although scream calls typically seem to elicit certain levels of urgency and alarm response, the level of this response seems to depend on the specific scream type, especially in humans1,6.
This scream-type dependency of the alarm judgment is specifically relevant in humans with a potentially broader scream call repertoire than other species (i.e., all non-human animals, which we refer to as “animals” throughout the manuscript). Humans, like animals, scream in states of fear and aggression, but humans also seem to scream in low-arousing negative affective states, such as sadness1,7, and specifically in positive affective states, such as elated joy and intense pleasure1. Human scream calls thus might show increased variability in terms of the scream contexts compared to animals, especially regarding screams in socially more complex contexts1. For the latter types of screams, consistent evidence from animal scream signaling and scream perception is so far largely missing. Human listeners can distinguish these different scream types based on acoustic variations and discriminate alarming (pain, fear, anger) from non-alarming screams (joy, pleasure, sadness). Again, this alarm/non-alarm distinction of human screams has been quantified based on other overt responses as a proxy to the alarm dimension. Thus, it still needs to be scientifically established that the alarm dimension is mechanistically relevant for the perceptual processing of a scream and what neurocognitive process leads to the decision of whether a screaming call is alarming or not. It also needs to be established how the judgment of the alarming quality of a scream is potentially relevant for an adaptive response choice.
Regarding the appropriate response choice to scream calls, a typical response to screams is to avoid the source of the threat. This can lead to fleeing behavior in case of fear8, but also to freezing or submissive/defensive behavior when confronted with an aggressive screamer9,10,11. This issue is again further complicated for humans, where scream calls not only signal danger but also positive affective states. Instead of avoidance responses, such scream calls could lead to approach behavior in listeners. This approach tendency could additionally be the case for some negative scream types (sadness, pain) that are typically expressed to attract allies for support. Thus, although the acoustic quality and affective scream information might evoke alarm response tendencies, they could motivate to approach rather than avoid the scream caller or scream context12,13,14.
The avoid-approach judgments and decisions are an established behaviorally relevant dimension related to the affective quality of stimuli in general and are thus not exclusive to screams. Some stimuli can even lead to an avoid-approach conflict15, given that such stimuli have a rewarding as well as an aversive and risky quality. Such a conflict might also be relevant for certain scream calls specifically as they could trigger both approach (help a person in pain) and avoidance tendencies (pain scream as an indicator of potential environmental threat or danger). There is an ambiguity of what the scream represents with regard to reward versus risk, requiring listeners to extract information from salient affective cues in the scream signal to form a decision.
Scream calls thus seem to imply two perceptually and behaviorally important dimensions, and listeners need to accurately decide between two options regarding each dimension. These dimensions concern two very important types of decoding and evaluation of scream signals, and there might be essential commonalities and differences in these assessment types. Both dimensions and assessments are similar in that one option represents a choice for urgency (alarm, avoid) while the other option can be regarded as the safe option (non-alarm, approach). The safe options, however, comprise certain risks in case of false decisions, especially when wrongly choosing the safe option in case of a true environmental threat. The alarm/non-alarm decision and the approach-avoidance decision thus seem to overlap considerably in their processing demands, and they might be assumed to be mutually dependent (e.g., alarm detection in screams might be directly linked to avoid tendencies). Similar neural mechanisms for alarm judgments and avoid-approach decisions also point to a neural overlap, such as in the amygdala as part of the limbic system for affective analysis2,16,17 and the sensory auditory cortex for acoustic signal analysis1,2,18.
Although there seems to be neural overlap in the processing of the alarm and approach/avoid (AA) dimension from the alarm signal, the neural codes for both dimensions partly show differential and opposite patterns in terms of either brain signal enhancement or suppression1,2,17. This differential neural code might even be evident regarding the larger brain network involved in the evaluative dynamics regarding alarm and AA judgments. The alarm judgment has a stronger affective evaluation component, which is predominantly supported by the limbic-cerebellar-brainstem network1,19,20 and specific amygdala-prefrontal association21. The AA judgment has a stronger focus on adaptive behavioral response evaluations, which is supported by striatal and insula regions22, as well as the specific role of the hippocampus23,24 and the hippocampus-prefrontal association25,26 for the optimal arbitration of response options27. These findings altogether point to both common and differential evaluative and decisional components in making alarm and AA judgments during the decoding of scream calls. Both judgments seem relevant and essential when animals and humans perceive scream calls, especially in humans given their diversity of scream call repertoire. A limitation from previous studies that we addressed in the present study was that the neural dynamics of alarm and AA judgment had been investigated in separate studies, which allows only indirect inferences about the communities and differences for types of judgments when confronted with threat scans. Furthermore, scream calls are very specific and intense alarm signals in communicative settings, and alarm and AA judgments might have crucial significance with this kind of alarm signaling. We therefore experimentally tested the common as well as differential neural dynamics that underlie alarm and AA judgments for different negative and positive human scream calls using functional neuroimaging and binary-choice experimental setups (Fig. 1a, b).
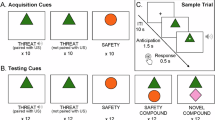
a Spectrograms of exemplary scream calls for the six different scream types and the “neutral” screams. b Two binary forced-choice tasks. For each scream call, participants had to decide if the scream call signaled “alarm” or “non-alarm” (ALARM task, left panel) or if they would “avoid” or “approach” the person who produced the scream (AA task, right panel). c Reaction times (RT) and choice probabilities for the two tasks (all n = 35). * Asterisk indicates a significant difference (p < 0.05, FWER corrected) between the urgent (alarm, avoid) and safe option (non-alarm, approach) for each scream type (left/middle panel) or the difference between the probabilities for the urgent decision (alarm versus avoid) across both tasks. Small points are participant data; dark points correspond to solid line, gray points correspond to dashed line (left/mid panel); colored points correspond to line color (right panel). Plots represent mean ± SEM d Correlation coefficients per participant (n = 35) between the probability pattern for alarm decision (DEC ala) across scream types and the RT for alarm (RT ala) and non-alarm decision (RT non) in red, and for correlations between the probability pattern for avoid decisions (DEC avo) across scream types and the RT for avoid (RT avo) and approach decision (RT app) in blue. * Asterisk indicates significant correlation p < 0.05 FWER corrected. Boxplots are median, box is 0.25-0.75 percentile, whiskers or most extreme data points. e Distribution pattern (n = 35) between all alarm choice probabilities (DEC ala) and non-alarm RT latencies (RT non) (left panel) and all avoid choice probabilities (DEC avo) and approach RT latencies (RT app) (right panel).
Results
Scream type considerably influences alarm but less avoid-approach judgments
We recorded brain activity using functional magnetic resonance imaging (fMRI) from n = 35 human participants (21 females, 14 males; mean age 27.20 years, SD 6.22) while they listened to six different human scream calls (pleasure, joy, sadness, pain, fear, anger) including a seventh scream type of “neutral” screams as intense vocalizations of the vowel/a/ (Fig. 1a). Participants were asked to either perform an alarm decision task (ALARM task, “alarm” or “non-alarm” as binary response options) or an approach-avoidance decision task (AA task, “avoid” and “approach” as binary response options) in each of the scream calls. Participants performed two blocks for the ALARM task and two blocks for the AA task, and each block contained the same 84 scream calls (12 per scream type) that were used and validated in a previous study1.
Both the ALARM task and the AA task involve a safe (non-alarm, approach) and an urgent decision option (alarm, avoid). For the latter, a speeded decision seems necessary and potentially vital. We therefore quantified reaction times (RT) for the two decision options for each of the seven scream-type calls and analyzed RTs in a 2 × 7 repeated measure ANOVA (Fig. 1c). For the ALARM task, RTs were different for the different scream types (F6,204 = 7.006, p < 0.001; Greenhouse-Geiser (GG) corrected) and for the two decision options (F1,34 = 6.277, p = 0.023; GG corrected).
Given an interaction effect between scream type and the decision options (F6,204 = 11.606, p < 0.001; GG corrected), we performed various posthoc comparisons. The alarm decision was faster for joy, sad, pain, and fear screams compared to neutral screams (all p’s < 0.048, all FWER corrected), and the non-alarm decision was slower for each of the negative screams (sad, pain, fear, anger) compared to each of the neutral (p’s < 0.036), pleasure (p’s < 0.002), and joy screams (p’s < 0.011). While the non-alarm compared to the alarm decision was faster for neutral screams (p = 0.012), the non-alarm decision was slower compared to the alarm decision for all negative screams (p’s < 0.013).
Overall, this mainly points to faster alarm decisions and slower non-alarm decisions for negative screams. While quick responses for the urgency (alarm) option are plausible, the significant response slowing to the safe option is an interesting observation. Decoding for the safe option contains some levels of risk in case of false negative decisions (i.e., choosing the safe option in case of real threat), and this risk arbitration can lead to response slowing. Given this RT pattern for the alarm and non-alarm decision, we classified the negative screams as “high-alarm” scream calls and the neutral and positive screams as “low-alarm” scream calls.
Concerning the RT patterns for the AA task, RTs differed between scream types (F6,204 = 2.544, p = 0.035, GG corrected) and between scream types depending on decisions (F6,204 = 2.639, p = 0.048, GG corrected). According to posthoc planned comparisons, the avoid decision was faster for fear than sadness scream (p = 0.021), and a tendency for avoid decisions being faster for pleasure screams than approach decision (p = 0.052). Overall and unlike for the ALARM task, there was little evidence that the speed for decisions in the AA task was differentially influenced by the scream call type.
We finally tested for significant differences in the choice probability for the urgent decision across the ALARM and the AA task. We quantified the probability of choosing the alarm and the avoid option for each scream type. The probability was different across scream types (F6,204 = 31.507, p < 0.001) and for scream types depending on choice (F6,204 = 27.354, p < 0.001). Anger screams had higher probabilities of avoid decisions (p’s < 0.001) than joy, sad, pain, and fear screams, and pleasure screams received higher avoid decisions (p’s < 0.044) than neutral and joy screams. For the probabilities of the alarm decision, each of the negative screams received higher probabilities of alarm decision compared to each of the neutral and positive screams (p’s < 0.006).
Comparing alarm and avoid choices for each scream type, neutral and pleasure screams received higher probabilities of avoid than alarm decision (p’s < 0.001), while sad, pain, and fear screams received higher probabilities of alarm than avoid decision (p’s < 0.010).
Probabilities for urgent decisions are coupled with the latency of opposite safe decision
An interesting observation reported in the previous section was that the statistical pattern for the choice probabilities of the alarm decision across the seven scream call types (Fig. 1c, right panel, red line) was highly similar to the RT latency pattern for the non-alarm decision (Fig. 1c, right panel, dotted line). Thus, the decision probability pattern for an urgent decision (alarm) followed the response RT latency pattern for the opposite safe option (non-alarm). To obtain a quantitative measure of this pattern similarity, we correlated the alarm probability pattern with the non-alarm RT pattern. The same correlation analysis was also performed for the association of the avoid probability pattern with the approach RT pattern. For full consistency in the data analysis, we finally also performed all other correlations between decision probabilities and RT latencies.
Only the correlation between alarm decision probabilities and non-alarm RT latencies (r = 0.690, p < 0.001, FWER corrected) as well as the correlation between avoid decision probabilities and approach RT latencies were significant (r = 0.689, p = 0.005, FWER corrected) (Fig. 1d). This surprising finding indicates that the likelihood of making an urgent decision towards a certain scream call is positively associated with the time it takes to decide about the safe option for that type of scream call. To analyze this further, we performed a correlation analysis between any urgent alarm or avoid decision independent of the screaming type and the RT latency for the opposite safe option. There was again a significant association between the alarm choice probability and the non-alarm RT latency (r = 0.238, p < 0.001), as well as between the avoid choice probability and the approach RT latency (r = 0.246, p < 0.001) (Fig. 1e).
Distinct functional brain circuits for alarm-related and avoid-approach-related decisions
Making alarm/non-alarm and avoid/approach decisions on scream calls involves recognizing and evaluating the screaming call as well as choosing and performing a decision option. We modeled these two processing levels in two separate analyses of the functional brain data.
Using a task-based analysis approach, we created a statistical model including all seven scream types for both the ALARM task and the AA task (Fig. 2a). We contrasted brain activity for the ALARM task against the AA task, and this contrast revealed a broad network of active brain regions including secondary (PTe planum temporale) and higher-order auditory cortical regions (ST superior temporal cortex), the intraparietal sulcus (IPS), and premotor areas (PMC premotor cortex, SMA supplementary motor area) and motor-sensory cortex (MC motor cortex, SC sensory cortex). These regions typically belong to the auditory dorsal pathway involved in sensory-to-motor mapping of auditory information28,29 and to prepare context-specific action programs30. Additional activity was found in areas both for basic affective evaluations in the limbic striatum (Put putamen) and the hippocampus (HC) as well as in areas for elaborate affective evaluations in the insular-orbitofrontal complex (OFC orbitofrontal cortex, aINS anterior insula) and the medial frontal cortex (MFC). Activity was finally also observed in the cerebellum (Cbll) to serve immediate response adaptionsadaptations to potential alarm signals31 (Fig. 2a, left panel; Table S2).
a Task-based analysis: Functional cortical and subcortical brain activities for comparing the ALARM task with the AA task. b Choice-based analysis: Functional activity for comparing the ALARM with the AA task. Functional activity (n = 35) is thresholded with voxel threshold of p = 0.05 FDR corrected, and cluster extent threshold of k = 30.
When performing the reverse contrast for the task-based analysis, significantly higher brain activity during the AA task to the ALARM task was found in regions for integrative sensory processing in the posterior superior temporal sulcus (STS) and inferior temporal lobe (IT) and elaborate cognitive processing (PCu precuneus, PFC prefrontal cortex, ACC anterior cingulate cortex). Besides this higher-order cognitive processing, we also found activity in the dorsal striatum (Cd Caudate) perhaps to integrate temporal auditory information32,33 and to shift relevant decision criteria34, as well as in the amygdala-parahippocampal circuit (Amy amygdala, PHC parahippocampal cortex), indicative of basic integrative affective processes and potential spatial coding also during the AA task (Fig. 2a, right panel; Table S2). Crucial components of the avoid-approach judgments thus seem to be the decoding of space information in the PHC35, specifically regarding the location of the scream caller in relation to the location of potential threat source and the intended recipient36.
Next to the task-based analysis, we performed a second analysis focusing on brain activity associated with participants’ choices. We therefore modeled and grouped trials by participants’ decisions, independent of the scream call types, resulting in four regressors (alarm/non-alarm and avoid/approach). For this choice-based analysis (Fig. 2b, Table S3), we again compared the ALARM task against the AA task [ALARM task > AA task] and found similar activity as for the task-based analysis in regions belonging to the auditory dorsal stream, the insular-orbitofrontal cortex, and the Cbll. Interestingly, further activity was found in regions that were significantly active for the AA task during the task-based analysis, which became now active during the ALARM task for the choice-based analysis. This especially concerned regions in the posterior STS, IT, PCu, ACC, PHC, and also in the amygdala. Comparing the AA task against the ALARM task during the choice-based analysis did not reveal any significant brain activity.
Low-alarm and high-alarm scream calls
The analysis of the choice behavior and choice latencies during the analysis of the performance data indicated there might be a dichotomy in the seven scream types according to their alarm level. Based on the pattern of alarm choice probabilities (Fig. 1c, right panel) and the pattern of RT latency for the non-alarm option, we divided all seven scream call types into low-alarm screams (neutral, pleasure, joy) and high-alarm screams (sadness, pain, fear, anger). Given this differential alarm level across different scream types, we accordingly tested for differential brain activity for the low- and high-alarm screams during the ALARM task against the AA task during the task-based analysis (Fig. 3a). Brain activity for low-alarm and high-alarm screams for the ALARM task (Fig. 3a, Tables S4, 5) was very similar to the brain activity across all scream types (Fig. 2a, Tables S1, 2), but brain activity was spatially more extended the high-alarm screams. Some brain areas were furthermore only active for high-alarm screams during the ALARM task, such as the Put activity in the limbic striatum.
a Comparison of functional brain activity for the ALARM task against the AA task for low-alarm (neutral, pleasure, joy; left panel) and high-alarm scream calls (sadness, pain, fear, anger; right panel). b Comparison of functional brain activity for the AA task against the ALARM task for high alarm scream calls. Functional activity (n = 35) with voxel threshold p = 0.05 FDR and cluster threshold k = 30.
We used the distinction between low-alarm and high-alarm screams also to test for higher brain activity during the AA task (Fig. 3b, Table S6). Only high-alarm screams but not low-alarm screams revealed higher brain activity during the AA task. Again, this activity pattern (Fig. 3b) was very similar to the brain activity pattern for all scream types (Fig. 2a). Overall, it seems that brain activity in the comparison between the ALARM and the AA task seems predominantly but not exclusively driven by the high-alarm screams.
Alarm decisions on scream calls involve distinctive neural mechanisms
As outlined above, both the ALARM task and the AA task involve a decisional dichotomy between an urgent and a safe option. Using the choice-based analysis approach, we accordingly also examined brain activity related to choosing between these options involving risk arbitrations concerning external threat sources and own decisional processes (Fig. 4). Choosing the urgent decision options (alarm, avoid) against the safe options (non-alarm, approach) was associated with activity in low-order (HG Heschl’s gyrus, PTe) and higher order auditory cortex (ST), MFC, and Cbll (Fig. 4a, Table S7). A similar but spatially much more extended brain activity pattern was found when comparing the urgent alarm decision with the urgent avoid decision, but with additional peak activity locations in aINS, inferior frontal cortex (IFC), and MC (Fig. 4b, Table S8). Overall, it seems that the urgent options might have common neural mechanisms that support their decisional mechanisms, but the alarm decision requires additional neural computations.
Alarm and avoid levels of scream calls are inversely predictive of brain activity
The decisional performance of participants indicated a pattern of association between the choice probabilities for the urgent option and the RT latency pattern for the safe option across the scream types. We accordingly calculated an “alarm quotient” and an “avoid quotient” to parametrically quantify these choice-latency associations. Each quotient quantifies the level of choice-latency associations, with low differences (i.e., high similarities) between the z-scored choice parameter and the z-scored latency parameters signifying a high alarm and avoid relevance. These quotients were then used to predict brain activity (Fig. 5).
a Brain activity predicted by the combined factors of alarm choice probability and RT latency for non-alarm choices; the combined factors represent the “alarm quotient”. b Brain activity is predicted by the combined factors of avoid choice probability and RT latency for approach choices; the combined factors represent the “avoid quotient”. The lower panels show scatter plots for neural activity in the amygdala and hippocampus depending on the alarm and avoid quotient. Functional activity (n = 35) with voxel threshold p = 0.05 FDR and cluster threshold k = 30.
The alarm quotient could only predict brain activity in a negative direction, such that higher levels of the alarm quotient were associated with decreasing brain activity in many regions of the neural network that we reported for the ALARM task above. Surprisingly, we also found decreasing amygdala activity with increasing levels of the alarm quotient (Fig. 5a, Table S9).
Interestingly, the avoid quotient could predict brain activity in a mostly similar brain network in a positive direction, with some important regional differences to the alarm quotient. While the avoid quotient did not reveal any association with amygdala activity, it revealed a positive association with hippocampal and parahippocampal activity (Fig. 5b, Table S10).
Minimal neural overlap between alarm and avoid-approach processing
All previous analyses of functional brain data according to the alarm and avoid-approach evaluation and decision on scream calls revealed differences in the neural mechanisms that support both processing dimensions, with some minor potential overlaps in neural codes. In the final step of the analysis, we used a multivoxel pattern analysis (MVPA) approach to examine common neural patterns that underly the processing of scream calls during alarm/non-alarm and avoid/approach decisions (Fig. 6, Table S11). Based on the task-based analysis model as described above, we trained a support vector machine (SVM) classifier to distinguish the seven scream call types during the ALARM task and used this training data to predict neural activity to distinguish the seven scream calls during the AA task and vice versa. This analysis revealed neural patterns in the ST, right STS, and left PCu, where activity in one task could predict activity during the other task. A similar MVPA approach based on the choice-based analysis as described above did not reveal any common neural pattern between the ALARM task and the AA task related to discriminating between the two choice options within each task. This indicates some common but rather minimal neural overlap in distinguishing the scream calls across tasks but no neural overlap in distinguishing the two choice options across tasks.
Functional activity patterns resulted from a machine learning approach where neural patterns across the scream call types were trained for the ALARM task to predict brain activity for the AA task (and vice versa). Functional activity (n = 35) with voxel threshold p = 0.05 FDR and cluster threshold k = 30.
Discussion
We examined the commonalities and differences in alarm judgments and avoid-approach decisions during scream perception in humans, especially because the scream type repertoire and scream affect signaling in humans are much broader and more diverse than in other species. There were only minimal reaction time differences between the avoid and approach decision options and across the scream types. Most of the scream types elicited an equal mix of avoid and approach responses, but especially anger and pleasure scream calls led to an increased level of avoid decisions. While this pattern is unsurprising for anger screams, which also evoked the highest levels of alarm decisions, the high avoid responses to positive pleasure screams are surprising as humans and other species tend to approach sources and signalers of affective pleasure37,38. While pleasure is a strong positive emotion, it is also highly context-dependent as it can involve some levels of intimacy between the signaler and the recipient. Pleasure is therefore only unambiguously positive in close and intimate relationships, whereas it can feel rather harassing and aversive in unfamiliar social encounters, such as in our experiment. In this case, perceiving pleasure screams can evoke avoid rather than approach tendencies.
Unlike the rather uniform decisional patterns for the avoid-approach dimension, the alarm and non-alarm decision patterns and latencies were much differentiated across the different scream types. Overall, there was a considerable difference between the neutral and positive screams on one side (neutral, pleasure, joy) and the negative screams on the other side (sadness, pain, fear, anger). Latencies for urgent alarm decisions were fastest and latencies for the safe non-alarm option were slowest for negative screams. Fast alarm decisions in case of threat signaled by screams are vital and have biological significance39, and slow decisions for the non-alarm option indicate risk arbitrations to prevent false and potentially harmful decisions40,41. Next to this response latency pattern, negative screams had largely higher probabilities for alarm than for avoiding decisions, indicating some predominance of the alarm over the avoid assessment. Based on these decisional patterns, we classified the neutral and positive screams as low-alarm scream calls and the negative screams as high-alarm scream calls. This distinction follows largely a natural distinction between positive and negative affective voice signals42.
High-alarm scream calls revealed a pattern of fast alarm decisions, high alarm classification probabilities, and slow non-alarm decisions, while low-alarm screams showed an almost reverse pattern. We parametrically quantified these opposing decisional patterns by correlation analyses in terms of choice-latency associations. Across all seven scream types, higher probabilities of classifying a scream type as alarming were associated with slower and more careful classifications of some exemplars of this scream type as potentially non-alarming. A similar pattern of high avoid classification for certain scream types and slower approach classifications for some scream calls of the same type was evident during the avoid-approach judgments. This specific pattern of high-urgent-probability coupled with slow-safe-classifications was also shown in scream-type independent alarm and avoid quotients. This seems to point to a general biological principle of probabilistic risk arbitration that seems to be a critical extension of the current description of risk assessment and response in animals and humans43.
Although speeded urgent decisions are common observations during imminent threats39, a choice-latency coupling between the probability and the speed of the urgent decision was not observed here, which would be a within-trial coupling of decision parameters. Contrarily, the observed high-urgent-probability to slow-safe-classifications coupling is an across-trial coupling (i.e., trials with urgent option decisions are separate from trials with safe option decisions because of the 2-alternative forced choice task) and thus might reflect a more general two-factorial processing bias in risk assessment and arbitration that is not accounted for in current models of threat processing44,45. Two important notions concern this two-factorial processing bias. First, as an extension to previous models, it highlights the significance of mechanistically also considering the safe option when modeling the decisional and neural dynamics in threat assessment. Second, the two-factorial processing bias is related to individual differences in the relative combination of choice probabilities and decisional speed. The outcome of the threat assessment of scream calls is thus a combination of alarm signal features (trial-specific) and individual predispositions (trial-general).
An overall observation from the decisional patterns across the alarm judgments and the avoid-approach judgment was a somehow larger distinctiveness of the alarm dimension in making decisions across different positive and negative scream types. We accordingly also examined the neural commonalities and differences between both dimensions during the decoding of affective information from scream calls. For the task-based analysis of brain data, we found neural activity for the alarm task in regions that could be summarized as sensory and affective evaluations for motor mirroring46,47 and preparation in response to scream calls28,29. The neural network for the avoid-approach judgments was rather represented by brain regions for cognitive processing with the support of affective and spatial decoding mechanisms, and this might be summarized as a neural mechanism of spatio-affective evaluations for social decision-making48,49. The notion of spatio-affective evaluations especially refers to the spatial feature of many affectively important communications signals. Scream calls are communication signs for potential threats, and the location of threats (directing and distance) in relation to the scream caller and scream recipients is crucial affective information. There seemed to be some minimal neural overlap between alarm and avoid-approach judgments, especially at the level of sensory scream and acoustic pattern analysis in higher-order AC, which was particularly indicated by the results of the MVPA analysis. However, given the overall distinctiveness of the neural networks, it is indicative of a largely neural separation between alarm and avoid-approach judgments on scream calls.
For the task-based functional brain data analysis, we performed additional analyses and contrasts based on the distinction between low-alarm and high-alarm screams. The neural networks generally observed for the alarm and the avoid-approach judgment were largely replicated and seemed to be more strongly driven by the high-alarm screams. Affect information encoded in the high-alarm screams seems to be more important for neurally distinguishing the alarm and avoid-approach dimension during scream decoding. The high-alarm level of the screams might increase the neural alertness level in recipients for an accurate judgment and distinction along both dimensions.
A special notion concerns amygdala activity observed for the avoid-approach judgments but not for the alarm judgments. The amygdala is commonly assumed to be central in alarm signal assessments and threat responding45, but was found here to be more relevant for evaluating and arbitrating between avoid-approach choice options50. Interestingly, when switching to a choice-based analysis related to participants’ actual decisions, the amygdala showed significant activity for the alarm task compared to the avoid-approach task. Similarly, other neural nodes that were observed for the avoid-approach task during the task-based analysis (STS, ACC, PHC, PCu) were found for the alarm task during the choice-based analysis. This could suggest that a large extent of neural activity for the avoid-approach judgments is basically choice- and decision-based. Once the neural activity for the alarm judgments is analyzed in a choice-based framework, it also includes neural nodes for arbitrating choice options (alarm, non-alarm) in addition to neural nodes for auditory-to-motor preparation. The amygdala specifically might be thus more important for evaluation and arbitrating choice options rather than in immediate alarm responding1,42.
Making relevant and accurate decisions between choice options in response to scream calls is biologically and socially essential. As mentioned, the two tasks in this study included a similar biologically and neurally relevant dichotomy between an urgent and a safe option, and additionally the two urgent choice options might have further neural distinctions. Processing scream calls for which participants chose an urgent decision option (alarm, avoid) included a neural network for auditory scream and affect analysis (AC)1,51 as well as tuning of motor programming (Cbll)20 and of social response programming (MFC)31,52. A similar and extended neural network, including integrative socio-affective discrimination nodes (OFC, aINS, IFC)28,53 and motor mirroring nodes (MC)47, was found specifically for scream calls followed by urgent alarm compared to urgent avoid decisions. This again points to some level of neural primacy of the alarm dimension over the avoid-approach dimension during the processing of scream calls.
Alarm and avoid-approach judgments on scream calls thus seem to involve differential and predominant neural mechanisms that support the processing of relevant affect and action information from scream signals. We described above the unique choice-latency association that was illustrated by significant variations of the alarm and the avoid quotient. These quotients not only had a decisional relevance but also strongly affected the brain decoding of scream signals. The avoid quotient could positively predict neural activity in the broad auditory-frontal network for scream signal processing1,2, including positive associations with bilateral hippocampus activity that is typically associated with neural mechanisms for avoid-approach arbitration23.
A surprising observation however was the inverted neural prediction of the alarm quotient, such that an increasing alarm quotient was associated with decreasing neural activity in regions belonging to the dorsal (auditory-to-motor mapping) and ventral auditory stream (auditory-to-identification mapping). This negative association was specifically evident in neural nodes for alarm decoding and responding, such as the cerebellum, motor system, and particularly the amygdala1,45. Unlike an expected upregulation of cortical and especially limbic activity with increasing alarm levels, we rather observed a consistent suppression of neural recruitment for the highest alarm levels. This is corroborated by recently emerging reports about a signal decrease rather than an increase in the amygdala to alarming scream signals1, which is often also accompanied by suppressions of physiological reactions54,55. This suppression of neural and physiological activity could be indicative of threat-anticipatory freezing55 but also of a suppression of unnecessary neural activity and neural noise56 to allow focusing on the external threat57 and to program an adaptive neural response to the threat45,58. Especially for the amygdala, the phase of neural suppression during the alarming assessment might be followed by a phase of neural enhancement while performing the alarm/non-alarm decision, as found in our choice-based analysis, thus switching to cost-benefit assessment of response options59.
In summary, scream calls are biological signals that mostly indicate threat, but they can also communicate positive affect signals and safe contexts in human communication. Different scream types accordingly evoke differential alarm and avoid responses, with the alarm response showing a more diversified pattern than the avoid-approach response. Two surprising and novel findings emerged from our study. First, instead of a likelihood-to-speed coupling focused on the urgent decision only, we found a likelihood-to-slowing coupling between the urgent decision likelihood and the safe decision latency, which together indicates a broader decisional bias for accurate risk arbitration across response options. Second, the human brain seems to have different mechanisms to support alarm and avoid-approach judgments, with a certain primacy of the alarm over the avoid-approach assessment. Besides an overall increased brain decoding of alarm signaling from screams on a mean level of brain activity, the neural alarm decoding however showed a cortico-limbic pattern of suppression with the highest alarm levels. This is potentially indicative of a neural reset for preparing the neural system to threat responding60 and performing actual alarm/non-alarm decisions and alarm actions45.
Methods
Participants
A total of 38 healthy human volunteers took part in the experiment. No statistical methods were used to pre-determine the number of participants, but our sample size is larger than in previous human neuroimaging studies in scream call processing1,2. Three participants were excluded from data analysis because of either excessive movement (threshold 2-times in-plane voxel resolution, >3.5 mm movement in relation to the first image of the functional scan) during fMRI data acquisition and noticeable movement-related functional activity artifacts (n = 1) or because of too many missing responses (>25%, n = 2) during the experimental trials. The final sample thus included 35 participants (21 females, 14 males; mean age 27.20 years, SD 6.22, age range 18–46). All participants had normal hearing and normal or corrected-to-normal vision. No participant reported having any neurological disorder, lesion in the central nervous system, cognitive impairments, or a psychiatric history. Participants were recruited via public announcements at the University of Oslo and online ads on Meta platforms. The experiment was approved by the internal review board at the Psychological Institute of the University of Oslo (IRB, ref #27349367) and by the national Norwegian Agency for Shared Services in Education and Research (SIKT, ref #531225). All ethical regulations relevant to human research participants were followed.
Statistics and reproducibility
The statistical significance of behavioral data and decision patterns was quantified using parametric methods, including repeated measures analysis of variance (rmANOVA) and Pearson correlation approaches. The resulting correlation coefficients were Fisher z-transformed to allow parametric comparisons between conditions. To account for potential false-positive issues due to multiple comparisons, a family-wise error correction (FWER) was applied.
Neural brain activations were statistically analyzed using both a general linear mixed model (GLM) approach and a multivoxel pattern analysis (MVPA) approach. To account for potential false-positive issues resulting from multiple comparisons, we applied false discovery rate (FDR) correction methods to group-level activation maps across the whole brain61.
Experimental stimuli
During the fMRI experiment, participants listened to 84 different scream calls that were presented in a randomized order. Each scream call had a duration of 800 ms. The screams consisted of 6 scream types (pleasure, joy, sadness, pain, fear, anger) and an additional category of “neutral” screams (intense vocalizations of the vowel/a/). All scream calls were expressed by 3 male and 3 female speakers, with two calls of each scream type performed by each person. The sample of 84 screams was selected from a larger sample of screams collected and used in a previous study1. The screams were selected from a total of 420 screams such that affect recognition rate from the scream calls was equal across the seven scream types1. All scream samples were RMS normalized and presented at 70DB SPL. The samples were presented using MR-compatible headphones, including active noise-cancellation (OptoActive II; https://www.optoacoustics.com) that reduced MRI scanner noise by ~20 dB.
Experimental tasks
The whole experiment was split into four different blocks. In each block, we presented all 84 scream calls in random order. Each trial started with a white fixation cross presented for 500 ms on a screen, followed by an 800 ms scream call. The fixation cross served to maintain participants’ attention and to cue them for an upcoming sound trial.
The inter-stimulus interval was randomly jittered between 4.5 and 6 s. The experiment included two different tasks for the participants, with two blocks per task. Both tasks were 2-alternative forced-choice tasks, and on every trial, participants had to choose between two response options using a left- or right-hand response with their index finger. Participants responded using MRI compatible response grips (NordicNeuroLab; https://www.nordicneurolab.com/product/fmri-acquisition). Response options were counterbalanced across runs and participants.
The first task tested for the perceived dimension of the “alarm” level perceived from scream calls (ALARM task), where participants were asked to decide if the scream call expressed an “alarm” signal or if it was rather of “not-alarm” quality (response options, alarm, non-alarm). An alarming scream call is represented by the feeling of urgency to respond to the perceived voice signal, while perceiving a scream as not-alarming would not induce such an urgency. The second task tested the approach-avoidance tendency (AA task) during the perception of scream calls. Participants were asked to decide if they would potentially “avoid” or “approach” the person expressing the scream based on the perceived acoustic and affective quality (response options: avoid, approach). The order of the tasks was randomized and counterbalanced across participants.
Both tasks are similar in involving a decision between two response options, specifically in that one option represents a choice for urgency (alarm, avoid) while the other option can be regarded as the safe option (non-alarm, approach).
Brain data acquisition
Structural and functional brain data was recorded on a Philips Ingenia 3T MR scanner. High-resolution structural MRI was acquired using T1-weighted scans (TR 7.91 ms, TE 3.71 ms, voxel size 0.57 mm3, in-plane resolution 256 × 256 voxels). Functional whole-brain images were recorded with a T2-weighted echo-planar pulse sequence (TR 1.65 s, TE 30 ms, FA 88 ̊; in-plane resolution 128 × 128 voxels, voxel size 1.7 × 1.7 × 3.5 mm; slice gap 0.4 mm; 19 slices). For each participant, 293 functional images were acquired per block, amounting to a total of 1172 functional images per participant.
Analysis of decisional performance
RT in each task condition were subjected to separate repeated measures analysis of variance (rmANOVA) for each of the two tasks separately. The rmANOVA included the factors decision (2 levels: alarm/not-alarm or avoid/approach) and the factor scream type (7 levels: neutral, pleasure, joy, sadness, pain, fear, anger) as within-subject factors.
Furthermore, the participants’ choice probabilities of alarm and avoid decisions were subjected to another rmANOVA model, with choice type (2 levels: alarm choice, avoid choice) and scream type (7 levels) as within-subject factors. Please note that the probabilities of non-alarm and approach decisions were not included in this model because they directly correspond to the alarm and avoid choices (i.e., %approach = 1 − %avoid). Significant results from any of the rmANOVA models were followed up with posthoc comparisons corrected for the FWER using a Bonferroni correction.
Based on the resulting pattern for the choice probabilities for the urgent decision (alarm, avoid) and the RT latencies for the safe decision (non-alarm, approach), we quantified the similarity of the choice-latency associations using correlation analysis. We quantified the Pearson correlation coefficient by cross-correlating all choice probability patterns with all RT latency patterns per participant. Correlation coefficients were Fisher z-transformed and averaged across participants and tested for significance using the two-sided t-distribution. All p values were corrected for multiple comparisons using FWER correction.
To further parametrically quantify the association between the alarm choice patterns and RT non-alarm latency pattern, as well as between the avoid choice pattern and the RT approach latency pattern, we quantified the “alarm quotient” and the “avoid quotient”, respectively. The alarm quotient represents the level of subjective alarmingness by quantifying the similarity between the increased level of an urgent alarm choice probability to a screaming call and the level of increased RT latency to the safe non-alarm decision. An increased RT to the safe non-alarm option indicates that participants are potentially aware of the risk of making a false non-alarm decision in case of a real threat. The avoid quotient quantifies the same pattern for the avoid-approach decision, with an increased avoid quotient indicating high avoid choices combined with increased latency and thus risk awareness for the (potentially wrong) approach choice. The quotients were quantified for each scream call type and each participant, and we used the following formula based on choice and RT parameters normalized to a range of [0, 1]:
quotient ~ [1 − abs(choice − RT)] * mean(choice + RT)
Preprocessing of brain data
Preprocessing and statistical analyses of the functional MRI images were performed with the Statistical Parametric Mapping software (SPM12, Wellcome Centre for Human Neuroimaging, London; https://www.fil.ion.ucl.ac.uk/spm/). Data preprocessing was done according to a standard pipeline. First, functional and structural data were manually aligned to the AC-PC axis, followed by motion correction (realignment, first image in the time series as reference image) of the functional images. The structural image of each participant was co-registered to the mean functional image, before the structural image was segmented to allow estimation of normalization parameters. Using the estimated normalization parameters, the anatomical and functional images were spatially normalized to the MNI stereotactic space. The functional images were then spatially smoothed with an 8 mm full-width half-maximum isotropic Gaussian kernel.
Statistical analysis of brain data
Statistical analysis of the functional brain data was also performed in SPM12. For a first-level GLM analysis, two separate models were created. First, the task-based GLM model included seven regressors for each of the seven scream types for each of the ALARM task and the AA task, resulting in a total of 14 different regressors. Second, the choice-based GLM model included a total of 4 regressors based on participants’ choice on each trial (alarm and non-alarm, avoid and approach). The reaction time was included as a covariate of no interest on a trial-by-trial basis in both GLM models to account for RT differences between59 trials that may produce signal changes that are unrelated to the decision or conditions of interest. Six movement regressors per block were also included as regressors of no interest to account for potential movement-related activation artifacts. For all trials, a stick function convolved with a standard hemodynamic response function was aligned to the onset of each stimulus.
The resulting contrast images per condition (14 conditions for the task-based GLM model, 4 conditions for the choice-based GLM model) from the first-level analysis were then taken to a random-effects group analysis. Several directed contrasts were performed to compare functional brain activity across conditions. Significant voxel activations were thresholded at p = 0.05 and adjusted for multiple testing using the FDR correction61. The minimum cluster size was set to k = 30 to determine clusters of activation with a reasonable spatial representation and to avoid relatively small clusters of less relevance for the experimental question.
Multivoxel pattern analysis
Using a searchlight brain decoding approach, we defined a local sphere of a 6 mm radius at each voxel to investigate the local multivoxel pattern information in the single-trial beta images that were able to differentiate across the seven scream types for the ALARM task and the AA task. This analysis is based on the task-based GLM analysis as described above. For this MVPA approach, we used The Decoding Toolbox (TDT, version 3.999F, https://sites.google.com/site/tdtdecodingtoolbox/). We trained a multivoxel SVM classifier by using a linear kernel (linear C-SVM), which was trained on 5/6 of the trials and tested on the remaining 1/6 of the trials of either the ALARM task trials or the AA task trials. The training of the classifier was performed on unsmoothed brain data.
Using a cross-classification approach, we used the training data from the ALARM task to predict brain patterns for the AA task and vice versa. This procedure resulted in brain maps of local classification accuracy across the scream types for each participant. The accuracy maps represented that relative accuracy above chance level (i.e., [accuracy − chance]). The resulting accuracy maps were spatially smoothed with an 8 mm full-width half-maximum isotropic Gaussian kernel and subjected to second-level random effects GLM analysis. On the second level, we tested the resulting map for statistical significance against the chance level by a voxel thresholded of p = 0.05 (FDR corrected) and a minimum cluster size was set to k = 30 to considerably minimize the possibility of false positive activity patterns.
The MVPA cross-classification analysis was also performed using the choice-based GLM analysis scheme as described above. We trained the SVM classifier to distinguish the alarm and non-alarm choice trials during the ALARM task and to predict brain activity to distinguish the avoid and approach choice trials during the AA task and vice versa. The training was performed on unsmoothed brain data using a searchlight analysis approach. The resulting relative accuracy maps (i.e., [accuracy − chance]) were again spatially smoothed with an 8 mm full-width half-maximum isotropic Gaussian kernel and subjected to second-level random effects GLM analysis (voxel thresholded p = 0.05 FDR corrected, cluster size threshold k = 30).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Data are available upon reasonable request. The conditions of ethics approval and consent procedures do not permit public archiving of anonymized study data. Numerical source data for graphs and charts are deposited here: https://github.com/caneuro/scream.decision.
Code availability
This paper does not report original codes. Codes are available on reasonable requests.
References
Frühholz, S., Dietziker, J., Staib, M. & Trost, W. Neurocognitive processing efficiency for discriminating human non-alarm rather than alarm scream calls. PLoS Biol. 19, e3000751 (2021).
Arnal, L. H., Flinker, A., Kleinschmidt, A., Giraud, A. L. & Poeppel, D. Human screams occupy a privileged niche in the communication soundscape. Curr. Biol. 25, 2051–2056 (2015).
Hogstedt, G. Adaptation unto death: function of fear screams. Am. Nat. 121, 562–570 (1983).
Beaurenaut, M., Tokarski, E., Dezecache, G. & Grèzes, J. The ‘Threat of Scream’ paradigm: a tool for studying sustained physiological and subjective anxiety. Sci. Rep. 10, 12496 (2020).
Schwartz, J. W., Engelberg, J. W. M. & Gouzoules, H. Evolving views on cognition in animal vocal communication: contributions from scream research. Anim. Behav. Cogn. 7, 192–213 (2020).
Schwartz, J. W., Engelberg, J. W. M. & Gouzoules, H. Was that a scream? Listener agreement and major distinguishing acoustic features. J. Nonverbal Behav. 44, 233–252 (2020).
Anikin, A. & Persson, T. Nonlinguistic vocalizations from online amateur videos for emotion research: a validated corpus. Behav. Res. Methods 49, 758–771 (2017).
Magrath, R. D., Haff, T. M., Fallow, P. M. & Radford, A. N. Eavesdropping on heterospecific alarm calls: from mechanisms to consequences. Biol. Rev. 90, 560–586 (2015).
Gouzoules, H., Gouzoules, S. & Tomaszycki, M. Agonistic screams and the classification of dominance relationships: are monkeys fuzzy logicians?. Anim. Behav. 55, 51–60 (1998).
Miller, D. B. & Blaich, C. F. Alarm call responsivity of mallard ducklings: VII. Auditory experience maintains freezing. Dev. Psychobiol. 21, 523–533 (1988).
Manser, M. B., Bell, M. B. & Fletcher, L. B. The information that receivers extract from alarm calls in suricates. Proc. Biol. Sci. R. Soc. 268, 2485 (2001).
Elliot, A. J. & Gable, S. L. Handbook of Approach and Avoidance Motivation. https://doi.org/10.1177/0146167205282153 (2013).
Hans Phaf, R., Mohr, S. E., Rotteveel, M. & Wicherts, J. M. Approach, avoidance, and affect: a meta-analysis of approach-avoidance tendencies in manual reaction time tasks. Front. Psychol. 5, 80231 (2014).
Higgins, E. T. Beyond pleasure and pain. Am. Psychol. 52, 1280–1300 (1997).
Epstein, S. Avoidance-approach: the fifth basic conflict. J. Consult. Clin. Psychol. 46, 1016–1022 (1978).
Kang, S. J. et al. A central alarm system that gates multi-sensory innate threat cues to the amygdala. Cell Rep. 40, 111222 (2022).
Freeman, A. R., Hare, J. F. & Caldwell, H. K. Call-specific patterns of neural activation in auditory processing of Richardson’s ground squirrel alarm calls. Brain Behav. 10, e01629 (2020).
Leon, M. I., Poytress, B. S. & Weinberger, N. M. Avoidance learning facilitates temporal processing in the primary auditory cortex. Neurobiol. Learn. Mem. 90, 347–357 (2008).
Rabellino, D., Boyd, J. E., McKinnon, M. C. & Lanius, R. A. The innate alarm system: a translational approach. Stress. Physiol. Biochem. Pathol. Handb. Stress. Ser. 3, 197–212 (2019).
Ceravolo, L., Frühholz, S., Pierce, J., Grandjean, D. & Péron, J. Basal ganglia and cerebellum contributions to vocal emotion processing as revealed by high-resolution fMRI. Sci. Rep. 11, 10645 (2021).
Williams, L. M. et al. Amygdala-prefrontal dissociation of subliminal and supraliminal fear. Hum. Brain Mapp. 27, 652–661 (2006).
Aupperle, R. L., Melrose, A. J., Francisco, A., Paulus, M. P. & Stein, M. B. Neural substrates of approach-avoidance conflict decision-making. Hum. Brain Mapp. 36, 449–462 (2015).
Bach, D. R. et al. Human hippocampus arbitrates approach-avoidance conflict. Curr. Biol. 24, 541–547 (2014).
McNaughton, N. & Gray, J. A. The Neuropsychology of Anxiety: An Enquiry Into the Functions of the Septo-hippocampal System 1–649 https://doi.org/10.1093/OSO/9780198843313.001.0001 (2024).
Sánchez-Bellot, C., AlSubaie, R., Mishchanchuk, K., Wee, R. W. S. & MacAskill, A. F. Two opposing hippocampus to prefrontal cortex pathways for the control of approach and avoidance behaviour. Nat. Commun. 2022 13, 1–17 (2022).
Korn, C. W. & Bach, D. R. Minimizing threat via heuristic and optimal policies recruits hippocampus and medial prefrontal cortex. Nat. Hum. Behav. 2019 3, 733–745 (2019).
Ito, R. & Lee, A. C. H. The role of the hippocampus in approach-avoidance conflict decision-making: evidence from rodent and human studies. Behav. brain Res. 313, 345–357 (2016).
Frühholz, S. & Grandjean, D. Processing of emotional vocalizations in bilateral inferior frontal cortex. Neurosci. Biobehav. Rev. 37, 2847–2855 (2013).
Rauschecker, J. P. Dual stream models of auditory vocal communication. The Oxford Handbook of Voice Perception 413 (Oxford University Press, 2018).
Rauschecker, J. P. & Scott, S. K. Maps and streams in the auditory cortex: nonhuman primates illuminate human speech processing. Nat. Neurosci. 12, 718–724 (2009).
Frühholz, S., Trost, W. & Kotz, S. A. The sound of emotions-towards a unifying neural network perspective of affective sound processing. Neurosci. Biobehav. Rev. 68, 1–15 (2016).
Pannese, A., Grandjean, D. & Frühholz, S. Subcortical processing in auditory communication. Hear. Res. 328, 67–77 (2015).
Kotz, S. A. & Schwartze, M. Cortical speech processing unplugged: a timely subcortico-cortical framework. Trends Cogn. Sci. 14, 392–399 (2010).
Kuchinke, L. et al. Human striatal activation during adjustment of the response criterion in visual word recognition. Neuroimage 54, 2412–2417 (2011).
Mormann, F. et al. Scene-selective coding by single neurons in the human parahippocampal cortex. Proc. Natl. Acad. Sci. USA 114, 1153–1158 (2017).
Ceravolo, L., Frühholz, S. & Grandjean, D. Proximal vocal threat recruits the right voice-sensitive auditory cortex. Soc. Cogn. Affect Neurosci. 11, 793–802 (2016).
Pradhan, G. R., Engelhardt, A., Van Schaik, C. P. & Maestripieri, D. The evolution of female copulation calls in primates: a review and a new model. Behav. Ecol. Sociobiol. 59, 333–343 (2006).
Bolt, L. M. Mating calls in non-human primates. Encyclopedia of Sexual Psychology and Behavior 1–3 https://doi.org/10.1007/978-3-031-08956-5_195-1 (2023).
McLachlan, J. R. & Magrath, R. D. Speedy revelations: how alarm calls can convey rapid, reliable information about urgent danger. Proc. R. Soc. B 287, 20192772 (2020).
Lima, S. L. & Dill, L. M. Behavioral decisions made under the risk of predation: a review and prospectus. 68, 619–640. https://doi.org/10.1139/z90-092 (2011).
Creel, S. & Christianson, D. Relationships between direct predation and risk effects. Trends Ecol. Evol. 23, 194–201 (2008).
Dricu, M. & Frühholz, S. Perceiving emotional expressions in others: activation likelihood estimation meta-analyses of explicit evaluation, passive perception and incidental perception of emotions. Neurosci. Biobehav. Rev. 71, 810–828 (2016).
Blanchard, D. C., Griebel, G., Pobbe, R. & Blanchard, R. J. Risk assessment as an evolved threat detection and analysis process. Neurosci. Biobehav. Rev. 35, 991–998 (2011).
Bentz, D. & Schiller, D. Threat processing: models and mechanisms. Wiley Interdiscip. Rev. Cogn. Sci. 6, 427–439 (2015).
Emerson, R. W., Ledoux, J. & Daw, N. D. Surviving threats: neural circuit and computational implications of a new taxonomy of defensive behaviour. Nat. Rev. Neurosci. 19, 269–282 (2018).
Schurz, M., Radua, J., Aichhorn, M., Richlan, F. & Perner, J. Fractionating theory of mind: a meta-analysis of functional brain imaging studies. Neurosci. Biobehav. Rev. 42, 9–34 (2014).
Van Overwalle, F. & Baetens, K. Understanding others’ actions and goals by mirror and mentalizing systems: a meta-analysis. Neuroimage 48, 564–584 (2009).
Terenzi, D., Liu, L., Bellucci, G. & Park, S. Q. Determinants and modulators of human social decisions. Neurosci. Biobehav. Rev. 128, 383–393 (2021).
Dricu, M. & Frühholz, S. A neurocognitive model of perceptual decision-making on emotional signals. Hum. Brain Mapp. 41, 1532–1556 (2020).
Schlund, M. W. & Cataldo, M. F. Amygdala involvement in human avoidance, escape and approach behavior. Neuroimage 53, 769 (2010).
Frühholz, S., Rodriguez, P., Bonard, M., Steiner, F. & Bobin, M. Psychoacoustic and Archeoacoustic nature of ancient Aztec skull whistles. Commun. Psychol. 2, 1–14 (2024).
Trevor, C. & Frühholz, S. Music as an evolved tool for socio-affective fiction. Emot. Rev. 16, 180–194 (2024).
Cohen, Y. E. et al. A functional role for the ventrolateral prefrontal cortex in non-spatial auditory cognition. Proc. Natl. Acad. Sci. USA 106, 20045–20050 (2009).
Olszyński, K. H., Polowy, R., Małż, M., Boguszewski, P. M. & Filipkowski, R. K. Playback of alarm and appetitive calls differentially impacts vocal, heart-rate, and motor response in rats. iScience 23, 101577 (2020).
de Voogd, L. D., Hagenberg, E., Zhou, Y. J., de Lange, F. P. & Roelofs, K. Acute threat enhances perceptual sensitivity without affecting the decision criterion. Sci. Rep. 12, 1–11 (2022).
Meyer, H. C., Sangha, S., Radley, J. J., LaLumiere, R. T. & Baratta, M. V. Environmental certainty influences the neural systems regulating responses to threat and stress. Neurosci. Biobehav. Rev. 131, 1037–1055 (2021).
Parasuraman, R. et al. Detecting threat-related intentional actions of others: effects of image quality, response mode, and target cuing on vigilance. https://doi.org/10.1037/a0017132 (2009).
Levy, I. & Schiller, D. Neural computations of threat. Trends Cogn. Sci. 25, 151–171 (2021).
Ghods-Sharifi, S., St. Onge, J. R. & Floresco, S. B. Fundamental contribution by the basolateral amygdala to different forms of decision making. J. Neurosci. 29, 5251–5259 (2009).
Schallmo, M. P. et al. Suppression and facilitation of human neural responses. Elife 7, e30334 (2018).
Genovese, C. R., Lazar, N. A. & Nichols, T. Thresholding of statistical maps in functional neuroimaging using the false discovery rate. Neuroimage 15, 870–878 (2002).
Acknowledgements
S.F. was supported by the Swiss National Science Foundation (SNSF 100014_182135/1 to S.F.). We thank Nicole Merz and Margrete Soya Heimvik for their help with data acquisition.
Author information
Authors and Affiliations
Contributions
G.B. contributed to data acquisition, data analysis, and writing the manuscript. C.S. contributed to data acquisition. S.F. contributed to designing the study, data acquisition, data analysis, and writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Philip Deming and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Beinnes, G.K., Skjegstad, C. & Frühholz, S. Differential neural decoding of alarm and avoidance information from vocal alarm calls in humans. Commun Biol 8, 818 (2025). https://doi.org/10.1038/s42003-025-08248-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-025-08248-9