Abstract
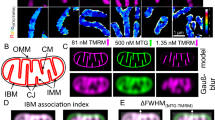
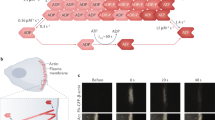
Campylobacter jejuni, a major cause of bacterial gastroenteritis, is capable of surviving in diverse hosts, including free-living amoebae such as Acanthamoeba. However, the molecular mechanisms that facilitate its intracellular persistence and subsequent transfer remain poorly defined. Here, we hypothesize that C. jejuni employs a biphasic actin-remodelling strategy, mediated by the effector proteins CiaI and CiaD, to reposition and remodel host mitochondria, promoting mitochondrial aggregation and iron homoeostasis. Using dual proteomics, microscopy, biochemical assays, and defined genetic mutants, we show that actin polymerization and CiaI are critical for mitochondrial interaction. We found that CiaI binds nucleotides with cooperative kinetics, acting as a molecular switch, and is crucial for C. jejuni localization near mitochondria, while CiaD promotes actin polymerization and acanthopodia formation to facilitate uptake. We propose a two-phase model: early actin polymerization repositions mitochondria, followed by localized actin depolymerization and mitochondrial remodelling. Iron chelation promotes bacterial survival, suggesting that oxidative stress functions as a host defence. These findings highlight a sophisticated mechanism of intracellular adaptation by C. jejuni that may be relevant to pathogenesis and identify new potential targets for disrupting its environmental and clinical persistence.
Similar content being viewed by others
Data availability
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD065850, and all analysed data underlying Table 1 can be found in Supplementary Data 1 and 2. All biological replicate flow cytometry data presented in this study can be found in the supplementary Information. Source values underlying all graphs presented in the study can be found in supplementary data 3.
References
Kellermann, M., Scharte, F. & Hensel, M. Manipulation of Host Cell Organelles by Intracellular Pathogens. Int. J. Mol. Sci. 22, https://doi.org/10.3390/ijms22126484 (2021).
Spier, A., Stavru, F. & Cossart, P. Interaction between Intracellular bacterial pathogens and host cell mitochondria. Microbiol Spectr. 7, 0016–2019 (2019).
Gray, M. W. Mitochondrial evolution. Cold Spring Harb. Perspect. Biol. 4, a011403 (2012).
Dan Dunn, J., Alvarez, L. A. J., Zhang, X. & Soldati, T. Reactive oxygen species and mitochondria: A nexus of cellular homeostasis. Redox Biol. 6, 472–485 (2015).
Silwal, P., Kim, J. K., Kim, Y. J. & Jo, E. K. Mitochondrial reactive oxygen species: double-edged weapon in host defense and pathological inflammation during infection. Front Immunol. 11, 1649 (2020).
Tiku, V., Tan, M. W. & Dikic, I. Mitochondrial functions in infection and immunity. Trends Cell Biol. 30, 263–275 (2020).
Lobet, E., Letesson, J.-J. & Arnould, T. Mitochondria: A target for bacteria. Biochem. Pharmacol. 94, 173–185 (2015).
Cervantes-Silva, M. P., Cox, S. L. & Curtis, A. M. Alterations in mitochondrial morphology as a key driver of immunity and host defence. EMBO Rep. 22, e53086 (2021).
Escoll, P. et al. Legionella pneumophila modulates mitochondrial dynamics to trigger metabolic repurposing of infected macrophages. Cell Host microbe 22, 302–316.e307 (2017).
Khan, S., Raj, D., Jaiswal, K. & Lahiri, A. Modulation of host mitochondrial dynamics during bacterial infection. Mitochondrion 53, 140–149 (2020).
Marchi, S., Morroni, G., Pinton, P. & Galluzzi, L. Control of host mitochondria by bacterial pathogens. Trends Microbiol. 30, 452–465 (2022).
Qi, B. & Han, M. Microbial Siderophore Enterobactin promotes mitochondrial iron uptake and development of the host via interaction with ATP Synthase. Cell 175, 571–582.e511 (2018).
Ruan, H. et al. The Salmonella effector SopB prevents ROS-induced apoptosis of epithelial cells by retarding TRAF6 recruitment to mitochondria. Biochem. Biophys. Res. Commun. 478, 618–623 (2016).
Omole, Z. et al. Pathogenicity and virulence of Campylobacter jejuni: What do we really know? Virulence 15, 2436060 (2024).
Lopes, G. V. et al. Virulence factors of the foodborne pathogen Campylobacter jejuni. Microb. Pathogenes. 161, 105265 (2021).
Desvaux, M., Hébraud, M., Henderson, I. R. & Pallen, M. J. Type III secretion: what’s in a name? Trends Microbiol. 14, 157–160 (2006).
Young, G. M., Schmiel, D. H. & Miller, V. L. A new pathway for the secretion of virulence factors by bacteria: the flagellar export apparatus functions as a protein-secretion system. Proc. Natl. Acad. Sci. 96, 6456–6461 (1999).
Konkel, M. E., Kim, B. J., Rivera-Amill, V. & Garvis, S. G. Bacterial secreted proteins are required for the internaliztion of Campylobacter jejuni into cultured mammalian cells. Mol. Microbiol. 32, 691–701 (1999).
Rivera-Amill, V., Kim, B. J., Seshu, J. & Konkel, M. E. Secretion of the virulence-associated Campylobacter invasion antigens from Campylobacter jejuni requires a stimulatory signal. J. Infect. Dis. 183, 1607–1616 (2001).
Gabbert, A. D. et al. The missing pieces: the role of secretion systems in Campylobacter jejuni Virulence. Biomolecules 13, 135 (2023).
Axelsson-Olsson, D., Waldenström, J., Broman, T., Olsen, B. & Holmberg, M. Protozoan Acanthamoeba polyphaga as a potential reservoir for Campylobacter jejuni. Appl. Environ. Microbiol. 71, 987–992 (2005).
Mohammadi, A., Dalimi, A., Ghaffarifar, F., Pirestani, M. & Akbari, M. Detection of Acanthamoeba Harboring Campylobacter jejuni Endosymbionts in Hospital Environments of Markazi Province, Iran. J. Parasitol. Res 2025, 6626888 (2025).
Nasher, F. & Wren, B. W. Transient internalization of Campylobacter jejuni in Amoebae enhances subsequent invasion of human cells. Microbiology. 168, https://doi.org/10.1099/mic.0.001143 (2022).
Vieira, A., Seddon, A. M. & Karlyshev, A. V. Campylobacter-Acanthamoeba interactions. Microbiology 161, 933–947 (2015).
Axelsson-Olsson, D. et al. Amoebae and algae can prolong the survival of Campylobacter species in co-culture. Exp. Parasitol. 126, 59–64 (2010).
Price, C. hristopher et al. Amoebae as training grounds for microbial pathogens. mBio 0, e00827–00824 (2024).
Nasher, F., Lehri, B., Stabler, R. A. & Wren, B. W. Acanthamoeba castellanii as a model for unveiling Campylobacter jejuni host–pathogen dynamics. Front. Cell. Infect. Microbiol. 15, https://doi.org/10.3389/fcimb.2025.1583830 (2025).
Canonico, B. et al. Campylobacter jejuni cell lysates differently target mitochondria and lysosomes on HeLa cells. Apoptosis 19, 1225–1242 (2014).
Hohmann, T. & Dehghani, F. The Cytoskeleton-A Complex interacting meshwork. Cells 8, https://doi.org/10.3390/cells8040362 (2019).
Pizarro-Cerdá, J., Chorev, D. S., Geiger, B. & Cossart, P. The diverse family of Arp2/3 complexes. Trends Cell Biol. 27, 93–100 (2017).
Blanchoin, L. & Pollard, T. D. Interaction of actin monomers with Acanthamoeba actophorin (ADF/cofilin) and profilin. J. Biol. Chem. 273, 25106–25111 (1998).
Mermall, V., Post, P. L. & Mooseker, M. S. Unconventional myosins in cell movement, membrane traffic, and signal transduction. Science 279, 527–533 (1998).
Krishnan, K. & Moens, P. D. J. Structure and functions of profilins. Biophys. Rev. 1, 71–81 (2009).
Watson, S. A. & McStay, G. P. Functions of Cytochrome c oxidase assembly factors. Int. J. Mol. Sci. 21, https://doi.org/10.3390/ijms21197254 (2020).
Sorribes-Dauden, R., Peris, D., Martínez-Pastor, M. T. & Puig, S. Structure and function of the vacuolar Ccc1/VIT1 family of iron transporters and its regulation in fungi. Comput. Struct. Biotechnol. J. 18, 3712–3722 (2020).
Ferrara, L. G. et al. MOMP from Campylobacter jejuni is a trimer of 18-stranded β-barrel monomers with a Ca2+ ion bound at the constriction zone. J. Mol. Biol. 428, 4528–4543 (2016).
Kabongo, A. T. et al. Biochemical characterization and identification of ferulenol and embelin as potent inhibitors of malate: quinone oxidoreductase from Campylobacter jejuni. Front. Mol. Biosci. 10, 1095026 (2023).
Grant, K. A. & Park, S. F. Molecular characterization of katA from Campylobacter jejuni and generation of a catalase-deficient mutant of Campylobacter coli by interspecific allelic exchange. Microbiology 141, 1369–1376 (1995).
van Vliet, A. H., Ketley, J. M., Park, S. F. & Penn, C. W. The role of iron in Campylobacter gene regulation, metabolism and oxidative stress defense. FEMS Microbiol. Rev. 26, 173–186 (2002).
Holmes, C. W., Penn, C. W. & Lund, P. A. The hrcA and hspR regulons of Campylobacter jejuni. Microbiology 156, 158–166 (2010).
Nachamkin, I., Yang, X. H. & Stern, N. J. Role of Campylobacter jejuni flagella as colonization factors for three-day-old chicks: analysis with flagellar mutants. Appl. Environ. Microbiol. 59, 1269–1273 (1993).
Nasher, F. & Wren, B. W. Flagellin O-linked glycans are required for the interactions between Campylobacter jejuni and Acanthamoebae castellanii. Microbiology 169, https://doi.org/10.1099/mic.0.001386 (2023).
Goley, E. D. & Welch, M. D. The ARP2/3 complex: an actin nucleator comes of age. Nat. Rev. Mol. Cell Biol. 7, 713–726 (2006).
Nasher, F., Lehri, B., Horney, M. F., Stabler, R. A. & Wren, B. W. Survival of Campylobacter jejuni 11168H in Acanthamoebae castellanii Provides Mechanistic Insight into Host Pathogen Interactions. Microorganisms 10, https://doi.org/10.3390/microorganisms10101894 (2022).
Barrero-Tobon, A. M. & Hendrixson, D. R. Flagellar biosynthesis exerts temporal regulation of secretion of specific Campylobacter jejuni colonization and virulence determinants. Mol. Microbiol. 93, 957–974 (2014).
Konkel, M. E. et al. Secretion of virulence proteins from Campylobacter jejuni is dependent on a functional flagellar export apparatus. J. Bacteriol. 186, 3296–3303 (2004).
Takai, Y., Sasaki, T. & Matozaki, T. Small GTP-Binding Proteins. Physiol. Rev. 81, 153–208 (2001).
Satoh, T. Diverse physiological functions and regulatory mechanisms for signal-transducing small GTPases. Int. J. Mol. Sci. 21, 7291 (2020).
Demkiv, A. O. et al. Redefining the limits of functional continuity in the early evolution of P-Loop NTPases. Mol. Biol. Evol. 42, https://doi.org/10.1093/molbev/msaf055 (2025).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).
Wang, J. et al. The conserved domain database in 2023. Nucleic Acids Res. 51, D384–D388 (2022).
Negretti, N. M. et al. The Campylobacter jejuni CiaD effector co-opts the host cell protein IQGAP1 to promote cell entry. Nat. Commun. 12, 1339 (2021).
Samuelson, D. R. & Konkel, M. E. Serine phosphorylation of cortactin is required for maximal host cell invasion by Campylobacter jejuni. Cell Commun. Signal. 11, 1–15 (2013).
Sharafutdinov, I., Knorr, J., Rottner, K., Backert, S. & Tegtmeyer, N. Cortactin: A universal host cytoskeletal target of Gram-negative and Gram-positive bacterial pathogens. Mol. Microbiol. 118, 623–636 (2022).
Dietz, J. V., Fox, J. L. & Khalimonchuk, O. Down the Iron Path: Mitochondrial Iron Homeostasis and Beyond. Cells 10, https://doi.org/10.3390/cells10092198 (2021).
Iovine, N. M. Resistance mechanisms in Campylobacter jejuni. Virulence 4, 230–240 (2013).
Albert, M. J. et al. Identification of a Campylobacter jejuni Protein That Cross-Reacts with Cholera Toxin. Infect. Immun. 75, 3070–3073 (2007).
Quintana, A. et al. T cell activation requires mitochondrial translocation to the immunological synapse. Proc. Natl. Acad. Sci. 104, 14418–14423 (2007).
Weinberg, S. amuelE., Sena, L. auraA. & Chandel, N. avdeepS. Mitochondria in the Regulation of Innate and Adaptive Immunity. Immunity 42, 406–417 (2015).
Fung, T. S., Chakrabarti, R. & Higgs, H. N. The multiple links between actin and mitochondria. Nat. Rev. Mol. Cell Biol. 24, 651–667 (2023).
Fang, F. C. Antimicrobial actions of reactive oxygen species. mBio 2, https://doi.org/10.1128/mBio.00141-11 (2011).
Buelow, D. R., Christensen, J. E., Neal-McKinney, J. M. & Konkel, M. E. Campylobacter jejuni survival within human epithelial cells is enhanced by the secreted protein CiaI. Mol. Microbiol. 80, 1296–1312 (2011).
Hardie, D. G. AMP-activated/SNF1 protein kinases: conserved guardians of cellular energy. Nat. Rev. Mol. Cell Biol. 8, 774–785 (2007).
Hardie, D. G. & Evans, A. M. (Oxford University Press, 2019).
Rayamajhee, B. et al. Acanthamoeba, an environmental phagocyte enhancing survival and transmission of human pathogens. Trends Parasitol. 38, 975–990 (2022).
Ben Zichri- David, S., Shkuri, L. & Ast, T. Pulling back the mitochondria’s iron curtain. npj Metab. Health Dis. 3, 6 (2025).
Galaris, D., Barbouti, A. & Pantopoulos, K. Iron homeostasis and oxidative stress: An intimate relationship. Biochim. Biophys. Acta (BBA) - Mol. Cell Res. 1866, 118535 (2019).
Handy, D. E. & Loscalzo, J. Redox regulation of mitochondrial function. Antioxid. Redox Signal 16, 1323–1367 (2012).
Zhang, Y. & Wong, H. S. Are mitochondria the main contributor of reactive oxygen species in cells? J. Exp. Biol. 224, https://doi.org/10.1242/jeb.221606 (2021).
Tirichen, H. et al. Mitochondrial reactive oxygen species and their contribution in chronic kidney disease progression through oxidative stress. Front. Physiol. 12, 627837 (2021).
Illescas, M., Peñas, A., Arenas, J., Martín, M. A. & Ugalde, C. Regulation of mitochondrial function by the actin cytoskeleton. Front. Cell Dev. Biol. 9, 795838 (2021).
Vickerman, K. Structural changes in mitochondria of Acanthamoeba at encystation. Nature 188, 248–249 (1960).
Cameron, A. & Gaynor, E. C. Hygromycin B and apramycin antibiotic resistance cassettes for use in Campylobacter jejuni. PLoS One 9, e95084 (2014).
Nasher, F. & Wren, B. W. Unravelling mechanisms of bacterial recognition by Acanthamoeba: insights into microbial ecology and immune responses. Front. Microbiol. 15, https://doi.org/10.3389/fmicb.2024.1405133 (2024).
Acknowledgements
We thank Christian Chiu for providing training, technical assistance and analyses using flow cytometry. We thank Steven Lynham and Xiaoping Yang from the Centre of Excellence for Mass Spectrometry at King’s College London for proteomic analyses. We thank Professor Serge Mostowy for his advice. We also acknowledge the Imaging and Cytometry Platform for Infection Biology (LSHTM). This work was supported by the Biotechnology and Biological Sciences Research Council Institute Strategic Programme BB/R012504/1 constituent project BBS/E/F/000PR10349 to B.W.W.
Author information
Authors and Affiliations
Contributions
F.N. conceptualized, designed, performed experiments and wrote the first draft; F.N. and B. W.W. edited the final draft of the manuscript
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
. Communications Biology thanks Huan Lian and the other anonymous reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Tobias Goris. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nasher, F., Wren, B.W. A two-step actin-mediated strategy enables Campylobacter jejuni to promote mitochondrial aggregation and iron homeostasis, for intracellular survival and persistence. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09713-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09713-9