Abstract
High entropy alloys (HEAs), with their highly tunable compositions and unique physicochemical properties, hold significant promise as advanced materials for electrocatalysis. Since catalytic reactions primarily occur at the catalyst surface, optimizing surface properties is paramount for maximizing the performance of nanostructured HEAs (nHEAs) in electrocatalytic applications. This review highlights the critical role of surface optimization and explores strategies such as composition regulation, phase engineering, dimensional control, heterogeneity design, and defect engineering as crucial avenues for fine-tuning nHEAs surface properties. These advancements enable nHEAs to achieve superior activity and durability across diverse electrocatalytic reactions, including water electrolysis, carbon- and nitrogen-based conversions, and the conversion of small organic molecules. We also explore the challenges and future research directions, emphasizing the transformative potential of nHEAs in sustainable energy technologies through innovative surface design.

Similar content being viewed by others
Introduction
High-entropy alloys (HEAs), first conceptualized by Ye et al. and Cantor et al. in 20041,2, have emerged as a promising class of material owing to their unique compositional complexity and structural diversity. Notably, they have garnered significant attention for their exceptional catalytic properties. HEAs are characterized by the presence of five or more principal elements, each contributing between 5% and 35% of the total atomic concentration. This high compositional complexity endows HEAs with outstanding catalytic performance, primarily attributed to four key effects: the high-entropy effect, lattice distortion effect, slow diffusion effect, and cocktail effect3. The high-entropy effect stabilizes the alloy structure by maximizing configurational entropy and minimizing Gibbs free energy, thereby preventing phase separation4,5. The lattice distortion effect, arising from atomic size disparities, alters the electronic environment and optimizes the coordination of surface atoms, consequently enhancing catalytic activity6,7. The slow diffusion effect, attributable to the complex atomic interactions within HEAs, restricts atomic migration, thereby improving both thermal stability and mechanical strength8,9,10. Finally, the cocktail effect, stemming from the synergistic interactions between multiple elements, endows HEAs to exhibit tunable electronic structures, excellent oxidation resistance, and customizable catalytic behaviors11,12,13.
The heart of catalysis lies in the interaction between the reactant molecules and the surface of the catalyst14,15. This interaction is primarily governed by the catalyst’s surface properties. In electrocatalytic reactions, the catalyst-electrolyte interface constitutes the primary reaction zone, rendering surface engineering paramount for enhancing catalytic efficiency. Surface properties, including composition, crystal phases, morphology, and defects structures, exert a profound influence on adsorbate-surface interactions, thereby governing the reaction kinetics and thermodynamics and determining the catalyst’s efficiency and selectivity16,17,18,19,20,21. In addition to surface engineering, the integration of nanostructuring can also significantly enhances the catalytic performance of nanostructured HEAs (nHEAs). Nanostructuring leads to an increase in the specific surface area of the catalyst, thereby exposing a greater number of active sites and concurrently augmenting the activity of surface atoms22,23,24. This synergistic effect optimizes the efficiency and selectivity of catalytic reactions. Consequently, the judicious combination of surface engineering with nanostructuring presents a powerful strategy for achieving substantial advancements in nHEAs performance within the domain of electrocatalysis.
Despite the considerable promise of nHEAs, their intrinsic complexity poses significant scientific and technological challenges. The complex interactions between structural attributes, compositional variations, and resultant catalytic performance necessitates the development of sophisticated design strategies. Furthermore, achieving precise control over elemental distribution, atomic arrangement, and surface properties remains a formidable obstacle in the pursuit of optimized nHEAs for diverse catalytic applications. Consequently, surface engineering has become a critical approach for unlocking the full potential of nHEAs. In this review, we focus on key surface engineering strategies for nHEAs catalysts, including dimensional control, composition modulation, phase engineering, surface heterogeneity, and defect engineering. Beyond the primary emphasis on surface properties, this review also introduces advanced methodologies for surface characterization. Through strategic manipulation of these surface properties, the catalytic performance of nHEAs can be significantly enhanced, thereby facilitating their widespread application in diverse electrochemical systems, including water splitting, the conversion of carbon- and nitrogen-containing substrates, and the electrochemical oxidation of small organic molecules. Finally, we discuss future research directions for advancing nHEAs catalysts, identifying critical challenges and outlining promising avenues for unlocking their full potential in sustainable energy technologies.
HEAs as advanced catalysts: overcoming the drawbacks of traditional materials
For decades, traditional catalysts, encompassing metal-based catalysts, metal oxide catalysts, acids and bases, and carbon-based materials, have served as the mainstay of catalysis25,26. However, these conventional systems exhibit inherent limitations concerning tunability, activity, selectivity and stability. A primary constraint of traditional catalysts lies in their reliance on a single or a limited number of active metal components dispersed on a support material. This restricted number of active sites inherently limits the tunability required for complex electrocatalytic reactions. Moreover, conventional metal alloys often possess a constrained compositional range, dictated by factors such as disparities in crystal structure and atomic size. This limitation restricts the ability to precisely engineer their electronic structure to achieve optimal adsorption of reaction intermediates. Traditional catalysts frequently encounter scaling relations, which establish correlations between the adsorption energies of different reaction intermediates. This phenomenon can impede the simultaneous optimization of the catalyst for multiple reaction steps, often necessitating a trade-off where enhancing one step detrimentally affects another. Selectivity also poses a challenge for traditional catalysts, frequently resulting in the formation of undesirable byproducts due to active sites that lack unique specificity for a particular reaction pathway. Finally, traditional catalysts, particularly those based on single metals or simple alloys, are susceptible to deactivation mechanisms such as sintering, poisoning, leaching, and fatigue, thereby impacting their long-term performance.
Compared to traditional catalysts, HEAs present promising strategies to address the aforementioned limitations by capitalizing on their inherent compositional complexity and the resultant synergistic phenomena. The intricate elemental composition and diverse atomic configurations within HEAs generate a wide array of potential active sites. The cocktail effect specifically refers to the synergistic interactions among the multiple constituent elements in HEAs, often leading to catalytic performance that surpasses that of their individual metallic components. This inherent heterogeneity can enhance catalytic activity by providing a spectrum of adsorption energies that may optimize the binding of different reaction intermediates, potentially circumventing the scaling relations that constrain the performance of traditional catalysts. The diverse atomic environments present on HEAs surfaces can deviate from these scaling relationships, theoretically enabling the optimization of multiple reaction steps and overcoming activity limitations observed in single-metal catalysts. Furthermore, the tunability of these diverse active sites in HEAs offers the potential to enhance selectivity by providing specific binding motifs for desired intermediates while simultaneously hindering the formation of undesired byproducts. HEAs frequently exhibit improved thermal and chemical stability, largely attributed to the high mixing entropy effect which impedes atomic diffusion and phase segregation, thereby enhancing resistance to sintering and leaching. The sluggish diffusion effect further contributes to their robust structural integrity.
Evolutions in high-entropy concepts
High entropy alloys
The compositional definition of HEAs delineates them as single-phase alloys comprising a minimum of five principal elements, with each constituent element exhibiting an atomic percentage within the range of 5% to 35% of the alloy’s total composition. This definition is fundamentally rooted in the quantitative proportions of the constituent elements. Conversely, the entropy-based definition characterizes HEAs by a configurational entropy (Sconf) exceeding 1.5 R in their randomly mixed solid-solution state, where R denotes the ideal gas constant27. This criterion highlights the thermodynamic significance of a high degree of disorder, facilitated by elevated entropy, in promoting the stabilization of single-phase solid solution microstructures. An alloy is classified as an HEA if it satisfies the criteria of either the composition-based or the entropy-based definition. The designation of HEA is particularly pertinent when the configurational entropy or the deliberate engineering of single-phase solid solution microstructures constitutes a primary focus in the alloy design and application.
High-entropy materials (HEMs)
Building upon the conceptual foundations established in HEAs, entropy-driven design principles have been increasingly generalized to diverse material systems, leading to the emergence of high-entropy materials (HEMs). This diverse family now encompasses high-entropy oxides (HEOs), high-entropy sulfides (HESs), high-entropy nitrides (HENs), high-entropy metal-organic frameworks (HE-MOFs), high-entropy layered double hydroxides (HE-LDHs), high-entropy chalcogenide fluorides (HEPFs)28. While HEAs constitute a metallic subset within this broader category, unified by configurational entropy as a stabilizing factor, the atomic-level origins of this entropy diverge across these systems. In HEAs, multiple principal metallic elements randomly occupy a single crystal lattice, forming a homogeneous solid solution. In contrast, high-entropy compounds typically exhibit disorder confined to specific sublattices—most often cationic positions—within a structurally ordered anion or framework scaffold. This distinction renders HEAs and other HEMs conceptually and structurally distinct, even as they converge under the broader umbrella of entropy-enabled materials design.
Multi-principal element alloys (MPEAs) and complex solid solutions (CSS)
While the nascent stage of HEA research was significantly characterized by the pursuit of single-phase solid solutions stabilized by high configurational entropy, a more contemporary and encompassing perspective recognizes that the central tenet of this burgeoning field lies in the systematic exploration of the expansive alloy systems residing within the central regions of high-dimensional composition space. This broader objective transcends the initial emphasis on single-phase formation as the sole defining characteristic. To more neutrally and accurately represent this evolving research direction, the academic community has introduced alternative terminologies such as MPEAs, CSS, and, less frequently, baseless alloys29,30,31. These concepts deliberately move beyond any presuppositions regarding the magnitude of configurational entropy or the resulting phase constitution. Instead, they strategically focus on the inherent multiplicity of constituent elements within the alloy and the virtually limitless compositional permutations available within the multidimensional composition space. This shift in terminology and focus underscores a fundamental move away from a narrow definition based on entropy-driven single-phase formation towards a more holistic and exploratory investigation of complex, multi-component metallic systems. This approach opens up unprecedented opportunities for discovering novel materials with tailored properties that may not necessarily conform to the traditional single-phase paradigm.
The interrelation of HEAs, HEMs, CSSs, and MPEAs
HEAs can be understood as a subset within the broader categories of HEMs, CSSs, and MPEAs. MPEAs are defined by the presence of multiple (four or more) principal elements in significant concentrations, representing a broad compositional design space. Compare to medium-entropy alloys (MEAs), which typically exhibit moderate configurational entropy values (~1.0–1.5 R), MPEAs span a broader entropy range, encompassing both medium- and high-entropy regimes. HEAs, often characterized by a configurational entropy above 1.5 R, represent a high-entropy subset of MPEAs. In contrast, CSSs refer to a specific solid-state phase characterized by the dissolution of multiple elements within a single crystal lattice, resulting in a complex atomic arrangement. CSSs can occur in both conventional and multi-principal element alloys (including HEAs). While the high configurational entropy in HEAs often promotes the formation of single-phase CSSs, it is not a universal outcome, as some HEAs exhibit multiphase structures, including high-entropy intermetallics (HEIs). Indeed, the observation that not all compositions designated as HEAs exhibit stabilization solely through high configurational entropy underscores the limitations of this initial definition. This unique atomic ordering and local chemical environment, often extending beyond simple solid solution behavior, plays a crucial role in determining the functional properties of these materials, highlighting the significance of compositional complexity beyond the entropic contribution to phase stability. Conversely, CSSs are not exclusive to HEAs and can form in alloys with fewer principal elements. Consequently, although overlap exists (many HEAs are MPEAs and may exhibit CSS phases), these terms describe distinct aspects: HEA denotes composition, while CSS describes the resulting phase structure. The definitions can exhibit nuances within the evolving field.
The atomic-scale synergistic interactions in nHEAs
The atomic-scale synergistic interactions among the multiple constituent elements within nHEAs establish a fundamental structural basis for the generation of diversified catalytic active sites32. This unique mechanistic attribute, originating from the intricate electronic coupling between adjacent atomic species, facilitates the formation of continuously distributed adsorption energy spectra across the material surface. Through the systematic optimization of component engineering, the density of active sites exhibiting optimal adsorption energy for targeted reaction pathways can be directionally enhanced and precisely controlled. Empirical evidence derived from experimental investigations demonstrably indicates that the judicious selection of principal elements, coupled with the meticulous optimization of their stoichiometric ratios, can significantly augment the intrinsic catalytic activity while preserving the underlying nHEAs crystal structure. Consequently, the term nHEAs offers a more appropriate and intuitively descriptive framework, given the inherent complexity of the material system under investigation. Furthermore, considering the prevalence of atomic-scale chemical heterogeneity and nanoscale phase coexistence, the descriptor nHEAs more accurately encapsulates the intricate interplay between multi-element lattice occupancy and metastable phase formations. This refined terminology aligns with the evolving scientific consensus that nHEAs comprehensively encompasses both entropy-driven nHEAs and purposefully engineered multi-phase systems, thereby providing a more nuanced and accurate representation of the material’s structural and compositional attributes.
Key surface properties of nHEAs
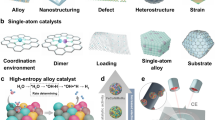
The surface properties of nHEAs play a pivotal role in determining their catalytic performance. Unlike conventional single-element or binary alloys, the inherent compositional complexity of nHEAs results in a heterogeneous elemental distribution, creating diverse active sites with distinct electronic structures. This heterogeneity promotes reactant adsorption and activation, while the deliberate selection and ratio of constituent elements enable precise tuning of electronic properties. In addition, the interplay of multiple crystal phases in nHEAs significantly influences surface reactivity and stability. Under reaction conditions, dynamic processes such as surface reconstruction, segregation, and alloy formation can further modify these characteristics, enhancing both catalytic activity and selectivity. In addition, the surface morphology—including particle size, shape, and porosity—critically impacts performance by providing a high surface area and abundant active sites. Structural features such as steps, kinks, and defects, as well as surface doping or oxide formation, further optimize catalytic performance. Various nanostructuring strategies that leverage these principles are illustrated in Fig. 1. Collectively, these intrinsic surface attributes not only underscore the potential of nHEAs as high-performance electrocatalysts but also lay the foundation for diverse surface engineering strategies.
Surface engineering strategies for designing nHEAs catalysts
HEAs goes to nanoscale
The catalytic activity of electrocatalysts is strongly influenced by the density of exposed active sites33. To maximize atomic utilization in nHEAs, reducing nanoparticle (NP) size has emerged as a key strategy due to the higher specific surface areas and enhanced atom utilization efficiency. As an illustration, a decrease in NP size from 11.7 nm to 3.9 nm correlates with a notable increase in atomic utilization, rising from 9.5% to 26%34. He et al. demonstrated this principle by synthesizing ultrasmall, size-tunable PtRuPdCoNi NPs ranging from 1.7 to 3.9 nm using a spatially confined strategy35. The 1.7 nm NPs exhibited superior activity and stability, attributed to their highest double-layer capacitance (indicating maximum active site exposure) and optimized noble metal utilization (Fig. 2a). Ultraviolet photoelectron spectroscopy (UPS) further revealed that smaller NPs possess lower work functions (WF), accelerating electron transfer from the catalyst surface to reaction intermediates and consequently enhancing reaction kinetics. Recent advancements in nHEAs synthesis have achieved sub-3 nm particle sizes through innovative methods: (i) ~2.68 nm PtFeCoNiMn HEA NPs synthesized via carbothermal shock pyrolysis36, where solvent polarity modulation and NP-support interactions played crucial roles in size control; (ii) ~2.3 nm PtRhPdIrRu HEA NPs synthesized through the microexplosion of Leidenfrost droplet37; (iii)~2 nm PtCuGaFeCo NPs fabricated using ethylene glycol and polyvinylpyrrolidone (PVP) as stabilizing agents38; (iv) ~2 nm PtFeCoNiCuZn NPs obtained via the hydrothermal decomposition of metal-organic frameworks (MOFs)39.
a TEM image and particle size distribution histogram of HEA-1.7 and scan rate dependence of the current densities in HEA. Reproduced with permission from ref. 35 (copyright Wiley, 2024). b HEA-PtPdIrRuAg ribbons. Reproduced with permission from ref. 40 (copyright, American Chemical Society, 2022). c HEA nanowires. Reproduced with permission from ref. 41 (copyright, Elsevier, 2023). Surface composition regulation. d NHEA@NHEA-Pd catalyst. g The line profiles. Reproduced with permission from ref. 48 (copyright Wiley, 2022). e PtPd HEA with PtPd-rich surface and the line profiles. Reproduced with permission from ref. 49 (copyright Elsevier, 2022). f PdAgSn/PtBi HEA NPs and elemental line-scanning results. Reproduced with permission from ref. 50 (copyright Wiley, 2023).
While ultrasmall 0D nanoparticles maximize active site exposure, one-dimensional (1D) and two-dimensional (2D) nHEAs exhibit unique structural features and remarkable electronic and physicochemical properties, including high aspect ratios, large specific surface areas, abundant surface unsaturated atoms, and enhanced electron mobility. For instance, Guo et al., synthesized sub-nanometer HEA-PtPdIrRuAg ribbons40 and HEA nanowires41 by leveraging anisotropic growth induced by Ag nanowire templates and a reduction-diffusion method, respectively (Fig. 2b, c). Similarly, Xu’s group developed a facile one-pot aqueous-phase approach to fabricate PdPtCuAgAu HEA nanowires with an average diameter of ~3.5 nm and lengths extending to hundreds of nanometers42. Additionally, advanced 3D morphologies such as hollow spheres43, nanocages44, and porous frameworks45 have gained significant attention due to their ability to enhance electrocatalytic performance. These structures offer a greater number of exposed active sites, shorter diffusion pathway for reactants, and improved utilization efficiency of noble metals.
Surface composition regulation
Surface composition engineering serves as a pivotal strategy for enhancing the catalytic performance of nHEAs. The strategic introduction of controlled compositional gradients via surface enrichment with noble metals (e.g., Pt, Pd) or catalytically active transition metals (e.g., Fe, Co) enables the selective localization of active sites at the near-surface region. This design effectively mitigates a critical limitation inherent in conventional single-phase HEAs, where a significant proportion of active sites remain embedded within the bulk matrix, rendering them inaccessible for catalytic processes. By concentrating active sites at the surface, this structure motif not only reduces metal consumption but also sustains or even enhances catalytic performance metrics compared to homogeneous solid-solution counterparts. Furthermore, this atomic-scale heterogeneity modulates local electronic structures and coordination environments, thereby optimizing adsorption energies of key reaction intermediates and facilitating efficient desorption of reaction products46,47.
For instance, Yao et al. developed an innovative surface-engineered HEA catalyst (NHEA@NHEA-Pd), featuring a noble metal-enriched shell supported by a stable non-noble metal core (Fig. 2d, g)48. The surface-engineered shell ensures a high dispersion and accessibility of Pd active sites, in contrast to conventional solid-solution or pure Pd systems where active sites are frequently embedded. Furthermore, the unique high-entropy coordination environment modulates the electronic structure of Pd, reducing the energy barrier for intermediate adsorption and surpassing the catalytic activity of pure Pd or binary Sn@Sn-Pd. Similarly, Yang et al. designed a PtPd-rich surface layer on a PtPdFeCoNiSnMn HEA, achieving a robust single-phase solid solution (Fig. 2e)49. Experimental evidences confirmed that the Pt and Pd atoms serve as the primary active sites for electrocatalytic reactions. Simultaneously, the presence of other constituent elements within the HEA matrix induced synergistic effects, which optimized the electronic environment and further boosted the catalytic activity of noble metal sites. Additionally, Guo et al. fabricated HEA NPs featuring a Pd-rich core and a Pt-dominant shell using an ambient low-temperature synthesis approach (Fig. 2f)50. Liu et al. designed Pd@PdPtRhIrRu core-shell nanocrystal by precisely controlling the precursor reduction kinetics51. Through precise control of surface composition, nHEAs can achieve superior catalytic performance and durability, making them highly promising for advanced electrocatalytic applications.
Surface heterogeneity
In contrast to surface composition regulation, which is characterized by the random distribution of surface metal elements to form a solid-solution phase, surface heterogeneity in nHEAs involves an uneven distribution of surface metal elements. Recent advancements have underscored the potential of surface heterostructure engineering in enhancing catalytic performance. Yin et al. developed a core-satellite HEA@Pt hybrid electrocatalyst comprising a PtRuFeCoNi HEA core with dendritic Pt structures grown on its surface (Fig. 3a)52. The uniform growth of Pt nanostructures along low-angle grain boundaries of HEA nanocrystals facilitated the formation of unique heterointerfaces with abundant active sites. The interfacial architecture, driven by the engineered electron redistribution at the heterointerface, further enhanced the catalytic activity. Similarly, Yao et al. designed a hybrid structure utilizing non-noble HEA NPs (FeCoNiCu) as a core to disperse and stabilize noble metals (Fig. 3b-c)53. The core was synthesized via rapid high-temperature shock on carbonized wood substrates, followed by galvanic displacement in H2PtCl6 to form HEA@Pt with Pt exposed on the surface.
a High-angle annular dark-field scanning transmission electron microscopy (HAADF STEM) image and corresponding energy-dispersive X-ray spectroscopy (EDS) mapping images of HEA@Pt. Reproduced with permission from ref. 52 (copyright Wiley, 2023). b The TEM images and element mapping of HEA@Pt. c HEA@Pt NP. Reproduced with permission from ref. 53 (copyright, Wiley, 2022). d Composition-structure relationship in HEAs with one constituent difference. Reproduced with permission from ref. 54 (copyright American Chemical Society, 2024). e FL-Ag/HEA NPs. Reproduced with permission from ref. 55 (copyright Springer Nature, 2024).
Hu et al. further demonstrated the tunability of local atomic ordering in nHEAs by incorporating a fifth element, such as Rh or Ru, into a PtPdFeCo substrate54. This modification facilitated a transition from a completely random alloy to one with localized single-element ordering. The degree of local ordering in RuPtPdFeCo was precisely controlled by varying the Ru concentration, enabling fine-tuning of heterostructure formation and localized clustering within the alloy (Fig. 3d). In contrast to the previous studies that primarily focused on creating localized heterogeneity by concentrating specific surface elements, our group has developed a surface entropy reduction method to selectively induce the leaching of components with weak affinities, such as Ag55. This process leads to the formation of few-atomic-layer Ag metals (FL-Ag) on the surface of FeCoNiVAgPd HEA (Fig. 3e). These leached FL-Ag components transcend the spatial limitations of conventional HEA architectures, enabling synergistic interactions with the underlying HEA substrate. They function as spatially distinct active sites, facilitating the adsorption and transformation of multiple reaction intermediates during complex catalytic processes.
Surface phase engineering
Surface phase engineering is a powerful strategy for enhancing nHEA catalysts by modulating surface composition and structure. Certain elements in nHEAs may segregate to the surface, forming alloys or oxides, which alters surface energy and electronic structure, ultimately improving catalytic activity56,57. By introducing phases such as oxides, hydroxides, or heterostructures, this approach increases active sites, facilitates charge transfer, and enhances stability under operating conditions. Liu et al. employed high-temperature treatment of nHEA in air to create a dual-phase catalyst featuring a high-entropy oxide (HEO) surface and a face-centered cubic (FCC)-structured core58. At 400 °C, transition metals diffused outward to form a disordered oxide layer with locally ordered lattices, while Pt remained in the core (Fig. 4a–c). The oxide layer acted as a diffusion barrier, preventing further oxidation and ensuring structural stability.
a Synthesis process, b EDS mappings and c corresponding at.% distribution of HEA NPs after in situ oxidation. Reproduced with permission from ref. 58 (copyright American Chemical Society, 2020). d Preparation process of HEA-NCFCC/NF@EA. e X-ray diffraction (XRD) patterns. Reproduced with permission from ref. 59 (copyright Elsevier, 2023). f Schematic illustration for the phase evolution of DP-B-HEA. g XRD patterns and in situ XRD patterns of DP-B-HEA/CNFs. Reproduced with permission from ref. 60 (copyright Royal Society of Chemistry, 2023). h The FeCoNiMnCr HEA-HEO catalyst. i XRD patterns. Reproduced with permission from ref. 62 (copyright Wiley, 2024). Surface defect engineering. j HAADF-STEM image of dendritic PdPtRhIrRu nanocrystal. Reproduced with permission from ref. 51 (copyright The American Association for the Advancement of Science, 2023). k, l High resolution transmission electron microscope (HRTEM) images of d-HEA NPs. m Schematic illustration of d-HEA lattice. Reproduced with permission from ref. 64 (copyright Wiley, 2024). n HRTEM image and integrated pixel intensity profile of the EE.300 s HEA film. Reproduced with permission from ref. 67 (copyright Royal Society of Chemistry, 2024).
Wang et al. synthesized a NiFeCuCoCe HEA on a nickel foam substrate (HEA-NCFCC/NF) using a one-step electrodeposition method59. Subsequently, multiple hydroxyl oxides were generated in situ on the HEA surface through a rapid electrochemical self-reconstruction process (HEA-NCFCC/NF@EA) (Fig. 4d, e). Ion exchange disrupted the crystal structure, creating defects and exposing active sites. Ce incorporation increased oxygen vacancies, boosting hydroxide adsorption and M-OOH species formation, which enhanced activity and stability in alkaline seawater. Zhu et al. employed a solid-state diffusion method to produce a dual-phase boron-doped FeCoNiCuPd HEA (DP-B-HEA)60. NiFe alloy NPs formed at 600 °C, followed by Co, Pd, and Cu diffusion at 900 °C. At 1000 °C, a dual-phase FCC structure emerged, where higher atomic packing density in FCC1 induced lattice distortion, improving catalytic performance (Fig. 4f, g). Maritza et al. synthesized a nanostructured FeNiCoCuMo HEA via mechanical alloying and hot-pressing, achieving a dual-phase structure comprising 99% FCC and 1% BCC with a crystallite size of ~5nm61. Zhang et al. used Cr-induced reconstruction to create HEA-HEO heterostructures (Fig. 4h, i)62. Density functional theory (DFT) calculations revealed that high-entropy oxide surfaces reduced 3d orbital pinning, providing active sites and promoting electron transfer, significantly enhancing catalytic performance and durability.
Surface defect engineering
Defect engineering represents a powerful strategy for enhancing catalytic performance by creating additional active sites and generating unsaturated coordination centers63. Capitalizing on their inherent compositional complexity, nHEAs intrinsically develop a diverse array of surface defects, including vacancies, dislocations, and grain boundaries, stemming from their thermodynamically metastable atomic configurations. These intrinsic defects originate from the disordered distribution of multiple metallic elements and significant lattice distortions, which collectively disrupt local coordination environments. Notably, nHEAs exhibit exceptional defect enrichment during synthesis. For example, Yang et al. synthesized dendritic PdPtRhIrRu HEA nanocrystals containing abundant step-edge atoms, localized lattice disorder, and vacancies (Fig. 4j) through kinetic-controlled growth51. These HEA nanocrystals showed superior hydrogen evolution reaction (HER) performance compared to both solid-solution and phase-separated PdPtRhIrRu counterparts. In situ X-ray absorption spectroscopy (XAS) and density functional theory (DFT) studies revealed that Ru sites act as catalytic centers, where adjacent defects weakened H* adsorption strength to enhance HER performance, while multi-element synergy optimizes electronic structures and entropy stabilization.
Similarly, Yu et al. developed a rapid synthesis strategy under boiling conditions to prepare disordered PtRhPdIrRu HEA NPs exhibiting nanotwinning, lattice distortions, and amorphous domains (Fig. 4k–m)64. The disordered structure induces a d-band shift in surface atoms, thereby optimizing adsorption energies for H* and OH* intermediates. Remarkably, extreme synthesis methods, such as Joule heating65, promote extensive nucleation and non-classical grain growth, thereby engineering nanocrystalline structures enriched in vacancies and dislocations. These investigations systematically elucidate the precise regulation of catalytic pathways by defect architectures in nHEAs establishing a paradigm for rational design of advanced catalysts.
Defects can also be intentionally engineered through the targeted removal of specific atomic species. Techniques such as acid leaching or the utilization of volatile sacrificial agents, such as zinc at elevated temperatures, facilitate the creation of controlled vacancies on the material’s surface. Huang et al. pioneered the synthesis of lattice-disordered nHEAs by employing laser irradiation to selectively remove sacrificial Zn components66. Similarly, Shen et al. utilized electrochemical treatment in acidic solutions on FeCoNiCrPt HEA thin films fabricated via pulsed laser deposition67. This process selectively removed surface atoms, resulting in the formation of vacancy-rich lattices and micro-strain fields (Fig. 4n). These modifications enabled the catalyst to achieve industrially viable water electrolysis performance with enhanced economic efficiency.
Surface characterization techniques for nHEAs
The catalytic performance of nHEAs is critically influenced by their complex surface structure. To facilitate the effective design of catalysts and the elucidation of reaction mechanisms, a comprehensive understanding of key surface characteristics is essential. These characteristics include morphology, elemental distribution, phase composition, surface chemical state, and defect configurations. Table 1 highlights the crucial methodologies employed for the investigation of nHEAs.
Imaging techniques represent some of the most direct and informative tools for characterizing nHEAs. Scanning electron microscopy (SEM) facilitates rapid morphological profiling, while transmission electron microscopy (TEM) and scanning TEM (STEM) enable atomic-resolution mapping of elemental distributions and chemical compositions. Importantly, electron energy loss spectroscopy (EELS) addresses the persistent challenge of light-element characterization by detecting characteristic energy-loss signals. Moreover, for applications demanding even higher resolution, aberration corrected STEM (AC-STEM) can be employed to achieve sub-angstrom resolution, permitting the observation of surface defects such as vacancies, surface step-edge atoms, and lattice distortions. However, these techniques primarily yield two-dimensional projections, which may not entirely capture the three-dimensional nature of the catalyst’s structure. Recent advancements in three-dimensional atomic distribution imaging, including atom probe tomography (APT) and atomistic electron tomography (AET), have significantly advanced catalyst characterization by providing a more comprehensive depiction of the atomic arrangement in nHEAs.
In addition to imaging techniques, various indirect characterization methods are also extensively used to complement the direct information provided by microscopy. Spectroscopic techniques, particularly those probing electronic transitions, play an essential role in revealing the chemical state and electronic structure of nHEAs. X-ray photoelectron spectroscopy (XPS) is widely employed for the determination of elemental composition and oxidation states of surface and near-surface regions. When combined with ion sputtering, XPS depth profiling enables the investigation of the chemical composition and oxidation state as a function of depth within the material. Other surface-sensitive techniques, such as Auger electron spectroscopy (AES) and low-energy ion scattering (LEIS), offer enhanced resolution in distinguishing oxidation states compared to XPS, providing detailed insights into the surface elemental composition and electronic structure. Expanding the scope of spectroscopic analysis further, XAS proves invaluable for investigating the electronic and geometric configurations surrounding target atoms. For example, Xia et al. elucidated the distribution characteristics of transition metal elements in NiCoFePtRh HEA NPs68. Their findings revealed a highly consistent coordination environments for Fe, Co, and Ni, with their atoms exhibiting a uniform and random distribution within the lattice. The interatomic distances of the noble metal elements Rh and Pt were significantly larger, suggesting that the presence of elements with larger atomic radius elements may induce local lattice distortions within the alloy system. To complement these spectroscopic techniques, diffraction-based methods, such as X-ray diffraction (XRD), neutron diffraction (ND), and electron diffraction (ED), provide valuable information about the atomic structure69. By converting total scattering data from XRD, ND, and ED into a pair distribution function (PDF), these techniques enable the extraction of structural details, including the structural order, nearest-neighbor distances, and coordination numbers of the atoms within the material.
In situ and operando characterization techniques are crucial for understanding the dynamic evolution of nHEAs catalysts under reaction conditions. In situ XRD monitors phase transitions and structural evolution in real time, enabling the identification of detrimental phases and the optimization of synthesis parameters. In situ TEM resolves atomic-scale geometric reconstruction of active sites and defect dynamics (e.g., twinning, dislocations) under operational conditions. Complementing these structural insights, operando XAS and in situ XPS probe the local coordination environment and surface chemical state evolution of active metal sites, respectively, thereby establishing a correlation between electronic structure to catalytic activity70. Xia et al. conducted pioneering operando XAS measurements to investigate the electrochemical behavior under varying applied potentials68. The work systematically acquired X-ray absorption near edge structure (XANES) spectra in fluorescence detection mode across multiple absorption edges (Rh K-edge, Ni K-edge, Fe K-edge, Pt L3-edge, and Co K-edge), encompassing both the initial state and different operational potentials. Notably, the XANES spectra features remained invariant throughout the potential application process, demonstrating exceptional structural stability of nHEAs during electrochemical reactions. In situ Raman and in situ Fourier transform infrared spectroscopy (FTIR) capture reaction intermediates, tracking critical reaction pathways.
Computational strategies for understanding nHEAs
Computational modeling plays a pivotal role in understanding reaction mechanisms, active-site configurations, and electronic interactions in nHEAs electrocatalysis. DFT has been widely employed to clarify catalytic reaction pathways, energy distributions of adsorbed intermediates, and the performance of different nHEAs compositions. For instance, DFT calculations have identified Cu sites in FeCoNiCuMn HEAs as primary active centers for HER and oxygen evolution reaction (OER) due to their lowest dissociation energy barrier and near-optimal hydrogen adsorption free energy17. These studies revealed that the electronegativity differences induce localized electron transfer, thereby modulating the reactivity of distinct metal sites.
The vast compositional space of nHEAs renders the acquisition of high-performance nHEAs with desired compositions through purely experimental methods challenging. Consequently, theoretical prediction is essential for identifying nHEAs catalysts with optimal catalytic performance. Zhuang et al. systematically utilized DFT to conduct a comprehensive investigation encompassing material composition design (special quasirandom structure (SQS) modeling and stability screening), active site analysis (adsorption energy and electronic structure), and reaction pathway optimization (free energy calculations and kinetic simulations)71. As a result, Ni0.3(FeCoCuPd)0.175 was identified as an efficient electrocatalytic nitrogen reduction reaction (eNRR) catalyst. Its superior performance is attributed to the electronic synergy of Fe-Co active sites, the Ni-induced upward shift of the d-band center, and the structural stability conferred by the high-entropy effect. Nonetheless, the high computational cost and time demands associated DFT limit its application in high-throughput screening of complex multi-component nHEAs systems. Furthermore, approximations inherent in exchange-correlation functionals and challenges in modeling non-equilibrium conditions introduce uncertainties when simulating nHEAs structures.
To address these limitations, machine learning (ML) has emerged as a powerful tool in nHEAs research. ML models trained on DFT data can rapidly predict catalytic properties, enabling the pre-screening of high-potential candidates prior to experimental synthesis. Key descriptors, such as d-band center, surface energy, and electronic density of states, are autonomously extracted to accelerate material discovery. For example, Yang et al. developed an ML workflow linking catalytic activity to structural parameters, achieving efficient predictions for multi-electron redox reactions72. Tomoya Shiota group further advanced this approach by introducing the Local Surface Energy (LSE) descriptor, which captures atomic-resolution surface reactivity73. By extracting LSE values from single-metal systems and applying linear regression models, this method enables high-throughput screening of nHEAs catalysts with DFT-level accuracy but significantly reduced computational complexity. Despite these advancements, ML efficacy remains constrained by data quality. Issues such as data scarcity, measurement errors, and inherent biases necessitate the development of standardized databases and physics-informed feature engineering.
The synergy between DFT and ML offers a transformative strategy for nHEAs electrocatalysis. ML models trained on DFT-generated data can prioritize promising candidates, drastically reducing computational costs, while DFT validation enhances the reliability of ML predictions. Recent studies highlight the potential of closed-loop optimization frameworks integrating DFT, ML, and experimental validation. For instance, Zhou et al. evaluated over 70 different catalysts utilizing large language models to design element combinations and curate a coordinated element library74. Subsequently, they employed micro-scale precursor printing and pulse high-temperature synthesis techniques to fabricate micro-scale HEAs. Using scanning electrochemical cell microscopy (SECCM), they screened for oxygen reduction reaction (ORR) activity and measured the relative current density to identify high-activity element combinations. In addition, multi-scale modeling approaches—combining quantum mechanics, molecular dynamics, and coarse-grained models—are gaining traction. These methods bridge atomic-scale interactions to macroscopic performance, enhancing both computational efficiency and mechanistic understanding of HEA structure-activity relationships.
Specific applications of nHEAs in electrocatalysis
The nHEAs offer unparalleled compositional flexibility, enabling precise modulation of their surface electronic structures and adsorption energies for reactants and intermediates. This inherent tunability facilitates the optimization of catalytic activity and selectivity across a diverse range of electrocatalytic reactions. Recent investigations have highlighted the potential of nHEAs in various electrocatalytic applications, such as water splitting, the conversion of carbon- and nitrogen-containing compounds, and electrochemical oxidation of small organic molecules (Table 2). The subsequent section delves into recent advancements that showcase nHEAs as promising electrocatalysts, with a particular emphasis on elucidating the mechanisms underlying their exceptional performance.
Hydrogen evolution reaction
Electrocatalytic water splitting is widely recognized as a promising avenue for addressing the escalating energy demands of modern society through the sustainable production of hydrogen75,76,77,78. However, conventional high-performance electrocatalysts often rely heavily on the utilization of noble metals, such as Pt, highlighting the critical need for the development of cost-effective yet efficient alternatives79. By leveraging surface engineering strategies, nHEAs can be optimized to achieve enhanced catalytic activity and stability while significantly reducing the reliance on expensive noble metals. For instance, Wang et al. meticulously designed a core-shell structure comprising a PtCoNiMoRh HEA core encapsulated by a Rh shell (PtCoNiMoRh@Rh) (Fig. 5a)80. This rationally designed catalyst demonstrated exceptional HER activity across a broad pH spectrum, encompassing acidic, neutral, and alkaline media, necessitating overpotentials of merely 9.1, 17.1, and 24.9 mV, respectively, to achieve 10 mA cm−2 (Fig. 5b). The observed enhancement in HER activity was attributed to the interaction between the HEA core and the Rh shell, resulting in a reduction of unoccupied Rh 4 d states, particularly the 4d5/2 orbital. Moreover, the synergistic orbital interactions among Rh-dyz, dx2, dxz, Pt-dxz, and Pt-dz2 orbitals with Ni-dyz, Ni-dz2, and Ni-dxz orbitals further enhanced the catalytic performance (Fig. 5c). Notably, the shell thickness played a crucial influence on modulating the adsorption energies of hydrogen intermediates (*H) and H2O, thereby fine-tuning the catalytic efficiency.
a PtCoNiMoRh@Rh. b Linear sweep voltammetry (LSV) curves with 90% IR correction in acidic, neutral, and alkaline electrolyte, respectively. c Rh-d orbitals states spectra of PtCoNiMoRh NPs. Reproduced with permission from ref. 80 (copyright Wiley, 2024). d LSV curves of different size HEA. e Correlation between HEA surface adsorption site (top, bridge, hollow) quantity/size and elemental composition. Reproduced with permission from ref. 81 (copyright Wiley, 2024) f PtMoPdRhNi NCs. g HER polarization curves. h Polarization curves before and after 5000 cycles. i XANES of Pt L3-edge. Reproduced with permission from ref. 82 (copyright Royal Society of Chemistry, 2023). OER. j Nanoporous HEA5IrB. k XPS spectra of HEA5, HEA5Ir, HEA5B and HEA5IrB and l OER polarization curves. Reproduced with permission from ref. 89 (copyright Royal Society of Chemistry, 2024).
Particle size engineering has emerged as a strategic manipulation to enhance catalytic active site density. Zhao et al. synthesized PdPtRuRhAu HEAs ranging from 2.81 to 7.74 nm81. Electrochemical evaluations revealed that the nHEAs with an average size of 3.14 nm (nHEA-3.14) exhibited the optimal catalytic performance (Fig. 5d). DFT calculations further elucidated the underlying mechanism, demonstrating that the catalytic enhancement originated from Pd-Au bridge adsorption sites, which played a dominant role in regulating intermediate binding energy. While Ru and Rh demonstrated stronger hydrogen adsorption capabilities. nHEA-3.14 possessed the highest density of these beneficial Pd-Au active sites (Fig. 5e), contributing to its superior catalytic activity.
While size-controlled synthesis enhances active site density, synergistic interactions among the constituent metallic components can further unlock catalytic potential through the modulation of electronic structures. For instance, Wang et al. developed PtMoPdRhNi nanocrystals (NCs) encapsulated by radially arranged nanosheets (Fig. 5f), resulting in exceptional HER performance in alkaline media82. This catalyst exhibited a remarkably low overpotential of merely 9.7 mV to reach a current density of 10 mA cm−2 in 1.0 M KOH, along with exceptional stability evidenced by negligible activity decay after 5000 cycles (Fig. 5g, h). The enhanced activity was attributed to the optimized utilization of Pt and strong d-d electron interactions among the constituent metals (Fig. 5i).
Oxygen evolution reaction
The OER is a key half-reaction for electrocatalytic water splitting, a pivotal technology for the sustainable production of hydrogen83,84,85,86. However, the OER mechanism is inherently complex, characterized by a sluggish four-step proton-coupled electron transfer process. This intricate pathway presents significant thermodynamic challenges, including substantial energy barriers, necessitating the application of high overpotentials. Consequently, the development of efficient, durable, and cost-effective electrocatalysts for OER remains a crucial research objective. Cheng et al. demonstrated the significant potential of nHEAs by synthesizing sub-2 nm nanoparticles with an Ir-rich oxide shell (IrRuCoNiMo@IrRuNiMoOx)87. This unique architecture exhibited an exceptionally low overpotential of 243 mV at 10 mA cm−2, significantly outperforming both IrO2 and Ir-black. The Ir-rich shell effectively suppressed the dissolution of transition metals, enhancing the long-term stability of the catalyst. Moreover, DFT calculations revealed that the synergistic interaction among the core elements optimized the binding energies of oxygen intermediates, thereby significantly enhancing OER activity.
Amorphization of nHEAs through the incorporation of elements with significantly larger atomic radii offers a promising avenue for enhancing catalytic performance. Feng et al. prepared amorphous FeCoNiCrMo HEA NPs by introducing Mo88, achieving an ultralow overpotential of 294 mV at 100 mA cm−2. The amorphous structure, with its random atomic arrangement, provides a diverse array of active sites and facilitates efficient electron transfer, outperforming its crystalline counterpart. Beyond structural amorphization, the strategic incorporation of light elements like boron introduces dual catalytic enhancement pathways: valence state elevation of transition metals to activate MOOH intermediates and surface electron density modulation to optimize reaction kinetics. Fujita et al. synthesized nanoporous HEA, incorporating B into CoCuFeMoNiIr catalysts (HEA5IrB) (Fig. 5j)89. This modification resulted in the oxidation of transition metals to higher valence states and a concomitant increase in electron density at the Ir-B surface (Fig. 5k). Consequently, the modified catalyst exhibited an overpotential of 277 mV at 10 mA cm−2, while simultaneously demonstrating improved durability (Fig. 5l).
Oxygen reduction reaction
The ORR plays a critical role in proton exchange membrane fuel cells, serving as a key half-reaction in energy conversion process90,91,92,93. Despite Pt being the most effective catalyst for ORR, its scarcity, high cost, and limited reserves necessitate the development of alternative catalysts that can provide comparable or even superior performance94. Gao’s group significantly advanced the field of nHEAs catalysis by developing Pt-based nHEAs mesoporous nanotubes (Fig. 6a)95, exhibited exceptional ORR activity, achieving a mass activity of 1.94 A mgPt−1 at 0.9 V versus reversible hydrogen electrode (RHE) in alkaline media (Fig. 6b). The unique mesoporous nanotube structure not only minimized Pt consumption but also maximized Pt utilization. The pronounced lattice distortions and abundant defect sites on the surfaces introduce unsaturated, highly active catalytic sites. XPS analysis revealed electron transfer from Fe and Cu to Pt, Pd, Ru, and Ir during ORR process, leading to electron enrichment of Pt and Pd and consequently accelerating the ORR kinetics (Fig. 6c). Furthermore, DFT calculations corroborated these findings, suggesting that synergistic interactions among the multiple metals within the nHEAs optimized the adsorption energies of oxygen intermediates, further enhancing catalytic performance (Fig. 6d).
a HAADF-STEM image of HEA mNTs. b LSV curves recorded in O2-bubbling 0.1 M KOH electrolyte. c The relationship between binding energy of Pt0 and specific activity. d ΔEO values of various HEAs. Reproduced with permission from ref. 95 (copyright Elsevier, 2023). e N-Pt/HEA/C. f ORR polarization curves. g Operando XANES measurements. h Charge density difference of surfaces. i ORR polarization curves tested at the beginning-of-life and after various potential cycles. Reproduced with permission from ref. 96 (copyright American Chemical Society, 2024). CO2RR. j AuAgPtPdCu NP. k TEM image. l i-t curves at different potentials (V vs AgCl). m Bar diagram of products. n Free-energy diagram. Reproduced with permission from ref. 102 (copyright American Chemical Society, 2020).
In another study, Sasaki et al. demonstrated the synergistic enhancement of ORR activity by incorporating N-doping into nHEAs catalysts (Fig. 6e)96. They developed a novel nHEAs catalyst architecture comprising a Pt-rich shell encapsulating an N-doped PtCoFeNiCu core, supported on carbon (N-Pt/HEA/C). This catalyst exhibits a mass activity of 1.34 A mgPt−1 at 0.9 V and exceptional durability with 79.1% retention of its initial mass activity after 30,000 cycles (Fig. 6f, g). Operando XAS and theoretical calculations revealed that the N-doping effectively optimized the adsorption energies of reaction intermediates while simultaneously improved the corrosion resistance of the catalyst, leading to significant enhancements in both long-term stability and overall performance (Fig. 6h, i).
Carbon dioxide reduction reaction
The electrochemical CO2 reduction reaction (CO2RR) offers a sustainable and versatile approach to mitigating climate change while simultaneously addressing the escalating growing demand for renewable fuels and value-added chemicals97,98,99,100,101. Biswas et al. demonstrate the remarkable potential of a nanocrystalline AuAgPtPdCu HEA (Fig. 6j, k) for CO2RR102, achieving nearly 100% Faradic efficiency (FE) for gaseous products at a low potential of -0.3 V vs RHE. The AuAgPtPdCu catalyst primarily produced CO, CH4, C2H4, and H2, with Cu identified as the dominant active site for the reaction (Fig. 6l, m). Notably, the other alloying elements, while not directly contributing to catalytic activity, were found to enhance performance through synergistic interactions, optimizing the electronic and geometric environment of the active sites. DFT calculations demonstrated that the nHEA surface exhibits a lower theoretical limiting potential for CO2RR compared to pure Cu, highlighting its inherent thermodynamic advantages. This enhancement is primarily attributed to the modified adsorption characteristics of key reaction intermediates: destabilization of the *OCH3 intermediate and stabilization of the *O intermediate. These modifications effectively lower the energy barriers for product formation, enabling the nHEAs catalyst to outperform the Cu (111) surface (Fig. 6n). The findings highlight the potential of nHEAs to surpass conventional monometallic catalysts in CO2RR by synergistically combining the intrinsic catalytic contributions of specific elements, such as Cu, with the emergent properties arising from alloying. Liu et al. developed PdCuAuAgBiIn HEA aerogels, achieving nearly 100% FE for C1 products from −0.7 to −1.1 V vs. RHE and a maximum FE of 98.1% for formic acid45. The exceptional CO2RR performance is attributed to strong intermetallic interactions and surface unsaturated sites, which optimize HCOO* intermediate adsorption and desorption, enhancing HCOOH production.
Ammonia electrosynthesis
Ammonia remains indispensable in industrial chemistry, yet its conventional production via the Haber-Bosch process is both energy-intensive and ecologically unsustainable, necessitating the adoption of electrocatalytic synthesis strategies103,104,105,106. Zhang et al. firstly demonstrated the application of nHEAs for nitrogen reduction reaction (NRR) by achieving a FE of 38.5% with 16 nm HEA RuFeCoNiCu NPs (Fig. 7a, b)107. Their study attributed the excellent NRR performance to a synergistic effect among multiple active sites. Specifically, the Fe site, surrounded by other elements, optimize N2 adsorption, while Ru-Ni and Co-Cu sites facilitated surface hydrogenation. This configuration promoted N2 hydrogenation while suppressing the undesirable H* combination (Fig. 7c).
a TEM image of RuFeCoNiCu NPs. b NH3 yields and FEs in 0.1 M KOH. c Schematic illustration of a possible mechanism. Reproduced with permission from ref. 107 (copyright Wiley, 2020). d LSV curves in 0.5 M K2SO4 with and without 0.1 M KNO3. e Comparison of the NH3 production rates. f Relay catalysis mechanism of NO3RR over FL-Ag/HEA. Reproduced with permission from ref. 55 (copyright Springer Nature, 2024). Electrochemical conversion of small organic molecules. g PdPtPbSnNi HEA NWs. h Cyclic voltammetry (CV) curves and i cycling stability in 1 M KOH + 1 M ethanol solution. j CV curves and k cycling stability in 1 M KOH + 1 M methanol solution. Reproduced with permission from ref. 113 (copyright Elsevier, 2024). l FOL conversion rate of different catalysts and different temperatures. m The adsorption energy of furfural on the NiCoCuZnFe/C-800. n The hydrogenation mechanism of furfural over NiCoCuZnFe/C-800. Reproduced with permission from ref. 117 (copyright Elsevier, 2023).
The nitrate reduction reaction (NO3RR) offers a promising alternative for ammonia production, leveraging the lower bond energy in nitrate compared to the strong triple bond in molecular nitrogen. This inherent advantage facilitates milder reaction conditions and higher energy efficiency. nHEAs, with their versatile multi-element compositions and inherent structural complexity, have demonstrated significant potential in catalyzing multistep tandem reactions, such as the NO3RR, due to their diverse and tunable active sites.
Expanding upon this concept, our group developed an innovative strategy by introducing Ag—a metal with weak interatomic affinity—onto the surface of an HEA to form few-atom-layer silverenes (FL-Ag/HEA)55. These interfacial FL-Ag structures interacted synergistically with adjacent HEA metal sites, creating unique active-site motifs that circumvent traditional scaling relationships. This structural configuration enabled an efficient cascade conversion of nitrate into ammonia (Fig. 7d-e). In situ FTIR and DFT calculations unraveled a relay catalysis mechanism at FL-Ag/HEA interfaces. Specifically, *NO3 was reduced to *NO on FL-Ag, while *NO intermediates migrated to the neighboring Pd sites at the interface for further hydrogenation to NH3 (Fig. 7f). This tandem mechanism exemplifies how tailored nHEAs architectures can optimize reaction pathways and improve catalytic efficiency in NO3RR applications.
Electrochemical conversion of small organic molecules
The sluggish kinetics of the OER often hinder the efficiency of electrosynthesis. Replacing OER with alcohol oxidation reactions (AOR) using renewable, biomass-derived alcohols offers a promising alternative108,109,110,111. AOR exhibits lower overpotentials, thereby reducing energy demands and simultaneously generating high-value chemicals thus enhancing both the efficiency and sustainability of electrosynthesis112.
Du et al. designed PdPtPbSnNi HEA nanowires exhibiting outstanding catalytic performance towards ethanol and methanol oxidation reactions (Fig. 7g)113, achieving mass activities of 4.36 and 4.34 A mgPd+Pt−1, respectively (Fig. 7h, i). The catalyst showed excellent stability, maintaining activities of 1.18 A mg−1 and 1.21 A mg−1 after 500 cycles, significantly surpassing commercial Pt/C and Pd/C catalysts (Fig. 7j, k). Moreover, the nHEAs also retained their structural integrity even after long-term chronoamperometry tests. The excellent performance is attributed to several key factors: The multi-element composition of the nHEAs provides a near-continuous distribution of binding energies, facilitating the multi-electron transfer reactions. The presence of Pd and Pt intrinsically enhance the activity. The incorporation of oxyphilic elements, such as Pb, Sn, and Ni effectively promote oxidation of adsorbed CO intermediates, a crucial step in alcohol oxidation. The nHEAs structure, combined with the 1D nanowire morphology, ensures excellent stability and durability. This work provides valuable insights into the controlled synthesis and growth of high-entropy PdPtSnPbNi nanowires, paving the way for the development of highly active and durable catalysts for alcohol electrooxidation.
Wen’s group developed CoNiCuMnMo HEA demonstrating remarkable electrocatalytic performance for the glycerol oxidation reaction (GOR). This HEA achieved a current density of 10 mA cm−2 at a low overpotential of 1.25 V vs RHE112. Additionally, it exhibited exceptional selectivity for formate production, maintaining a FE exceeding 90% over a broad potential range (1.27–1.47 V vs RHE). DFT calculations revealed that the catalytic activity stems from Mo sites coordinated with Mn, Mo, and Ni, highlighting the crucial role of synergistic atomic configurations in enhancing the GOR performance. Zhu et al. synthesized a high-performance L10-(PtIr)(FeMoBi) high-entropy intermetallic catalyst using a lattice compensation strategy to counter entropy reduction114. This approach enabled a homogeneous distribution of active Pt and Fe sites, enhancing catalytic efficiency. The catalyst achieved a FE of 95% and a mass activity of 5.2 A mgPt−1 for glycolic acid production via ethylene glycol oxidation.
The electrocatalytic oxidation of 5-hydroxymethylfurfural (HMFOR) to 2,5-furandicarboxylic acid (FDCA) is of paramount importance for sustainable biomass valorization, given FDCA’s potential to substitute fossil-derived plastics. Yin et al. developed AuCuAgPdNi HEA NWs via a one-pot method, achieving 95.5% FDCA yield and 98.4% selectivity at 1.60 V vs RHE115. The exceptional performance stems from defect-induced strain, enhancing charge transfer and mass transport, and high surface area providing abundant active sites. Additionally, the d–d orbital coupling and self-complementary effects among the five elements optimize electronic structure and facilitate surface electron exchange. DFT confirms strengthened d–d interactions lower energy barriers, enabling efficient HMFOR. Zhu et al. synthesized a sub-10 nm CoNiMnMoPd HEA catalyst that achieved 92.5% FE for FDCA production with 89.5% HMF conversion and 95.8% selectivity116. The enhanced performance stems from the unique multielement environment, where Ni sites stabilized in a high-valence state in HEA selectively adsorbed HMF, while non-Ni neighboring sites promoted dehydrogenation. This spatial synergy, combined with optimized electronic effects, reduced the energy barrier of the rate-determining step from 0.770 eV (monometallic Ni) to 0.567 eV. While oxidation pathways of furfural have been widely investigated, its reductive transformation through catalytic hydrogenation demonstrates equally significant industrial potential.
Furfuryl alcohol (FOL), produced via catalytic hydrogenation of furfural, has emerged as a valuable intermediate with broad application prospects in industrial processes. Xu et al. synthesized NiCoCuZnFe/C-800, which achieved an FOL conversion rate of 87.32% with 100% selectivity at 90 °C (Fig. 7l)117. DFT calculations indicate that Zn and Fe sites serve as the preferred adsorption centers for the –CHO functional group of furfurals (Fig. 7m). In contrast, in ultra-dilute HEA systems, the aggregation and leaching of these oxyphilic metals weaken the catalyst’s ability to adsorb furfural. Additionally, the partial overlap between the three-dimensional energy band of Cu atoms and the antibonding orbitals of the furan ring induces a strong repulsive interaction between furfural and the Cu(111) surface. This electronic interaction favors a vertical adsorption orientation of furfural on ultra-dilute nHEAs, minimizing the interaction between the furan ring and the metal surface (Fig. 7n). Such an adsorption configuration enhances the selectivity for furfuryl alcohol, thereby improving catalytic performance.
Outlook
In this review, we delve into the rational surface design strategies for nHEAs electrocatalysts, including dimensional control, composition regulation, phase engineering, surface heterogeneity manipulation and defect engineering. Besides, we introduce a series of advanced surface characterizations. Subsequently, we present a comprehensive overview of the applications of nHEAs in various electrocatalytic processes. The underlying reasons for their enhanced electrocatalytic performance, including the associated reaction mechanisms, are thoroughly analyzed. Finally, we offer perspectives on the future development trends and potential research directions for nHEAs electrocatalysts.
Achieving precise control over the synthesis of nHEAs remains a significant challenge. Current methodologies often yield nanoparticles exhibiting limited control over elemental distribution, morphology, or architecture, thereby restricting their functional versatility. Future research should prioritize the development of strategies enabling meticulous control over elemental composition, size, morphology, and architecture (e.g., core-shell, hierarchical, or heterostructure) to facilitate tailored phase engineering. Such structural precision creates diverse active sites that simultaneously improve reaction selectivity and stability. The synergistic effects derived from these sites additionally confer multifunctional properties to the catalyst system.
A critical gap persists in the mechanistic understanding of complex reactions such as CO2RR, eNRR, NO3RR, and C-N coupling, where multi-step pathways and diverse intermediates demand precise control over adsorption energetics. Traditional trial-and-error approaches prove insufficient for deciphering these processes due to the stochastic distribution of surface metal atoms. Recent advances in in situ and operando characterization techniques, such as XAS, electrochemical TEM, and Raman spectroscopy, now enable real-time monitoring of dynamic surface reconstruction, intermediate adsorption, and electronic state evolution. Furthermore, the integration of these advanced characterization methods with machine learning-driven microstructure optimization—through active learning loops combining high-throughput DFT calculations and experimental validation—offers a promising avenue for navigating the expansive nHEAs compositional space and deciphering structure-activity relationships. However, the efficacy of current machine learning approaches depends on the availability of high-quality, standardized datasets, a bottleneck stemming from inconsistent synthesis protocols and characterization methods across different studies. Addressing these limitations is paramount for the advancement of nHEAs research.
While nHEAs exhibit the potential to surpass traditional catalysts in multifunctionality and stability, their practical implementation requires a balanced assessment. Conventional catalysts such as Pt/C and IrO2 still demonstrate superior simplicity, reproducibility, and industrial maturity, notwithstanding the tunable electronic properties and synergistic active sites offered by nHEAs. Consequently, future research should focus on the precision synthesis for controlled atomic heterogeneity, integrated operando analysis coupled with machine learning for mechanistic insights, and the development of scalable, sustainable synthesis routes. By addressing these limitations, nHEAs could redefine the frontiers of catalysis and offer innovative solutions to pressing energy and environmental challenges.
Change history
14 November 2025
In the Acknowledgements section of this article the project number relating to Basic Research Program of Jiangsu was incorrectly given as BK2022030167 and should have been BK20220156. The original article has been corrected.
References
Yeh, J. W. et al. Nanostructured high-entropy alloys with multiple principal elements: Novel alloy design concepts and outcomes. Adv. Eng. Mater. 6, 299–303 (2004).
Cantor, B., Chang, I. T. H., Knight, P. & Vincent, A. J. B. Microstructural development in equiatomic multicomponent alloys. Mater. Sci. Eng. A 375-377, 213–218 (2004). This paper introduces the concept of high-entropy alloys for the first time.
Hsu, W. L., Tsai, C. W., Yeh, A. C. & Yeh, J. W. Clarifying the four core effects of high-entropy materials. Nat. Rev. Chem. 8, 471–485 (2024).
Xin, Y. et al. High-entropy alloys as a platform for catalysis: Progress, challenges, and opportunities. ACS Catal. 10, 11280–11306 (2020).
Chen, Y. et al. Synthesis of monodisperse high entropy alloy nanocatalysts from core@shell nanoparticles. Nanoscale Horiz. 6, 231–237 (2021).
Wang, D. et al. Tailoring lattice strain in ultra-fine high-entropy alloys for active and stable methanol oxidation. Sci. Chi. Mater. 64, 2454–2466 (2021).
Jiang, Y. et al. Stimulating electron delocalization of lanthanide elements through high-entropy confinement to promote electrocatalytic water splitting. ACS Nano 18, 19137–19149 (2024).
Yu, Y. et al. High-entropy alloy nanoparticles as a promising electrocatalyst to enhance activity and durability for oxygen reduction. Nano Res. 15, 7868–7876 (2022).
Wang, R. et al. Effect of lattice distortion on the diffusion behavior of high-entropy alloys. J. Alloys Compd. 825, 154099 (2020).
Huang, X. et al. Noble-metal-based high-entropy-alloy nanoparticles for electrocatalysis. J. Energy Chem. 68, 721–751 (2022).
Zhang, L., Cai, W., Bao, N. & Yang, H. Implanting an electron donor to enlarge the d–p hybridization of high-entropy (oxy)hydroxide: A novel design to boost oxygen evolution. Adv. Mater. 34, 2110511 (2022).
Ma, Y. et al. High-entropy energy materials: challenges and new opportunities. Energy Environ. Sci. 14, 2883–2905 (2021).
Zhang, Q. et al. High-entropy alloys in water electrolysis: Recent advances, fundamentals, and challenges. Sci. Chi. Mater. 66, 1681–1701 (2023).
Cheng, W., Sun, C., Liu, W. & Wang, Z. High-entropy alloy PtCuNiCoMn nanoparticles on rGO for electrooxidation of methanol and formic acid. Langmuir 40, 2343–2351 (2024).
Kang, N. et al. Electrochemical performance of in-situ electrodeposited spinel-structured high entropy oxides/NiFeCuMoMn-high entropy alloy multiphase electrocatalysts for seawater electrolysis. Electrochim. Acta 506, 144931 (2024).
Hao, J. et al. Unraveling the electronegativity-dominated intermediate adsorption on high-entropy alloy electrocatalysts. Nat. Commun. 13, 2662 (2022).
Zhu, H. et al. A high-entropy atomic environment converts inactive to active sites for electrocatalysis. Energy Environ. Sci. 16, 619–628 (2023).
Zhu, H. et al. High-entropy alloy stabilized active Ir for highly efficient acidic oxygen evolution. Chem. Eng. J. 431, 133251 (2022).
Wang, S. et al. Engineering multiple nano-twinned high entropy alloy electrocatalysts toward efficient water electrolysis. Appl. Catal. B: Environ. Energy 363, 124791 (2025).
Chen, S., Duan, J., Ran, J. & Qiao, S.-Z. Paper-based N-doped carbon films for enhanced oxygen evolution electrocatalysis. Adv. Sci. 2, 1400015 (2015).
Monestel, H. G. R. et al. Robust MOF-253-derived N-doped carbon confinement of Pt single nanocrystal electrocatalysts for oxygen evolution reaction. Chin. J. Catal. 41, 839–846 (2020).
Al Zoubi, W., Putri, R. A. K., Abukhadra, M. R. & Ko, Y. G. Recent experimental and theoretical advances in the design and science of high-entropy alloy nanoparticles. Nano Energy 110, 108362 (2023).
Luo, J. et al. Progressive fabrication of a Pt-based high-entropy-alloy catalyst toward highly efficient propane dehydrogenation. Angew. Chem. Int. Ed. 64, e202419093 (2024).
Shao, M., Peles, A. & Shoemaker, K. Electrocatalysis on platinum nanoparticles: particle size effect on oxygen reduction reaction activity. Nano Lett. 11, 3714–3719 (2011).
He, Q. F., Ding, Z. Y., Ye, Y. F. & Yang, Y. Design of high-entropy alloy: a perspective from nonideal mixing. JOM 69, 2092–2098 (2017).
Ye, Y. F., Wang, Q., Lu, J., Liu, C. T. & Yang, Y. High-entropy alloy: challenges and prospects. Mater. Today 19, 349–362 (2016).
Chang, X., Zeng, M., Liu, K. & Fu, L. Phase engineering of high-entropy alloys. Adv. Mater. 32, 1907226 (2020).
Lee, J., Seo, J. H., Gao, B. & Jang, H. W. Transition metal-based high-entropy naterials for catalysis. MetalMat 2, e31 (2025).
Batchelor, T. A. A. et al. Complex-solid-solution electrocatalyst discovery by computational prediction and high-throughput experimentation. Angew. Chem. Int. Ed. 60, 6932–6937 (2021).
Singh, P. et al. Design of high-strength refractory complex solid-solution alloys. npj Comput. Mater. 4, 16 (2018).
Miracle, D. B. & Senkov, O. N. A critical review of high entropy alloys and related concepts. Acta Mater. 122, 448–511 (2017). This study systematically compares existing concepts related to high-entropy alloys.
Lee, S. A., Bu, J., Lee, J. & Jang, H. W. High-entropy nanomaterials for advanced electrocatalysis. Small Sci. 3, 2200109 (2023).
Wang, H. et al. Disentangling the size-dependent geometric and electronic effects of palladium nanocatalysts beyond selectivity. Sci. Adv. 5, eaat6413 (2019).
Ai, Y. et al. Ultra-small high-entropy alloy nanoparticles: Efficient nanozyme for enhancing tumor photothermal therapy. Adv. Mater. 35, 2302335 (2023).
Cai, H. et al. Size-adjustable high-entropy alloy nanoparticles as an efficient platform for electrocatalysis. Angew. Chem. Int. Ed. 64, e202423765 (2025).
Huang, Z. et al. Microenvironment regulation to synthesize sub-3 nm Pt-based high-entropy alloy nanoparticles enabling compressed lattice to boost electrocatalysis. Appl. Catal. B: Environ. Energy 363, 124775 (2025).
Wu, Z. et al. Explosive leidenfrost-droplet-mediated synthesis of monodispersed high-entropy-alloy nanoparticles for electrocatalysis. Nano Lett. 24, 7158–7165 (2024).
Kuang, H., Xu, Z., Tan, X., Yu, K. & Chen, C. Highly dispersed ultrasmall high-entropy alloys nanoparticles as efficient electrocatalysts for oxygen reduction in acidic media. Small 20, 2308421 (2024).
Chen, T. et al. An ultrasmall ordered high-entropy intermetallic with multiple active sites for the oxygen reduction reaction. J. Am. Chem. Soc. 146, 1174–1184 (2024).
Tao, L. et al. A general synthetic method for high-entropy alloy subnanometer ribbons. J. Am. Chem. Soc. 144, 10582–10590 (2022).
Sun, Y. et al. A general approach to high-entropy metallic nanowire electrocatalysts. Matter 6, 193–205 (2023).
Fan, D. et al. Engineering high-entropy alloy nanowires network for alcohol electrooxidation. J. Colloid Interface Sci. 625, 1012–1021 (2022).
Wang, X. et al. Continuous synthesis of hollow high-entropy nanoparticles for energy and catalysis applications. Adv. Mater. 32, 2002853 (2020).
Su, K. et al. High-entropy alloy nanocages with highly ordered {100} facets and ultrathin features for water splitting in an acidic medium. J. Mater. Chem. A 12, 16043–16051 (2024).
Li, H. et al. High-Entropy Alloy Aerogels: A New Platform for Carbon Dioxide Reduction. Advanced Materials 35, 2209242 (2023).
Hsiao, Y.-C. et al. A library of seed@high-entropy-alloy core–shell nanocrystals with controlled facets for catalysis. Adv. Mater. 37, 2411464 (2024).
Qiu, Z. et al. High-entropy Ag−Ru-based electrocatalysts with dual-active-center for highly stable ultra-low-temperature zinc-air batteries. Angew. Chem. Int. Ed. 64, e202415216 (2024).
Zeng, K. et al. Surface-decorated high-entropy alloy catalysts with significantly boosted activity and stability. Adv. Funct. Mater. 32, 2204643 (2022).
Chang, J. et al. Rational design of septenary high-entropy alloy for direct ethanol fuel cells. Joule 7, 587–602 (2023).
Lao, X. et al. Pd-enriched-core/Pt-enriched-shell high-entropy alloy with face-centred cubic structure for C1 and C2 alcohol oxidation. Angew. Chem. Int. Ed. 62, e202304510 (2023). This study presents an innovative strategy for synthesizing core-shell high-entropy alloys.
Liu, Y.-H. et al. Toward controllable and predictable synthesis of high-entropy alloy nanocrystals. Sci. Adv. 9, eadf9931 (2023). This paper leads to a paradigm shift in the design of HEA nanocrystals, pushing away from the trial-and-error approach.
Zhang, P. et al. New conceptual catalyst on spatial high-entropy alloy heterostructures for high-performance Li-O2 batteries. Small 19, 2206742 (2023).
Shi, W. et al. High-entropy alloy stabilized and activated Pt clusters for highly efficient electrocatalysis. SusMat 2, 186–196 (2022).
Huang, Z. et al. Tailoring local chemical ordering via elemental tuning in high-entropy alloys. J. Am. Chem. Soc. 146, 2167–2173 (2024).
Hao, J. et al. Integrating few-atom layer metal on high-entropy alloys to catalyze nitrate reduction in tandem. Nat. Commun. 15, 9020 (2024). This paper firstly reports few-atomic-layer-thick heterostructures for nitrate reduction reaction.
Jiang, H., He, Q., Zhang, Y. & Song, L. Structural self-reconstruction of catalysts in electrocatalysis. Acc. Chem. Res. 51, 2968–2977 (2018).
Song, B. Are metal chalcogenides, nitrides, and phosphides oxygen evolution catalysts or bifunctional catalysts?. ACS Energy Lett. 2, 1937–1938 (2017).
Song, B. et al. In situ oxidation studies of high-entropy alloy nanoparticles. ACS Nano 14, 15131–15143 (2020).
Bian, H. et al. HEA-NiFeCuCoCe/NF through ultra-fast electrochemical self-reconstruction with high catalytic activity and corrosion resistance for seawater electrolysis. Chem. Eng. J. 477, 147286 (2023).
Wen, Y. et al. Dual-phase B-doped FeCoNiCuPd high-entropy alloys for nitrogen electroreduction to ammonia. Chem. Commun. 59, 13371–13374 (2023).
Jimenez-Arevalo, V. M. et al. Energy transfer by mechanical alloying and electrocatalytic performance of the as-sintered self-supported high-entropy alloy FeNiCoCuMo. Adv. Eng. Mater. 27, 2400807 (2025).
Hu, J. et al. In situ reconstruction of high-entropy heterostructure catalysts for stable oxygen evolution electrocatalysis under industrial conditions. Adv. Mater. 36, 2310918 (2024).
He, Q. et al. Highly defective Fe-based oxyhydroxides from electrochemical reconstruction for efficient oxygen evolution catalysis. ACS Energy Lett. 3, 861–868 (2018).
Huang, X. et al. Formation of disordered high-entropy-alloy nanoparticles for highly efficient hydrogen electrocatalysis. Small 20, 2311631 (2024).
Wang, Y. et al. Self-encapsulation of high-entropy alloy nanoparticles inside carbonized wood for highly durable electrocatalysis. Adv. Mater. 36, 2402391 (2024).
Huang, K. et al. Lattice-disordered high-entropy alloy engineered by thermal dezincification for improved catalytic hydrogen evolution reaction. Adv. Mater. 36, 2304867 (2024).
Yang, Y. et al. Vacancy induced microstrain in high-entropy alloy film for sustainable hydrogen production under universal pH conditions. Energy Environ. Sci. 17, 5854–5865 (2024).
Feng, G. et al. Sub-2 nm ultrasmall high-entropy alloy nanoparticles for extremely superior electrocatalytic hydrogen evolution. J. Am. Chem. Soc. 143, 17117–17127 (2021).
Sarkar, A. et al. High-entropy oxides: Fundamental aspects and electrochemical properties. Adv. Mater. 31, 1806236 (2019).
Ren, J. T., Chen, L., Wang, H. Y. & Yuan, Z. Y. High-entropy alloys in electrocatalysis: from fundamentals to applications. Chem. Soc. Rev. 52, 8319–8373 (2023).
Yu, Y. -f et al. High electrocatalytical performance of FeCoNiCuPd high-entropy alloy for nitrogen reduction reaction. Mol. Catal. 519, 112141 (2022).
Yang, Z., Gao, W. & Jiang, Q. A machine learning scheme for the catalytic activity of alloys with intrinsic descriptors. J. Mater. Chem. A 8, 17507–17515 (2020).
Shiota, T., Ishihara, K. & Mizukami, W. Lowering the exponential wall: accelerating high-entropy alloy catalysts screening using local surface energy descriptors from neural network potentials. Digit. Discov. 4, 738–751 (2025).
Pan, Y. et al. Accelerating the discovery of oxygen reduction electrocatalysts: High-throughput screening of element combinations in Pt-based high-entropy alloys. Angew. Chem. Int. Ed. 63, e202407116 (2024).
Kim, J., Choi, S., Cho, J., Kim, S. Y. & Jang, H. W. Toward multicomponent single-atom catalysis for efficient electrochemical energy conversion. ACS Mater Au 2, 1–20 (2022).
Zhang, Z. et al. Simple construction of ruthenium single atoms on electrospun nanofibers for superior alkaline hydrogen evolution: A dynamic transformation from clusters to single atoms. Chem. Eng. J. 392, 123655 (2020).
Xu, Y., Wang, C., Huang, Y. & Fu, J. Recent advances in electrocatalysts for neutral and large-current-density water electrolysis. Nano Energy 80, 105545 (2021).
Liu, F. et al. Rational design of better hydrogen evolution electrocatalysts for water splitting: a review. Adv. Sci. 9, 2200307 (2022).
Gao, L. et al. Autocatalytic surface reduction-assisted synthesis of PtW ultrathin alloy nanowires for highly efficient hydrogen evolution reaction. Adv. Energy Mater. 12, 2103943 (2022).
Wei, M., Sun, Y., Zhang, J., Zeng, J. & Wang, J. Coupling high-entropy core with Rh shell for efficient pH-universal hydrogen evolution. Small 20, 2403353 (2024).
Wang, Q. et al. Size-controllable high-entropy alloys toward stable hydrogen production at industrial-scale current densities. Adv. Mater. 37, 2420173 (2025).
Wei, M. et al. High-entropy alloy nanocrystal assembled by nanosheets with d–d electron interaction for hydrogen evolution reaction. Energy Environ. Sci. 16, 4009–4019 (2023).
Song, J. et al. A review on fundamentals for designing oxygen evolution electrocatalysts. Chem. Soc. Rev. 49, 2196–2214 (2020).
Park, H. et al. Surface-tailored medium entropy alloys as radically low overpotential oxygen evolution electrocatalysts. Small 18, 2105611 (2022).
Wu, Y. et al. Triggering lattice oxygen activation of single-atomic Mo sites anchored on Ni–Fe oxyhydroxides nanoarrays for electrochemical water oxidation. Adv. Mater. 34, 2202523 (2022).
Qiu, H.-J. et al. Noble metal-free nanoporous high-entropy alloys as highly efficient electrocatalysts for oxygen evolution reaction. ACS Mater. Lett. 1, 526–533 (2019).
Yao, L. et al. Sub-2 nm IrRuNiMoCo high-entropy alloy with iridium-rich medium-entropy oxide shell to boost acidic oxygen evolution. Adv. Mater. 36, 2314049 (2024).
Zhou, X. et al. Atomic structure amorphization and electronic structure reconstruction of FeCoNiCrMox high-entropy alloy nanoparticles for highly efficient water oxidation. Small 20, 2405596 (2024).
Cai, Z.-X., Bolar, S., Ito, Y. & Fujita, T. Enhancing oxygen evolution reactions in nanoporous high-entropy catalysts using boron and phosphorus additives. Nanoscale 16, 4803–4810 (2024).
Xu, H. et al. A hierarchically porous Fe-N-C synthesized by dual melt-salt-mediated template as advanced electrocatalyst for efficient oxygen reduction in zinc-air battery. Appl. Catal. B: Environ. 305, 121040 (2022).
Yao, Z. C., Tang, T., Hu, J. S. & Wan, L. J. Recent advances on nonprecious-metal-based bifunctional oxygen electrocatalysts for zinc–air batteries. Energy Fuels 35, 6380–6401 (2021).
Zhang, J., Yang, H. B., Zhou, D. & Liu, B. Adsorption energy in oxygen electrocatalysis. Chem. Rev. 122, 17028–17072 (2022).
Zhao, X. et al. Designing a built-in electric field for efficient energy electrocatalysis. ACS Nano 16, 19959–19979 (2022).
Li, J. et al. Metal-organic framework-derived graphene mesh: a robust scaffold for highly exposed Fe–N4 active sites toward an excellent oxygen reduction catalyst in acid media. J. Am. Chem. Soc. 144, 9280–9291 (2022).
Liu, Q. et al. A fine 3d transition metal regulation strategy toward high-entropy alloy mesoporous nanotubes as efficient electrocatalysts. Chem. Eng. J. 477, 147099 (2023).
Zhao, X. et al. Multiple metal–nitrogen bonds synergistically boosting the activity and durability of high-entropy alloy electrocatalysts. J. Am. Chem. Soc. 146, 3010–3022 (2024).
Zhu, H. et al. Lewis acid sites incorporation promotes CO2 electroreduction to multicarbon oxygenates over B-CuO nanotubes. Appl. Catal. B: Environ. 339, 123082 (2023).
Hao, J. et al. Interatomic electron transfer promotes electroreduction CO2-to-CO efficiency over a CuZn diatomic site. Nano Res. 16, 8863–8870 (2023).
Wang, G. et al. Electrocatalysis for CO2 conversion: from fundamentals to value-added products. Chem. Soc. Rev. 50, 4993–5061 (2021).
Reske, R., Mistry, H., Behafarid, F., Roldan Cuenya, B. & Strasser, P. Particle size effects in the catalytic electroreduction of CO2 on Cu nanoparticles. J. Am. Chem. Soc. 136, 6978–6986 (2014).
Shi, P. et al. Enhanced strength–ductility synergy in ultrafine-grained eutectic high-entropy alloys by inheriting microstructural lamellae. Nat. Commun. 10, 489 (2019).
Nellaiappan, S. et al. High-entropy alloys as catalysts for the CO2 and CO reduction reactions: experimental realization. ACS Catal. 10, 3658–3663 (2020).
Wen, Y. et al. A coherent Pd–Pd16B3 core–shell electrocatalyst for controlled hydrogenation in nitrogen reduction reaction. Adv. Funct. Mater. 34, 2400849 (2024).
Zhu, X. et al. Aqueous electrocatalytic N2 reduction for ambient NH3 synthesis: recent advances in catalyst development and performance improvement. J. Mater. Chem. A 8, 1545–1556 (2020).
Wan, Y., Xu, J. & Lv, R. Heterogeneous electrocatalysts design for nitrogen reduction reaction under ambient conditions. Mater. Today 27, 69–90 (2019).
Iqbal, M. S. et al. Single-atom catalysts for electrochemical N2 reduction to NH3. Rare Metals 42, 1075–1097 (2023).
Zhang, D. et al. Multi-site electrocatalysts boost pH-universal nitrogen reduction by high-entropy alloys. Adv. Funct. Mater. 31, 2006939 (2021).
Sheng, H. et al. Linear paired electrochemical valorization of glycerol enabled by the electro-Fenton process using a stable NiSe2 cathode. Nat. Catal. 5, 716–725 (2022).
Zhao, G., Fang, C., Hu, J. & Zhang, D. Platinum-based electrocatalysts for direct alcohol fuel cells: Enhanced performances toward alcohol oxidation reactions. ChemPlusChem 86, 574–586 (2021).
Lamy, C. et al. Recent advances in the development of direct alcohol fuel cells (DAFC). J. Power Sources 105, 283–296 (2002).
Zhao, X. et al. Recent advances in catalysts for direct methanol fuel cells. Energy Environ. Sci. 4, 2736–2753 (2011).
Fan, L. et al. High Entropy Alloy Electrocatalytic Electrode toward Alkaline Glycerol Valorization Coupling with Acidic Hydrogen Production. J. Am. Chem. Soc. 144, 7224–7235 (2022).
Hu, M., Li, J., Liu, T., Wu, Z. & Du, Y. Insights into the formation and growth of high entropy PdPtSnPbNi nanowires to obtain catalysts with high alcohol electrocatalytic oxidation activity. J. Colloid Interface Sci. 675, 481–487 (2024).
Hao, J. et al. Suppression of structural heterogeneity in high-entropy intermetallics for electrocatalytic upgrading of waste plastics. Angew. Chem. Int. Ed. 64, e202419369 (2024).
Geng, R. et al. Self-complementary effect through d-d orbital coupling in high-entropy alloy nanowires boosting electrocatalytic biomass upgrading. Appl. Catal. B: Environ 365, 124979 (2025).
Ren, P. et al. High-entropy environments enable metal surface-catalyzed nucleophilic electrooxidation. Angew. Chem. Int. Ed. 64, e202502776 (2025). This paper offers novel insights for the development of Ni-based HEA catalysts for biomass valorization.
Tu, R. et al. Ultra-dilute high-entropy alloy catalyst with core-shell structure for high-active hydrogenation of furfural to furfuryl alcohol at mild temperature. Chem Eng J 452, 139526 (2023).
Li, W.-H. et al. A single-atom cobalt catalyst for the fluorination of acyl chlorides at parts-per-million catalyst loading. Angew. Chem. Int. Ed. 61, e202209749 (2022).
Lin, L. et al. High-entropy sulfides as electrode materials for Li-ion batteries. Adv. Energy Mater. 12, 2103090 (2022).
Zaid, H. et al. Self-organized growth of 111-oriented (VNbTaMoW)N nanorods on MgO(001). Nano Lett. 21, 577–582 (2021).
Yang, Y. et al. Determining the three-dimensional atomic structure of an amorphous solid. Nature 592, 60–64 (2021).
Jia, Z. et al. A novel multinary intermetallic as an active electrocatalyst for hydrogen evolution. Adv. Mater. 32, 2000385 (2020).
Gu, K. et al. Defect-rich high-entropy oxide nanosheets for efficient 5-Hydroxymethylfurfural electrooxidation. Angew. Chem. Int. Ed. 60, 20253–20258 (2021).
Sun, Y. et al. Interface-mediated noble metal deposition on transition metal dichalcogenide nanostructures. Nat. Chem. 12, 284–293 (2020).
Perim, E. et al. Spectral descriptors for bulk metallic glasses based on the thermodynamics of competing crystalline phases. Nat. Commun. 7, 12315 (2016).
Santodonato, L. J. et al. Deviation from high-entropy configurations in the atomic distributions of a multi-principal-element alloy. Nat. Commun. 6, 5964 (2015).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (NSFC) (52273058), Basic Research Program of Jiangsu (BK20220156), and the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Contributions
J.C. conducted the literature search and prepared the first draft of the paper and the figures. H.Z. proposed the idea. H.Z. supervised, reviewed, edited the paper, and acquired the funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Materials thanks Ho Won Jang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: [EBM name(s)] and Jet-Sing Lee. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cai, J., Zhu, H. Surface-engineered nanostructured high-entropy alloys for advanced electrocatalysis. Commun Mater 6, 118 (2025). https://doi.org/10.1038/s43246-025-00838-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43246-025-00838-8