Abstract
The rising demand for lithium in energy storage technologies requires the development of sustainable and selective recovery methods from unconventional, earth-abundant brine resources. Moving beyond traditional lithium mining and pH-swing-driven ion exchange, electrochemical pathways offer a promising, environmentally friendly alternative for lithium capture. In this perspective, we explore the potential of H2TiO3 (HTO) ion-sieve materials, widely known for their pH-driven lithium selectivity, in a membrane-free, single-cell electrochemical system. This approach leverages an applied voltage bias (−1.2 V vs. Ag/AgCl) to enhance lithium concentration at the electrode surface, driving the H⁺-Li⁺ exchange without external pH adjustment. Under these conditions, lithium adsorption capacity reached 9.61 ± 0.2 mg/g, over six times higher than in the physisorption control. The system also demonstrated superior lithium selectivity over competing cations in complex brine, with separation factors of 32.41 for Li+/Na+, 43.5 for Li+/K+ and 7.6 for Li+/Mg2+. By eliminating the need for chemical pH swings and enabling electricity-driven lithium enrichment at the electrode interface, this approach highlights the potential of electrochemical pathways for sustainable, selective lithium recovery from complex brine feedstocks. Future directions for advancing material design, selectivity mechanisms, and process-level optimization are discussed to guide further research in this emerging field.
Similar content being viewed by others
Motivation and background
The rapid growth of renewable energy and storage technologies relies on developing sustainable and cost-effective methods to recover critical metals. These metals are essential for tackling the challenges posed by the inconsistent availability and scaling of solar and wind power1. To unlock the full potential of these intermittent power sources, advanced storage solutions, particularly batteries, are vital for delivering reliable, dispatchable power. Such energy storage technologies play a crucial role in enhancing energy security and enabling the large-scale integration of renewable power into the grid2. Among electrochemical energy storage technologies, lithium ion batteries are growing rapidly due to their versatility for applications ranging from grid-scale storage to electric vehicles (EVs)3. In 2023, global lithium demand surged to 165,000 tons, with batteries accounting for 85% of this demand, representing 30% increase from 20224. Global lithium demand is expected to reach 0.7 million tons in 2030 and double to 1.4 million tons in 2040, due to growing energy storage needs1,5. Over 70% of global lithium is currently sourced from salt lake brines, but rising demand is accelerating interest in extraction from abundant, lower-concentration sources such as geothermal, oilfield brines, and seawater3,6.
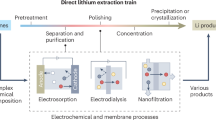
Since most lithium is currently obtained through economical but slow and environmentally challenging solar evaporation processes from brine sources, there is a need for more sustainable pathways to recover lithium7,8 Geothermal and Salt Lake brines pose an even greater challenge, as they contain significantly lower Li+ concentration compared to other competing cations (Na+ and Mg2+) further complicating selective extraction. To address this challenge, direct lithium extraction techniques such as ion exchange9,10, solvent extraction11, nanofiltration, and electrochemical separations12 have emerged as promising alternatives. Nevertheless, these pathways involve significant use of chemicals for pH adjustment or solvents for ion separation, posing challenges related to cost, scalability, and environmental sustainability. Therefore, novel economical, energy–efficient, and environmentally benign routes are needed for the selective separation of lithium from complex brines to advance sustainable solutions.
Electrochemical approaches for selective lithium recovery from brines remain underexplored, yet they offer a sustainable alternative to conventional chemical extraction methods. By minimizing the use of acids, bases, and organic solvents, and leveraging renewable electricity, these approaches have the potential for lower environmental and energy footprints. However, several critical challenges such as (a) low Li selectivity, (b) poor electrode stability under highly saline conditions, and (c) energy inefficiencies, demand further research. Use of membrane-assisted electrochemical systems improve selectivity but introduce additional issues including increased electrical resistance, fouling, and added system complexity and cost. Addressing these challenges is essential for realizing practical, scalable, and economically viable electrochemical lithium recovery technologies.
Electrochemical lithium capture pathways
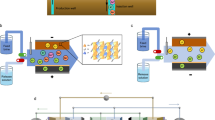
Proposed electrochemical lithium pathways in literature can be broadly categorized into electromembrane/electrodialysis processes and electrochemical ion pumping/electrosorption1. Electrodialysis (ED) uses an applied electric field and ion-exchange membranes to drive the migration of dissolved ions, enabling their selective removal from saline streams. For lithium recovery from brines, ED offers a promising, energy-efficient alternative to conventional evaporation and precipitation methods, which are slow and resource-intensive. The effectiveness of ED depends critically on the properties of the membranes, which must deliver high selectivity, low resistance, and long-term chemical and mechanical stability in highly saline, multicomponent environments13,14. Fig. 1A is a schematic representation of selective electrodialysis for lithium extraction from brine. A major advantage of ED over traditional processes is its ability to directly produce lithium hydroxide (LiOH) which is a preferred precursor for lithium-ion batteries, without first forming lithium carbonate (Li2CO3), thereby minimizing CO2 emissions associated with intermediate conversion steps15,16. Recent progress in membrane materials and system design has advanced ED toward industrial viability15, indicating their technological readiness and commercial viability.
Electrochemical ion pumping is another approach to selectively capture lithium17,18. Fig. 1B represents the steps involved in electrochemical ion-pumping route: (1) intercalation of lithium ions onto electrode materials at negative bias, (2) replacement of the lithium source solution with a recovery solution, (3) application of positive bias to the electrode to release and recover lithium, and (4) replacement of the recovery solution with the source solution to reinitiate this cycle19. In contrast to the ED, electrodes in electrosorption processes are directly involved in the selective capture of lithium ions. Lithium-ion battery cathode and anode materials are often explored for this application, owing to the lithium intercalation mechanism which enables lithium extraction20. The absence of a membrane, ion pumping methods can operate in harsh geothermal brine conditions such as high salinity, pressure, and temperature21,22. Moreover, the operational voltage required for electrosorption is generally less than 2.0 V23, in contrast to electromembrane that can operates at voltage range from 5 to 30 V24. However, a primary challenge in the electrochemical ion pumping process is identifying selective electrode materials capable of efficiently capturing lithium with high-selectivity. Examples of such electrode materials25,26,27,28,29 are presented in Table 1. Electrosorption-based lithium recovery is primarily constrained by low ion selectivity, as competing cations such as magnesium and sodium co-adsorb with lithium, and by the inherently limited sorption capacity of available electrode materials. For example, materials like LiB2O4 show less than 0.5% lithium uptake experimentally. These factors, along with electrode stability and durability, directly influence the efficiency, cost-effectiveness, and long-term sustainability of the process30.
Electrosorption materials and their lithium capture mechanism
Lithium capture materials and mechanisms are inspired by battery electrodes due to their similar charge-discharge behavior. Figure 2A illustrates the two primary electrochemical lithium capture mechanisms: capacitive response (Fig. 2A right), where lithium ions accumulate in the electrical double layer, and Faradaic intercalation (Fig. 2A left), where lithium ions insert into the electrode lattice via redox reactions. In Faradaic intercalation, the lithium capture occurs through redox reactions and intercalation into the electrode lattice, which offers higher selectivity due to the crystal structure’s inherent preference for Li⁺ over other cations. This structural selectivity arises from factors such as ionic size compatibility and lattice binding energies. In contrast, capacitive systems rely on the accumulation of Li⁺ in the Helmholtz double layer at the electrode-electrolyte interface, which is a non-Faradaic and surface-limited process. While capacitive capture enables faster kinetics and lower energy consumption, the lithium selectivity arises from a combination of field-enhanced Li⁺ concentration near the surface and the favorable interaction of Li⁺ with the electrode surface. Amongst various Li capture materials, lithium iron phosphate (LFP, LiFePO4), lithium manganese oxide (LMO, LiMn2O4), and lithium nickel cobalt manganese oxide (NCM, LiNi1/3Co1/3Mn1/3O2) have been broadly proposed and studied for the lithium recovery31,32. The Faradaic current response in these materials arises from electrochemical redox reactions at the electrode- electrolyte interface33, followed by lithium capture through the intercalation of lithium into the material, (Fig. 2B), as illustrated by a few example reactions below:
The LFP system demonstrates excellent long-term stability, making it attractive for lithium capture applications. However, LFP suffers from co-intercalation of competing cations, which reduces insertion capacity and selectivity34. In contrast, LMO is favored for its high Li+ selectivity and considerable insertion capacity12,31 attributed to its spinel structure that promotes high ion-conductivity. However, LMO suffers from structural instability due to the Jahn-Taller effect of the manganese during charge and discharge cycling, causing manganese species leaching into the electrolyte affecting the long-term electrode stability9. Another promising approach for lithium capture leverages electrochemical capacitive desalination (CDI), which operates on the principle of electrostatic adsorption of ions into the electrical double layers formed at the surface of porous, conductive electrodes35. Compared to conventional desalination technologies, CDI demonstrates significantly lower specific energy consumption, making it an increasingly attractive solution for water purification35,36. While conventional capacitive deionization (CDI) typically exhibits limited ion selectivity, recent advances have demonstrated the feasibility of targeted separation of specific ions, such as phosphate, nitrate, and lithium. Integrating ion-exchange membranes into CDI systems, known as membrane capacitive deionization, has been shown to significantly enhance ion removal efficiency, charge utilization, and selectivity compared to conventional CDI37. Another promising strategy for selective lithium recovery involves incorporating lithium-ion sieve (LIS) materials into the CDI electrodes. For example, layered hydrated titanates, such as H2TiO3 (Fig. 2C), have been widely studied as lithium-selective materials, traditionally applied in pH-driven ion exchange processes, and are now being explored for electrochemical lithium capture10,38,39,40. In this class of material, lithium capture is achieved in a similar mechanism as occurs in pH driven approach via ion exchange reaction as represented below:
The capacitive response caused by the voltage application across the electrodes results in increased lithium surface concentration in contrast with pH - driven approaches via breaking of Li–O bonds by electrons. This results in a more effective lithium capture without the need for chemically intensive pH adjustment processes.
Electrochemical lithium capture with layered titanate material
Prior advances suggested that H2TiO3 is promising material due to its theoretical lithium adsorption capacity of ~128 mg/g, which is significantly higher than alternative widely used but less stable materials such as λ-MnO2 that has a theoretical capacity of ~60 mg/g10,41. Li and co-workers showed that titania dissolution in H2TiO3 is 0.31% per adsorption and desorption cycle41,42, which is more than an order of magnitude lower than Li2MnO4. Despite the favorable characteristics of H2TiO3 as an active material for electrochemical lithium capture, there remains a limited number of studies that explore its use for electrochemical lithium capture. Bhaskaran et al.28, developed a membrane capacitive desalination approach utilizing H2TiO3. Lithium insertion capacity as high as 13.67 mg/g was demonstrated in the optimal condition utilizing 60% reduced graphene oxide additive. However, investigation into the selectivity of the material is limited, focusing mainly on comparisons with pure sodium chloride and lacking experiments with complex brine solutions28. Zhang et al.29, explored ways to enhance H2TiO3 based electrode performance through tannic acid treatment in addition to the use of reduced graphene oxide as the conducting agent. Acid treatment enhanced the electrode’s hydrophilicity, improving lithium adsorption with a demonstrated capacity of up to 25.2 mg/g in the optimal configuration. The process is also shown to have a high separation factor over sodium and magnesium. However, the feed brine utilized for the study has a high lithium concentration of 1412 mg/L and a pH of 10.9829. These conditions are quite favorable for extraction, but most deposits have lithium concentration less than 400 mg/L8. These challenges motivate innovative advances in electrochemical capture of lithium selectively by harnessing synthetic brines over H2TiO3 layered electrodes.
In this perspective, we investigate the potential of H2TiO3 (HTO)-based electrodes for electrochemical lithium recovery from brines using a membrane-free system, offering a promising alternative to conventional chemical ion-exchange processes, which are inherently slow. By applying an electric field, the local cation concentration at the electrode surface is enhanced, accelerating the lithium exchange kinetics. However, this approach also introduces challenges related to differences in hydration radius, ion mobility, ion-exchange selectivity, and hydration energy among competing ions present in complex brines. Here, we systematically examine the influence of solution pH on lithium selectivity and uptake, assess the energy efficiency of the process, and critically discuss the limitations and research needs for advancing HTO-based electrochemical capture systems toward practical, competitive lithium recovery technologies.
Materials and methods
Synthesis of H2TiO3 (HTO) powder
Li2TiO3 (LTO) is synthesized through a solid-state reaction1, by mixing Li2CO3 (Sigma Aldrich) and anatase TiO2 (Thermo Fischer) in 2: 1 molar ratio of Li/Ti with a mortar and pestle for 20 min. Furthermore, as synthesized LTO was annealed at 700 °C for 4 h (ramp rate = 6 °C/min) in a muffle furnace. Moreover, to remove any unreacted lithium carbonate, the annealed LTO powder was washed thoroughly with deionized water (DI) water and dried overnight in an oven at 80 °C. Subsequently, HTO powder is synthesized by magnetically stirring the LTO powder at 800 rpm in 0.2 M hydrochloric acid (HCl) with a solid to liquid ratio of 1:200 for 24 h. Finally, HTO powder was obtained with repeated washing and centrifugation followed by overnight drying at 80 °C in vacuum oven.
Preparation of electrode
To prepare electrodes for lithium capture, a carbon cloth was used as conductive support. Prior to the coating process, carbon cloth (Fuel Cell Store, 360 µm) was cut into 3 × 3 cm2 and sonicated in a 1:1:1 volumetric ratio of acetone, ethanol, DI water solutions for 10 min and dried at 80 °C for 15 min. Further, as synthesized HTO powder, C65 (Timcal), and PVDF (Sigma Aldrich) were mixed in NMP (Thermo Fischer) using mortar pestle with a mass ratio of 8:1:1, respectively. The as-prepared slurry was uniformly coated on the surface of carbon cloth with the help of glass slides. Furthermore, resultant electrodes were dried in a vacuum oven at 60 °C for 12 h, to evaporate the volatile solvent and fixation of the coatings (Fig. S1). The difference of mass between the electrode and plain carbon cloth is measured and is used to calculate the active mass loading of the electrode.
Electrochemical lithium adsorption study
Electrochemical lithium adsorption was conducted in a three-electrode system electrolytic cell, where the prepared HTO electrode was used as working electrode, platinum mesh as counter electrode, and Ag/AgCl in saturated KCl was used as reference electrode. The working HTO electrode was suspended in the electrolyte by a PTFE holder with platinum contact. The distance between the working and counter electrode was approximately 2 cm. The synthesized brine solution was used as feed, prepared from chloride salts by mimicking Atacama brine2,3 and the composition is presented in Table S1. In a typical electrochemical lithium capture process, the electrodes were immersed in 35 mL (dipped area 3 × 2 cm2) of the feed solution. Lithium capture experiments in this study are conducted in batch mode with applied potential of −1.2 vs Ag/AgCl while magnetically stirred at 50 rpm for 2 h. The influence of pH on the Li capture process was studied by varying the pH from 5.8, 7.0, and 9.0 using 0.1 M ammonium hydroxide. A set of lithium physisorption experiments were also conducted for comparison in the similar configuration in absence of electrical potential.
Material characterizations
The crystal structures and crystallinity of the synthesized HTO powder and electrode samples were confirmed through X-Ray Diffraction (XRD, Bruker D8 Advance ECO powder diffractometer) with Cu Kα radiation at 40 kV, and 25 mA. Typical scan was done from a 2θ angle of 10° to 80° with and scan step size of 0.019°. Raman Spectroscopy (WITec Alpha300R) was used to determine the chemical bond formation in different states of the electrode. Raman Spectroscopy was carried out using 532 nm laser. The scanning electron microscopy (SEM) analysis was done on Zeiss Sigma 500 SEM. The electrolyte concentration was measured with Inductively Coupled Plasma-Optical Emission Spectrometry (ICP-OES, Thermo Scientific, iCAP 7400). The elemental states of the elements were measured using X-ray photoelectron spectroscopy (XPS) on Thermo Fisher Scientific Nexsa G2 XPS instrument with monochromatic Al-Kα X-ray source.
Results and discussion
Electrode development for electrochemical lithium capture
The approach to synthesize and prepare HTO electrodes is shown in Fig. S1. The structural features of synthesized Li2TiO3 and H2TiO3 powder and electrode are investigated using X-Ray Diffractogram (XRD) and shown in Fig. 3A. Figure 3A(a, b) displays the XRD patterns of Li2TiO3 powder and electrode, respectively. The characteristic XRD peaks are located at 2θ values of 18.01 °, 43.45 °, 63.19 ° and are indexed to (002), (22-2) and (312) planes of monoclinic-structured Li2TiO3 (ICDD PDF 00-033-0831). The structure of Li2TiO3 powder and electrode are in agreement with the monoclinic phase of β-Li2TiO310,43. Fig. 3A(c, d) show the disappearance of the (111), (22-2) and (312) and (062) XRD peaks in the H2TiO3 powder and electrode demonstrating the structural disorders arising due to the delithiation process. The diffraction pattern of H2TiO3 powder and electrode is consistent with that of layered structure of H2TiO310. To investigate the influence of acid delithiation on O-Li bonds in LTO, the characteristic features of the synthesized Li2TiO3 and H2TiO3 powder and electrode are investigated using Raman spectroscopy (Fig. 3B(a–d)). The Li-O stretching and O-Li-O bending vibration results in shifts in the Raman peaks at 293, 308, and 405 cm−15 on the LTO powder and electrode samples (Fig. 3B(a, b)). The intensity corresponding to these peaks is found to reduce or disappear after the acid delithiation (Fig. 3B(c, d)), further providing evidence of successful HTO synthesis. The peak at 663 cm−1 is assigned to Ti-O bonds stretching, as the region between 550–700 cm−1 is associated with TiO6 octahedra in lithium titanate materials44. This Ti–O stretching peak appears broadened after delithiation, suggesting the presence of hydrogen in different coordination states, which could correspond to hydrogen in H and HTi2 layers. Both X-Ray Diffraction (XRD) patterns and Raman spectra show no notable differences between the powder and electrode form of the LTO and HTO, which indicate the successful preparation of the electrode that retains the characteristics of the LTO and HTO powders. The structural and morphological features of the H2TiO3 electrode were analyzed, SEM imaging revealed agglomerated particles with primary particle sizes in the range of 100–200 nm (Fig. S3), consistent with prior studies10. Literature reports further indicate that H2TiO3 exhibits a mesoporous structure, with BET surface area measurements showing type IV nitrogen adsorption–desorption isotherms and H1-type hysteresis, characteristic of uniform mesoporosity and cylindrical pores36,45. This mesoporosity, together with the observed particle morphology, provides a high surface area and accessible pore network, facilitating lithium adsorption through enhanced ion accessibility and electric double-layer formation within the pores. Furthermore, XPS analyses were conducted on the H2TiO3 electrodes to examine the surface chemical valence states of the elements. Figure S4A presents the deconvoluted Ti 2p spectrum of HTO, showing binding energies at 458.08 and 456.50 eV for Ti 2p3/2, and 463.97 and 461.58 eV for Ti 2p1/2. This observation confirms the coexistence of Ti+4 and Ti+3 oxidation states, where the presence of Ti+3 enhances the electronic conductivity by introducing additional charge carriers. These results are consistent with previous reports46,47. The O 1 s peak near 530 eV corresponds to lattice oxygen in the Ti-O framework (Fig. S4B). A second peak, centered around 531.5–532.5 eV, is commonly attributed to surface hydroxyl groups due to surface defects(Ti+3). Additionally, the appearance of a peak near 534 eV is typically associated with adsorbed water, which may be due to the material’s high surface area and strong affinity for moisture48. The mixed-valence state is expected to facilitate charge transport within the electrode matrix for electrochemical application.
A X-ray diffraction patterns showing characteristic peaks of the synthesized materials at different synthesis stages: (a) Li2TiO3 powder, (b) Li2TiO3 electrode, (c) H2TiO3 powder, and (d) H2TiO3 electrodes. B Raman spectra of the same samples, showing Li-O stretching and O-Li-O bending vibrational modes at 293 cm⁻¹, 308 cm⁻¹, and 405 cm⁻¹ respectively. The series (a–d) correspond to the same samples as in (A), with light blue, blue, light green, and dark green traces respectively.
Lithium capture using HTO materials
The synthesized HTO electrodes are used to investigate physisorption and electrochemical lithium capture behavior. The feed for lithium uptake is the simulated Atacama brine with composition as outlined in Table S1. The lithium uptake of the HTO electrodes is investigated by varying the initial pH conditions of feed in three electrode setup (Fig. S2). Figure 4A shows that during physisorption, lithium uptake increases with a slight increase in the initial pH of the solution. In contrast, in the electrochemical capture mode, the amount of lithium captured decreases as the initial pH increases. However, the application of a −1.2 V potential to the working electrode in electrochemical lithium adsorption results in a significant improvement in lithium uptake, averaging more than six times the amount captured compared to the baseline physisorption process. During the electrochemical mode, lithium capture decreases with an increase in the initial pH, which is surprising since alkaline conditions are known to favor lithium uptake in wide-ranging materials including ion exchange resins10 and solvents11. Typically, during the pH driven ion exchange adsorption mechanism, hydroxyl ions in solution assist weak acid dissociation in H2TiO3 thereby promoting Li-O bond formation10. The electrochemical lithium uptake indicates that the acid dissociation step is not essential for the adsorption process and electric double layer formation may facilitate a distinct favorable condition for the lithium uptake. This apparent contradiction can be attributed to the use of a NH4Cl/NH4OH buffer for pH adjustment, which introduces NH4⁺ cations into the electrolyte. These NH4⁺ ions compete with Li⁺ for adsorption sites within the Helmholtz layer at the negatively charged electrode surface. As the pH increases, the concentration of NH4⁺ in the solution also increases, leading to a concentration-dependent suppression of Li-H exchange. This competitive effect of NH4⁺ likely explains the observed decrease in lithium capture at higher pH in the electrochemical configuration. Interestingly, final pH of the brine in both electrochemical and physisorption modes decreases alongside the lithium adsorption process, as shown in Fig. 4 (B). Notably, the final pH in the electrochemical mode across all initial pH conditions converged to a similar value of 4.1. The drop in pH indicates that lithium adsorption is accompanied by the release of H+ ions, following an ion exchange reaction as shown below:
A Lithium uptake (mg/g) from Atacama brine after 2 h at 50 rpm under different pH conditions, comparing physisorption and electrochemical sorption. B pH of the solution before and after sorption under both conditions. C Selectivity coefficients of lithium over potassium (blue bars), sodium (red bars), and magnesium (gray bars) at different pH values during electrochemical sorption. D Schematic representation of ion distribution and sorption behavior at the electrode surface during electrochemical sorption.
In this reaction, MO-H refers to the metal oxide surface. The observed increase in lithium adsorption with higher initial pH in the physisorption mode (Fig. 4A) can be explained by the alkaline conditions depleting released H+ ions, thereby shifting the equilibrium to the right, favoring lithium adsorption. Conversely, the significant increase of lithium adsorption in the electrochemical mode (Fig. 4A) is attributed to the applied field increasing lithium concentration at the electrode surface by ~4-5x (complete description in Supplementary section), which accelerates H⁺-Li⁺ exchange. Lithium selectivity over other cations in the brine separation process is shown in Fig. 4C and Table S2. The observed selectivity stems from the H2TiO3’s mesoporosity and stronger O-Li bonding favoring lithium uptake over larger or more strongly hydrated cations. It is observed that lithium separation is generally most selective over potassium, followed by magnesium and sodium. Selectivity over potassium is expected to be high due to a significant difference in ionic radii compared to lithium49. However, selectivity over sodium is found to be lower than magnesium despite sodium’s larger ionic radii, which can possibly be attributed to the large concentration of sodium in the initial brine solution (Table S1). Based on the results of lithium uptake and initial feed brine pH variation during the physisorption and electrochemical lithium uptake, the influence of charged surfaces on sorption behavior is proposed in Fig. 4D. As shown in Fig. 4D, in the absence of the external electric field, the cations in the brine can diffuse freely within the solution, whereas application of negative potential to working electrode leads to migration of the cations towards the electrode surface and specifically forms the layer of adsorbed cations50. This phenomenon increases the surface lithium ion concentration, which shifts the equilibrium to the right (see Eq. 1), thereby increasing the lithium uptake. To assess the material’s stability in the electrochemical configuration, multiple cyclic voltammetry analysis was conducted at a slow scan rate for 100 cycles Fig. S5. SEM (Fig. S6) and XRD analyses (Fig. S7) after cycling confirmed no significant morphological degradation of the electrode, although additional phases corresponding to adsorbed cations such as Li⁺, Ca+2, and Mg+2 were detected after the adsorption process. These demonstrate the structural robustness of H2TiO3 under repeated electrochemical operation, underscoring its potential for sustainable and reusable lithium recovery from complex brines.
A preliminary estimate of the energy consumption for lithium recovery in this system was calculated as approximately 5.6 kWh per kilogram of lithium (complete description in Supplementary section). This compares favorably with conventional processes such as evaporation ponds (30–70 kWh/kg-Li) and electrodialysis or membrane-based methods (10–20 kWh/kg-Li)37,51. Competitive energy efficiency, coupled with the elimination of harsh chemicals, large land requirements, and pH swings, highlights the potential of this approach as a sustainable alternative. However, lithium selectivity remains dependent on the composition of cations in the Stern layer, where the higher hydration enthalpy and larger hydration radius of Li⁺ limit its relative accumulation under an applied field, indicating a trade-off between energy input and selectivity that warrants further optimization.
Mechanistic insights into electrochemical lithium capture using HTO materials
To assess the efficacy of electrochemical separation, the cyclic voltammetry of HTO electrode is performed. The cyclic voltammetry curves of the electrode show largely capacitive behavior without any obvious faradaic peak (Fig. 5A). The peak current can also be correlated with scan rate variations to gain further insight into electrochemical behavior. A current response that is proportional to the square root of scan rate (v1/2) is said to be limited by internal diffusion52, implying a fast faradaic electron transfer as well as a freely diffusing analyte53 from the bulk to the surface and vice versa. The current of such a process is governed by the following expression54:
A Cyclic voltammograms recorded at scan rates ranging from 10 to 200 mV/s in 1 M LiCl electrolyte with platinum mesh counter electrode. B Peak current as a function of scan rate (ν) for oxidation (red circles) and reduction (black circles) processes. C Peak current as a function of square root of scan rate (ν1/2) for oxidation (red circles) and reduction (black circles) indicating linear correlation between peak current and scan rate (ν), along with the deviation from linearity with (ν1/2).
In this expression, i is current (A), C is capacitance (F), and \(v\) is scan rate (mV/s). In contrast, a current response that is proportional to the scan rate (\(v\)) is said to be surface limited52, and the current arises from capacitive current due to the Helmholtz double layer formation. Formally, the current response can be described as follows52:
In this expression, Cd is the capacitance of the electrode.
Figure 5 (B) and (C) show that both the oxidation and reduction current responses exhibit a better linear fit against the scan rate (\(v\)) rather than against the square root of the scan rate (\(v\)1/2). The deviation from linearity with \(v\)1/2, as described by Eq. (5) and the good linear fit with v as described by Eq. (6) indicates that the exchange process is dominated by surface-controlled kinetics (Fig. 2A). These results suggest that the observed current response from the cyclic voltammetry arises from capacitive current due to Helmholtz double layer formation, and the current response is due to an electrode-adsorbed species52,55. This suggests that there is no electron transfer to the lithium in the process. Instead, the applied voltages enable the formation of inner Helmholtz plane consisting of lithium ion, which then further interacts with H2TiO3 active sites. In similar lithium capture experiments, Zhang and co-workers showed that there is no change on the oxidation state of titanium before and after lithium adsorption29, which further supports the evidence of the absence of electron transfer in the lithium capture process.
Key conclusions, research directions, and opportunities
Electrochemical lithium extraction offers a promising pathway for industrial-scale production of lithium-based products such as LiOH.H2O and Li2CO3, combining lower process complexity with the ability to utilize renewable electricity sources and tolerate diverse brine compositions. Its adaptability to varying feedstocks and operation across a wide pH range reduces the need for extensive pretreatment, and its faster kinetics compared to slow evaporation-based processes make it an attractive, sustainable alternative to conventional chemical and membrane-based methods for meeting the growing lithium demand.
While prior studies have largely focused on conventional battery materials and intercalation-based mechanisms, our findings show that ion-exchange materials such as H2TiO3 can also be effectively utilized in an electrochemical system. Although the lithium capture process is non-Faradaic, the application of an electric field enhances the local lithium concentration at the electrode surface, thereby accelerating the Li-H exchange. In this study, a lithium extraction capacity of 9.6 mg/g was achieved without pH adjustment, using a single-compartment, membrane-free system operated in batch mode.
Despite demonstrating the feasibility of H2TiO3-based electrochemical lithium recovery, several challenges remain. First, the observed lithium capture capacity of 9.6 mg/g is significantly lower than the theoretical capacity of ~128 mg/g, as the exchange primarily occurs at solvent-accessible surface sites. This highlights the need to synthesize highly porous materials with greater surface area and accessibility to active sites. Second, the separation factor of lithium over competing cations depends on the thermodynamics and kinetics of the cation-H⁺ exchange and the local cation concentrations in the Stern layer. This warrants further investigation using atomistic simulations, such as DFT or ab initio molecular dynamics, to elucidate these critical properties and their contributions to selectivity. Third, material modification strategies, such as metal doping and surface functionalization, should be explored computationally and experimentally to assess their effects on the thermodynamics and kinetics of Li-H exchange and suppression of competing reactions. Fourth, since the Stern layer cation composition can also be influenced by operational parameters, such as voltage pulsing and other potentiodynamic methods, systematic studies are needed to optimize operating conditions for improved selectivity and efficiency. Finally, comprehensive life cycle assessments and techno-economic analyses are crucial to evaluate the environmental and economic viability of this approach at scale. These findings provide a foundation for advancing H2TiO3-based electrochemical lithium recovery through material innovation, mechanistic understanding, and process optimization toward sustainable and scalable deployment.
Data availability
Data is provided within the manuscript or supplementary information files.
References
Zavahir, S. et al. A review on lithium recovery using electrochemical capturing systems. Desalination 500, 114883 (2021).
Azzuni, A. & Breyer, C. Energy security and energy storage technologies. Energy Procedia 155, 237–258 (2018).
Zhang, Y., Hu, Y., Wang, L. & Sun, W. Systematic review of lithium extraction from salt-lake brines via precipitation approaches. Miner. Eng. 139, 105868 (2019).
Global EV Outlook 2024. https://www.iea.org/reports/global-ev-outlook-2024 (2024).
Global Critical Minerals Outlook 2024. https://www.iea.org/reports/global-critical-minerals-outlook-2024 (2024).
Zhao, X., Li, G., Feng, M. & Wang, Y. Semi-continuous electrochemical extraction of lithium from brine using CF-NMMO/AC asymmetric hybrid capacitors. Electrochim. Acta 331, 135285 (2020).
Xu, X. et al. Extraction of lithium with functionalized lithium ion-sieves. Prog. Mater. Sci. 84, 276–313 (2016).
Vera, M. L., Torres, W. R., Galli, C. I., Chagnes, A. & Flexer, V. Environmental impact of direct lithium extraction from brines. Nat. Rev. Earth Environ. 4, 149–165 (2023).
He, G., Zhang, L., Zhou, D., Zou, Y. & Wang, F. The optimal condition for H2TiO3–lithium adsorbent preparation and Li+ adsorption confirmed by an orthogonal test design. Ionics 21, 2219–2226 (2015).
Marthi, R., Asgar, H., Gadikota, G. & Smith, Y. R. On the structure and lithium adsorption mechanism of layered H2TiO3. ACS Appl. Mater. Interfaces 13, 8361–8369 (2021).
Fernández-Escalante, E., Ibañez, R. & San-Román, M. F. Selective lithium separation from desalination concentrates via the synergy of extractant mixtures. Desalination 556, 116525 (2023).
Kim, S., Joo, H., Moon, T., Kim, S.-H. & Yoon, J. Rapid and selective lithium recovery from desalination brine using an electrochemical system. Environ. Sci. Process. Impacts 21, 667–676 (2019).
Zhang, Y., Paepen, S., Pinoy, L., Meesschaert, B. & Bruggen, B. V. Selectrodialysis: fractionation of divalent ions from monovalent ions in a novel electrodialysis stack. Sep. Purif. Technol. 88, 191–201 (2012).
Nie, X. Y., Sun, S. Y., Song, X. & Yu, J. G. Further investigation into lithium recovery from Salt Lake brines with different feed characteristics by electrodialysis. J. Membr. Sci. 530, 185–191 (2017).
Kong, L. et al. Electro-driven direct lithium extraction from geothermal brines to generate battery-grade lithium hydroxide. Nat. Commun. 16, 806 (2025).
Chen, X. et al. Production of lithium hydroxide by electrodialysis with bipolar membranes. Sep. Purif. Technol. 274, 119026 (2021).
Trócoli, R., Erinmwingbovo, C. & Mantia, F. L. Optimized lithium recovery from brines by using an electrochemical ion-pumping process based on λ-MnO2 and nickel hexacyanoferrate. ChemElectroChem 4, 143 (2017).
Luo, G., Li, X., Chen, L., Chao, Y. & Zhu, W. Electrochemical lithium ion pumps for lithium recovery: a systematic review and influencing factors analysis. Desalination 548, 116228 (2023).
Joo, H., Lee, J. & Yoon, J. Short review: timeline of the electrochemical lithium recovery system using the spinel LiMn2O4 as a positive electrode. Energies 13, 6235 (2020).
Marchini, F., Rubi, D., Del Pozo, M., Williams, F. J. & Calvo, E. J. Surface chemistry and lithium-ion exchange in LiMn2O4 for the electrochemical selective extraction of LiCl from Natural Salt Lake Brines. J. Phys. Chem. C. 120, 15875 (2016).
Calvo, E. J. Electrochemical methods for sustainable recovery of lithium from natural brines and battery recycling. Curr. Opin. Electrochem. 15, 102–108 (2019).
Zhao, X. et al. Efficient lithium extraction from brine using a three-dimensional nanostructured hybrid inorganic-gel framework electrode. ACS Sustain. Chem. Eng. 8, 4827–483 (2020).
Tusekile, A., Elisadiki, J., Dahbi, M., King’ondu, C. K. & Jande, Y. A. C. Electrosorption of paraquat pesticide on activated carbon modified by aluminium oxide (Al2O3) with capacitive deionization. Desalination 572, 117116 (2024).
Dowlatshah, S. et al. Electromembrane extraction of peptides based on hydrogen bond interactions. Anal. Chim. Acta 1275, 341610 (2023).
Pasta, M., Battistel, A. & La Mantia, F. Batteries for lithium recovery from brines. Energy Environ. Sci. 5, 9487 (2012).
Lawagon, C. P. et al. Li1−xNi0.33Co1/3Mn1/3O2/Ag for electrochemical lithium recovery from brine. Chem. Eng. J. 348, 1000–1011 (2018).
Du, X. et al. A novel electroactive λ-MnO2/PPy/PSS core–shell nanorod coated electrode for selective recovery of lithium ions at low concentration. J. Mater. Chem. A 4, 3989–13996 (2016).
Bhaskaran, G. et al. Layered hydrated-titanium-oxide-laden reduced graphene oxide composite as a high-performance negative electrode for selective extraction of Li via membrane capacitive deionization. J. Colloid Interface Sci. 650, 752–763 (2023).
Zhang, J. et al. Bifunctional modification enhances lithium extraction from brine using a titanium-based ion sieve membrane electrode. ACS Appl. Mater. Interfaces 15, 29586–29596 (2023).
Lim, Y. J., Goh, K., Zhao, A. G. Y. & Wang, R. Uranium and lithium extraction from seawater: challenges and opportunities for a sustainable energy future. J. Mater. Chem. A 11, 22551–22589 (2023).
Wu, L. et al. Lithium recovery using electrochemical technologies: advances and challenges. Water Res. 221, 118822 (2022).
Battistel, A., Palagonia, M. S., Brogioli, D., La Mantia, F. & Trócoli, R. Electrochemical Methods for lithium recovery: a comprehensive and critical review. Adv. Mater. 32, 1905440 (2020).
Schoetz, T. et al. Disentangling faradaic, pseudocapacitive, and capacitive charge storage: a tutorial for the characterization of batteries, supercapacitors, and hybrid systems. Electrochim. Acta 412, 140072 (2022).
Liu, S. et al. Reviving the lithium-manganese-based layered oxide cathodes for lithium-ion batteries. Matter 4, 1511–1527 (2021).
Shi, W. et al. Efficient lithium extraction by membrane capacitive deionization incorporated with monovalent selective cation exchange membrane. Sep. Purif. Technol. 210, 885–890 (2019).
Sun, J. et al. Preparation of high hydrophilic H2TiO3 ion sieve for lithium recovery from liquid lithium resources. Chem. Eng. J. 453, 139485 (2023).
Saif, H. M., Crespo, J. G. & Pawlowski, S. Lithium recovery from brines by lithium membrane flow capacitive deionization (Li-MFCDI)—a proof of concept. J. Membr. Sci. Lett. 3, 100059 (2023).
Lawagon, C. P. et al. Adsorptive Li+ mining from liquid resources by H2TiO3: equilibrium, kinetics, thermodynamics, and mechanisms. J. Ind. Eng. Chem. 35, 347–356 (2016).
Lawagon, C. P. et al. Development of high-capacity Li+ adsorbents from H2TiO3/polymer nanofiber composites: systematic polymer screening, characterization and evaluation. J. Ind. Eng. Chem. 70, 124–135 (2019).
Han, H. J., Qu, W., Zhang, Y. L., Lu, H. D. & Zhang, C. L. Enhanced performance of Li+ adsorption for H1.6Mn1.6O4 ion-sieves modified by Co doping and micro array morphology. Ceram. Int. 47, 21777–21784 (2021).
Li, X., Chao, Y., Linlin, C. & Zhu, W. Taming wettability of lithium-ion sieve via different TiO2 precursors for effective Li recovery from aqueous lithium resources. Chem. Eng. J. 392, 123731 (2020).
Zhao, K. et al. A novel Co-doped H2TiO3 spinning composite for efficient lithium recovery from alkaline lithium precipitation mother liquor. Chem. Eng. J. 482, 148989 (2024).
Nakazawa, T. et al. High energy heavy ion induced structural disorder in Li2TiO3. J. Nucl. Mater. 367–370, 1398–1403 (2007).
Marthi, R., Yang, P., Owusu-Fordjour, E. Y. & Smith, Y. R. Role of stacking faults and hydroxyl groups on the lithium adsorption/desorption properties of layered H2TiO3. Mater. Today Adv. 14, 100237 (2022).
Wang, S. et al. Superior lithium adsorption and required magnetic separation behavior of iron-doped lithium ion-sieves. Chem. Eng. J. 332, 160–168 (2018).
Wang, Q. et al. Titanium-based lithium ion sieve adsorbent H2TiO3 with enhanced Li+ adsorption properties by magnetic Fe doping. Sep. Purif. Technol. 346, 127455 (2024).
Wang, J. et al. Enhanced photoelectrochemical properties of Ti3+ self-doped branched TiO2 nanorod arrays with visible light absorption. Materials 11, 1791 (2018).
Guo, L. Q., Qin, S. X., Yang, B. J., Liang, D. & Qiao, L. J. Effect of hydrogen on semiconductive properties of passive film on ferrite and austenite phases in a duplex stainless steel. Sci. Rep. 7, 3317 (2017).
Bard, A. J. & Faulkner, L. R. Electrochemical Methods: Fundamentals and Applications (Wiley, 2001).
Augustyn, V. et al. High-rate electrochemical energy storage through Li+ intercalation pseudocapacitance. Nat. Mater. 12, 518–522 (2013).
González, A., Choque, G., Grágeda, M. & Ushak, S. The development and analysis of a preliminary electrodialysis process for the purification of complex lithium solutions for the production of Li2CO3 and LiOH. Membranes 15, 50 (2025).
Elgrishi, N. et al. A practical beginner’s guide to cyclic voltammetry. J. Chem. Educ. 95, 197–206 (2018).
Liu, Y., Jiang, S. P. & Shao, Z. Intercalation pseudocapacitance in electrochemical energy storage: recent advances in fundamental understanding and materials development. Mater. Today Adv. 7, 100072 (2020).
Wang, J., Polleux, J., Lim, J. & Dunn, B. Pseudocapacitive contributions to electrochemical energy storage in TiO2 (Anatase) nanoparticles. J. Phys. Chem. C. 111, 14925–14931 (2007).
Yang, L. et al. Review—Challenges and opportunities in lithium metal battery technology. J. Electrochem. Soc. 171, 060504 (2024).
Acknowledgements
The authors acknowledge the use of the shared facilities at the Cornell Center for Materials Research (CCMR) which are supported through the National Science Foundation Materials Research Science and Engineering Centers (NSF MRSEC) program (DMR-1719875). The work is also supported by DOE Lithium Extraction Prize.
Author information
Authors and Affiliations
Contributions
L.N. conducted the experiments, analyzed and plotted the data, contributed to literature review, writing and editing of the manuscript. M.A.M. and S.S. contributed to literature review, writing, analysis and editing of the manuscript. A.M. conducted ICP analyses to determine the concentrations of metals in the fluids. G.G. proposed the concept of electrochemical Li uptake and mechanism from brine, oversaw the development and execution of the concept and experimental plan, and edited the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
G.G. is an Editorial Board Member of Scientific Reports, Associate Editor of npj Materials Sustainability, and is the co-founder of Carbon To Stone advancing technologies for resource recovery and carbon management for industrial efficiency. The other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nugroho, L., Mahadik, M.A., Singh, S. et al. Electrochemical lithium capture using titanate materials: mechanistic insights and proposed advances. npj Mater. Sustain. 4, 1 (2026). https://doi.org/10.1038/s44296-025-00076-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44296-025-00076-6