Abstract
Study design:
Experimental investigation of intrathecal transplantation of stem cells by lumbar puncture (LP) in a rat model that simulates human thoracic spinal cord injury (SCI).
Objectives:
To examine the distribution and phenotype of spinal cord-derived neural stem/progenitor cells (NSPCs) and bone marrow-derived mesenchymal stromal cells (BMSCs) following LP transplantation in SCI rats.
Setting:
Toronto Western Research Institute, Toronto, Ontario, Canada.
Methods:
NSPCs or BMSCs were transplanted via LP at level L3–5 1 week after compression SCI at T8. Rats were killed at 3, 17 and 27 days after LP transplantation and the relative distribution of cells at C4, T8 and L3–5 was quantitated. The phenotype of the NSPC and BMSC was assessed with immunocytochemistry in vitro and following LP transplantation.
Results:
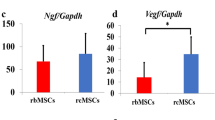
By 4 weeks, more NSPC migrated to the lesion site relative to BMSC and uninjured animals. However, there was no preferential homing of either of these types of cells into the parenchyma of the injury site, and most of the transplanted cells remained in the intrathecal space. In vitro, spinal cord-derived NSPC proliferated and expressed nestin, but after LP transplantation, NSPC became post-mitotic and primarily expressed oligodendrocyte markers. In contrast, BMSC did not express any neural antigens in vivo.
Conclusion:
LP is a minimally invasive method of cell transplantation that produces wide dissemination of cells in the subarachnoid space of the spinal cord. This is the first study to report and quantify the phenotype and spatial distribution of LP transplanted NSPC and BMSC in the intact and injured spinal cord.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Tator CH . Review of treatment trials in human spinal cord injury: issues, difficulties, and recommendations. Neurosurgery 2006; 59: 957–982; discussion 982–987.
Bakshi A, Barshinger AL, Swanger SA, Madhavani V, Shumsky JS, Neuhuber B et al. Lumbar puncture delivery of bone marrow stromal cells in spinal cord contusion: a novel method for minimally invasive cell transplantation. J Neurotrauma 2006; 23: 55–65.
Bakshi A, Hunter C, Swanger S, Lepore A, Fischer I . Minimally invasive delivery of stem cells for spinal cord injury: advantages of the lumbar puncture technique. J Neurosurg Spine 2004; 1: 330–337.
Lepore AC, Bakshi A, Swanger SA, Rao MS, Fischer I . Neural precursor cells can be delivered into the injured cervical spinal cord by intrathecal injection at the lumbar cord. Brain Res 2005; 1045: 206–216.
Jung DI, Ha J, Kang BT, Kim JW, Quan FS, Lee JH et al. A comparison of autologous and allogenic bone marrow-derived mesenchymal stem cell transplantation in canine spinal cord injury. J Neurol Sci 2009; 285: 67–77.
Shi E, Jiang X, Wang L, Akuzawa S, Nakajima Y, Kazui T . Intrathecal injection of hepatocyte growth factor gene-modified marrow stromal cells attenuates neurologic injury induced by transient spinal cord ischemia in rabbits. Anesthesiology 2010; 113: 1109–1117.
Takahashi Y, Tsuji O, Kumagai G, Hara CM, Okano HJ, Miyawaki A et al. Comparative study of methods for administering neural stem/progenitor cells to treat spinal cord injury in mice. Cell Transplant 2010.
Pluchino S, Quattrini A, Brambilla E, Gritti A, Salani G, Dina G et al. Injection of adult neurospheres induces recovery in a chronic model of multiple sclerosis. Nature 2003; 422: 688–694.
Weiss S, Dunne C, Hewson J, Wohl C, Wheatley M, Peterson AC et al. Multipotent CNS stem cells are present in the adult mammalian spinal cord and ventricular neuroaxis. J Neurosci 1996; 16: 7599–7609.
Kulbatski I, Mothe AJ, Keating A, Hakamata Y, Kobayashi E, Tator CH . Oligodendrocytes and radial glia derived from adult rat spinal cord progenitors: morphological and immunocytochemical characterization. J Histochem Cytochem 2007; 55: 209–222.
Martens DJ, Seaberg RM, van der Kooy D . In vivo infusions of exogenous growth factors into the fourth ventricle of the adult mouse brain increase the proliferation of neural progenitors around the fourth ventricle and the central canal of the spinal cord. Eur J Neurosci 2002; 16: 1045–1057.
Mothe AJ, Tator CH . Transplanted neural stem/progenitor cells generate myelinating oligodendrocytes and Schwann cells in spinal cord demyelination and dysmyelination. Exp Neurol 2008; 213: 176–190.
Parr AM, Kulbatski I, Zahir T, Wang X, Yue C, Keating A et al. Transplanted adult spinal cord-derived neural stem/progenitor cells promote early functional recovery after rat spinal cord injury. Neuroscience 2008; 155: 760–770.
Ohta M, Suzuki Y, Noda T, Ejiri Y, Dezawa M, Kataoka K et al. Bone marrow stromal cells infused into the cerebrospinal fluid promote functional recovery of the injured rat spinal cord with reduced cavity formation. Exp Neurol 2004; 187: 266–278.
Clark BR, Jamieson C, Keating A . Human long-term bone marrow culture. In: Walker JM, Pollard JW (eds). Basic Cell Culture Protocols, 2nd edn. The Humana Press Inc: Totowa, NJ, pp 75:249–256, 1997.
Parr AM, Kulbatski I, Wang XH, Keating A, Tator CH . Fate of transplanted adult neural stem/progenitor cells and bone marrow-derived mesenchymal stromal cells in the injured adult rat spinal cord and impact on functional recovery. Surg Neurol 2008; 70: 600–607; discussion 607.
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006; 8: 315–317.
Rivlin AS, Tator CH . Effect of duration of acute spinal cord compression in a new acute cord injury model in the rat. Surg Neurol 1978; 10: 38–43.
De la Calle JL, Paino CL . A procedure for direct lumbar puncture in rats. Brain Res Bull 2002; 59: 245–250.
Mothe AJ, Kulbatski I, Parr A, Mohareb M, Tator CH . Adult spinal cord stem/progenitor cells transplanted as neurospheres preferentially differentiate into oligodendrocytes in the adult rat spinal cord. Cell Transplant 2008; 17: 735–751.
Ankeny DP, McTigue DM, Jakeman LB . Bone marrow transplants provide tissue protection and directional guidance for axons after contusive spinal cord injury in rats. Exp Neurol 2004; 190: 17–31.
Chopp M, Zhang XH, Li Y, Wang L, Chen J, Lu D et al. Spinal cord injury in rat: treatment with bone marrow stromal cell transplantation. Neuroreport 2000; 11: 3001–3005.
Woodbury D, Schwarz EJ, Prockop DJ, Black IB . Adult rat and human bone marrow stromal cells differentiate into neurons. J Neurosci Res 2000; 61: 364–370.
Gelderd JB, Chopin SF . The vertebral level of origin of spinal nerves in the rat. Anat Rec 1977; 188: 45–47.
Acknowledgements
This work was supported by the Canadian Institutes of Health Research (CIHR NET), the Multiple Sclerosis Society of Canada, and by the Canadian Paraplegic Association (Ontario Branch). We thank Kurt van Bendegem for preparing the digitized schematic diagram in Figure 1. We also thank Linda Lee and Rita van Bendegem for tissue processing and Kenny Zhan for help with data analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Mothe, A., Bozkurt, G., Catapano, J. et al. Intrathecal transplantation of stem cells by lumbar puncture for thoracic spinal cord injury in the rat. Spinal Cord 49, 967–973 (2011). https://doi.org/10.1038/sc.2011.46
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sc.2011.46
Keywords
This article is cited by
-
The Effect of Different Routes of Xenogeneic Mesenchymal Stem Cell Transplantation on the Regenerative Potential of Spinal Cord Injury
Regenerative Engineering and Translational Medicine (2023)
-
Pretreatment of Mouse Neural Stem Cells with Carbon Monoxide-Releasing Molecule-2 Interferes with NF-κB p65 Signaling and Suppresses Iron Overload-Induced Apoptosis
Cellular and Molecular Neurobiology (2016)
-
Comparison of intraspinal and intrathecal implantation of induced pluripotent stem cell-derived neural precursors for the treatment of spinal cord injury in rats
Stem Cell Research & Therapy (2015)
-
Diffusion Efficiency and Bioavailability of Resveratrol Administered to Rat Brain by Different Routes: Therapeutic Implications
Neurotherapeutics (2015)
-
Comparison of functional and histological outcomes after intralesional, intracisternal, and intravenous transplantation of human bone marrow-derived mesenchymal stromal cells in a rat model of spinal cord injury
Acta Neurochirurgica (2013)