Abstract
Study design:
Case–control study.
Objectives:
To assess serum myostatin levels, bone mineral density (BMD), appendicular skeletal muscle mass (ASMM) and serum sclerostin levels in chronic spinal cord injured (SCI) patients and healthy controls.
Setting:
SCI centre in Italy.
Methods:
Blood samples, whole-body bioelectrical impedance analysis and BMD measurement with the ultrasound technique at the calcaneus level were taken from patients suffering from chronic SCI (both motor complete and incomplete) and healthy control subjects.
Results:
A total of 28 SCI patients and 15 healthy controls were enrolled. Serum myostatin levels were statistically higher (P<0.01) in SCI patients compared with healthy controls. Similar results were found comparing both the motor complete and the motor incomplete SCI subgroups to healthy controls. Serum sclerostin was significantly higher in patients with SCI compared with healthy controls (P<0.01). BMD, stiffness and mean T-score values in SCI patients were significantly lower than those in healthy controls. Serum myostatin concentrations in the motor complete SCI subgroups correlated only with serum sclerostin levels (r2=0.42; P=0.001) and ASMM (r2=0.70; P=0.002) but not in healthy controls.
Discussion:
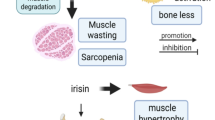
Serum myostatin and serum sclerostin are significantly higher in chronic SCI patients compared with healthy controls. They are potential biomarkers of muscle and bone modifications after SCI. This is the first study reporting an increase in serum myostatin in patients suffering from chronic SCI and a correlation with ASMM.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Morse LR, Battaglino RA, Stolzmann KL, Hallett LD, Waddimba A, Gagnon D et al. Osteoporotic fractures and hospitalization risk in chronic spinal cord injury. Osteoporos Int 2009; 20: 385–392.
Morse LR, Geller A, Battaglino RA, Stolzmann KL, Matthess K, Lazzari AA et al. Barriers to providing dual energy x-ray absorptiometry services to individuals with spinal cord injury. Am J Phys Med Rehabil 2009; 88: 57–60.
Khosla S . Minireview: the OPG/RANKL/RANK system. Endocrinology 2001; 142: 5050–5055.
Hauschka PV, Lian JB, Cole DE, Gundberg CM . Osteocalcin and matrix Gla protein: vitamin K-dependent proteins in bone. Physiol Rev 1989; 69: 990–1047.
Patterson-Buckendahl P . Osteocalcin is a stress-responsive neuropeptide. Endocr Regul 2011; 45: 99–110.
Morse LR, Sudhakar S, Danilack V, Tun C, Lazzari A, Gagnon DR et al. Association between sclerostin and bone density in chronic spinal cord injury. J Bone Miner Res 2012; 27: 352–359.
Morse LR, Sudhakar S, Lazzari AA, Tun C, Garshick E, Zafonte R et al. Sclerostin: a candidate biomarker of SCI-induced osteoporosis. Osteoporos Int 2013; 24: 961–968.
Garshick E, Kelley A, Cohen SA, Garrison A, Tun CG, Gagnon D et al. A prospective assessment of mortality in chronic spinal cord injury. Spinal Cord 2005; 43: 408–416.
Carda S, Cisari C, Invernizzi M . Sarcopenia or muscle modifications in neurologic diseases: a lexical or patophysiological difference? Eur J Phys Rehabil Med 2013; 49: 119–130.
Schoenau E . From mechanostat theory to development of the ‘Functional Muscle-Bone-Unit’. J Musculoskelet Neuronal Interact 2005; 5: 232–238.
Cianferotti L, Brandi ML . Muscle-bone interactions: basic and clinical aspects. Endocrine 2014; 45: 165–177.
Mo C, Romero-Suarez S, Bonewald L, Johnson M, Brotto M . Prostaglandin E2: from clinical applications to its potential role in bone- muscle crosstalk and myogenic differentiation. Recent Pat Biotechnol 2012; 6: 223–229.
Brotto M . Editorial: welcome to our Recent Patents on Biotechnology Special Edition on skeletal muscle diseases!. Recent Pat Biotechnol 2012; 6: 155–156.
Brotto M, Abreu EL . Sarcopenia: pharmacology of today and tomorrow. J Pharmacol Exp Ther 2012; 343: 540–546.
Huang J, Hsu YH, Mo C, Abreu E, Kiel DP, Bonewald LF et al. METTL21C is a Potential Pleiotropic Gene for Osteoporosis and Sarcopenia Acting through the Modulation of the NFkappaB Signaling Pathway. J Bone Miner Res 2014; 29: 1531–1540.
Marimuthu K, Murton AJ, Greenhaff PL . Mechanisms regulating muscle mass during disuse atrophy and rehabilitation in humans. J Appl Physiol 2011; 110: 555–560.
Lang T, Streeper T, Cawthon P, Baldwin K, Taaffe DR, Harris TB . Sarcopenia: etiology, clinical consequences, intervention, and assessment. Osteoporos Int 2009; 21: 543–559.
American Spinal Injury Association International Standards for Neurological Classification of Spinal Cord Injury. Revised 2011. American Spinal Injury Association: Atlanta, GA, USA. 2011.
Ditunno JF Jr., Young W, Donovan WH, Creasey G . The international standards booklet for neurological and functional classification of spinal cord injury. American Spinal Injury Association. Paraplegia 1994; 32: 70–80.
Kirshblum SC, Memmo P, Kim N, Campagnolo D, Millis S . American Spinal Injury Association. Comparison of the revised 2000 American Spinal Injury Association classification standards with the 1996 guidelines. Am J Phys Med Rehabil 2002; 81: 502–505.
Invernizzi M, Carda S, Milani P, Mattana F, Fletzer D, Iolascon G et al. Development and validation of the Italian version of the Spinal Cord Independence Measure III. Disabil Rehabil 2010; 32: 1194–1203.
Lukaski HC, Johnson PE, Bolanchuk WW, Lykken GI . Assessment of fat-free mass using bioelectrical impedance measurement of the human body. Am J Clin Nutr 1985; 41: 810–817.
Kyle UG, Genton L, Hans D, Pichard C . Validation of a bioelectrical impedance analysis equation to predict appendicular skeletal muscle mass (ASMM). Clin Nutr 2003; 22: 537–543.
Melton LJ 3rd, Khosla S, Crowson CS, O’Connor MK, O’Fallon WM, RIggs BL . Epidemiology of sarcopenia. J Am Geriatr Soc 2000; 48: 625–630.
Fleming TR, Powers JH . Biomarkers and surrogate endpoints in clinical trials. Stat Med 2012; 31: 2973–2984.
Grimby G, Broberg C, Krotkiewska I, Krotkiewski M . Muscle fiber composition in patients with traumatic cord lesion. Scand J Rehabil Med 1976; 8: 37–42.
Mohr T, Andersen JL, Biering-Sorensen F, Galbo H, Bangsbo J, Wagner A et al. Long-term adaptation to electrically induced cycle training in severe spinal cord injured individuals. Spinal Cord 1997; 35: 1–16.
Amato AA, Sivakumar K, Goyal N, Davis WS, Salajegheb M, Praestgaard J et al. Treatment of sporadic inclusion body myositis with bimagrumab. Neurology 2014; 83: 2239–2246.
Giangregorio L, McCartney N . Bone loss and muscle atrophy in spinal cord injury: epidemiology, fracture prediction, and rehabilitation strategies. J Spinal Cord Med 2006; 29: 489–500.
Castro MJ, Apple DF Jr., Hillegass EA, Dudley GA . Influence of complete spinal cord injury on skeletal muscle cross-sectional area within the first 6 months of injury. Eur J Appl Physiol Occup Physiol 1999; 80: 373–378.
Drummond MJ, Glynn EL, Lujan HL, Dicarlo SE, Rasmussen BB . Gene and protein expression associated with protein synthesis and breakdown in paraplegic skeletal muscle. Muscle Nerve 2008; 37: 505–513.
Zhang D, Liu M, Ding F, Gu X . Expression of myostatin RNA transcript and protein in gastrocnemius muscle of rats after sciatic nerve resection. J Muscle Res Cell Motil 2006; 27: 37–44.
Léger B, Senese R, Al-Khodairy AW, Dériaz O, Gobelet C, Giacobino J-P et al. Atrogin-1, MuRF1, and FoXO, as well as phosphorylated GSK-3β and 4E-BP1 are reduced in skeletal muscle of chronic spinal cord-injured patients. Muscle Nerve 2009; 40: 69–78.
Mirza FS, Padhi ID, Raisz LG, Lorenzo JA . Serum sclerostin levels negatively correlate with parathyroid hormone levels and free estrogen index in postmenopausal women. J Clin Endocrinol Metab 2010; 95: 1991–1997.
Lewiecki EM . Sclerostin: a novel target for intervention in the treatment of osteoporosis. Discov Med 2011; 12: 263–273.
Lewiecki EM . Sclerostin monoclonal antibody therapy with AMG 785: a potential treatment for osteoporosis. Expert Opin Biol Ther 2011; 11: 117–127.
Papapoulos SE . Targeting sclerostin as potential treatment of osteoporosis. Ann Rheum Dis 2011; 70 (): i119–i122.
Warden SJ, Bennell KL, Matthews B, Brown DJ, McMeeken JM, Wark JD . Quantitative ultrasound assessment of acute bone loss following spinal cord injury: a longitudinal pilot study. Osteoporos Int 2002; 13: 586–592.
Jiang SD, Dai LY, Jiang LS . Osteoporosis after spinal cord injury. Osteoporos Int 2006; 17: 180–192.
NOF. National Osteoporosis Foundation Clinician's Guide to Prevention and Treatment of Osteoporosis http://nof.org/hcp/clinicians-guide 2013.
Hollaender R, Hartl F, Krieg MA, Tyndall A, Geuckel C, Buitrago-Tellez C et al. Prospective evaluation of risk of vertebral fractures using quantitative ultrasound measurements and bone mineral density in a population-based sample of postmenopausal women: results of the Basel Osteoporosis Study. Ann Rheum Dis 2009; 68: 391–396.
Khaw KT, Reeve J, Luben R, Bingham S, Welch A, Wareham N et al. Prediction of total and hip fracture risk in men and women by quantitative ultrasound of the calcaneus: EPIC-Norfolk prospective population study. Lancet 2004; 363: 197–202.
Gonnelli S, Cepollaro C, Montagnani A, Martini S, Gennari L, Mangeri M et al. Heel ultrasonography in monitoring alendronate therapy: a four-year longitudinal study. Osteoporos Int 2002; 13: 415–421.
Spungen AM, Wang J, Pierson RN Jr, Bauman WA . Soft tissue body composition differences in monozygotic twins discordant for spinal cord injury. J Appl Physiol 2000; 88: 1310–1315.
Modlesky CM, Bickel CS, Slade JM, Meyer RA, Cureton JK, Dudley GA . Assessment of skeletal muscle mass in men with spinal cord injury using dual-energy X-ray absorptiometry and magnetic resonance imaging. J Appl Physiol 2004; 96: 561–565.
Osterthun R, Post MW, van Asbeck FW, van Leeuwen CM, van Koppenhagen CF . Causes of death following spinal cord injury during inpatient rehabilitation and the first five years after discharge. A Dutch cohort study. Spinal Cord 2014; 52: 483–488.
Shields RK, Dudley-Javoroski S . Musculoskeletal adaptations in chronic spinal cord injury: effects of long-term soleus electrical stimulation training. Neurorehabil Neural Repair 2007; 21: 169–179.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest
Rights and permissions
About this article
Cite this article
Invernizzi, M., Carda, S., Rizzi, M. et al. Evaluation of serum myostatin and sclerostin levels in chronic spinal cord injured patients. Spinal Cord 53, 615–620 (2015). https://doi.org/10.1038/sc.2015.61
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sc.2015.61
This article is cited by
-
Association of serum sclerostin levels with marrow adiposity in postmenopausal women with glucocorticoid-induced osteoporosis
BMC Endocrine Disorders (2024)
-
Endocrinological and inflammatory markers in individuals with spinal cord injury: A systematic review and meta-analysis
Reviews in Endocrine and Metabolic Disorders (2022)
-
Association of serum bone- and muscle-derived factors with age, sex, body composition, and physical function in community-dwelling middle-aged and elderly adults: a cross-sectional study
BMC Musculoskeletal Disorders (2019)
-
Mitochondrial health and muscle plasticity after spinal cord injury
European Journal of Applied Physiology (2019)
-
Osteoporosis in Veterans with Spinal Cord Injury: an Overview of Pathophysiology, Diagnosis, and Treatments
Clinical Reviews in Bone and Mineral Metabolism (2019)