Abstract
Synthesis of a half metallic material on a substrate is highly desirable for diverse applications. Herein, we have investigated structural, adsorptive and magnetic properties of metal free graphitic carbon nitride (g-C4N3) layer on hexagonal BN layer (h-BN) using the optB88-vdW van der Waals density functional theory. It is found that g-C4N3 layer can be adsorbed on BN layer due to the change of lattice constant of the hybridized system. The newly found lattice constant of g-C4N3 was 9.89 Å, which is approximately 2% lower and larger than to those of free standing BN and g-C4N3, respectively. Also, 2 × 2 surface reconstruction geometry predicted in free standing g-C4N3 layer disappears on the BN layer. Interestingly, we have found that metal free half metallic behavior in g-C4N3 can be preserved even on BN layer and the characters of spin polarized planar orbitals suggest that our theoretical prediction can be verified using normal incidence of K-edge X-ray magnetic circular dichroism (XMCD) measurement.
Similar content being viewed by others
3d transition metal elements are believed to be essential to introduce magnetism. However, many studies showed that magnetism could occur in the absence of the 3d transition metal elements and it is referred to as d0 magnetism. Examples of d0 magnetism from various types of oxide materials1,2 and semiconductors3,4 have been reported and in most of discussions the d0 magnetism is attributed to the effect of vacancy defects or lattice distortion. Half metallic magnetism also received great research interest because half metallic materials have various potential applications in spintronics. In most of previous studies on half metallic systems, the combination of various types of materials with 3d transition metal elements has been investigated5,6. Besides the magnetism of conventional 3d systems, 2p electron systems also yield another interesting property. Usually, 2p electron systems are known to display non-magnetic behavior in bulk state. Nonetheless, further studies in two dimensional structures have indicated that magnetic state is likely to be achieved in low dimensional geometry. For instance, many reports have shown the development of magnetic state in graphene or BN; the influence of vacancy defect or adatom defect has been discussed7,8. Here, the local magnetic moment has been discussed in most cases.
For spintronics, it is of interest to examine ferromagnetism in pure 2p electron systems in the absence of transition metal elements because large spin relaxation time is expected owing to the relatively weak spin-orbit coupling. Hence, the investigation of biocompatible material with half metallic properties of particular interest. Among various candidates, C-N based materials show great potential. In this respect, considerable efforts have been devoted to polymetric graphitic carbon nitride (g-C3N4)9,10,11. This structure features intrinsic vacancies that are expected to produce spin polarized state. However, g-C3N4 displays non-magnetic properties. Recently, a similar material g-C4N3 in the framework of g-C3N4 has been synthesized12. This metal free material showed half metallic properties in the free standing state, as reported by Du et al13,14,15,16. Additionally, the authors claimed that the g-C4N3 has a (2 × 2) surface reconstructed geometry due to a buckling feature in the free standing layer.
It is highly desirable to synthesize of a half metallic material on a substrate for diverse applications because one can avoid various difficulties of manipulating free standing layer. It is therefore necessary to choose a supporting material that does not disturb the intrinsic half metallic properties of g-C4N3. However, investigation of half metallic properties of metal-free g-C4N3 on a supporting material has yet to be reported. Herein, we for the first time investigate structural, adsorption and magnetic properties of g-C4N3/BN.
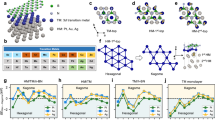
Vienna ab initio simulation package (VASP) was used. Herein, the van der Waals interaction was included. In particular, van der Waals interaction of optB88-vdW method17,18,19 was employed to investigate the physical properties of g-C4N3/BN. A vacuum distance of 15 Å was applied along the perpendicular direction to the film surface. All calculations were performed based on using 9 × 9 × 1 k-points and a kinetic energy of 700 eV cutoff. The most stable lattice constant of g-C4N3/BN was investigated by calculating the total energy as a function of lattice parameter. For the adsorptive and magnetic properties of hybridized g-C4N3/BN system, we have considered a (2 × 2) g-C4N3 layer onto a (4 × 4) BN layer. Structure optimization was performed via force and energy minimization process. In addition, ferromagnetic (FM), antiferromagnetic (AFM) and non-magnetic spin configurations were considered for the magnetic ground state. It is worth noting that the lattice constant of (1 × 1) g-C3N4 unit cell is 4.74 Å, but that of (1 × 1) g-C4N3 is 4.84 Å. Moreover, the g-C4N3 is known to have a (2 × 2) reconstructured surface geometry, thus the ground state lattice constant becomes 9.68 Å. This suggests that the lattice constant of g-C4N3 is likely to be modified in the presence of external effect. Consequently, we will explore the most stable lattice structure of g-C4N3/BN hybridized system by changing its lattice constant.
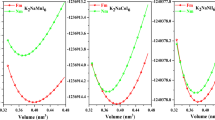
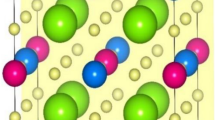
Fig. 1 and Fig. 2 show the schematic configurations of g-C4N3/BN systems featuring 6 different adsorption sites. The green and pink spheres represent the N, B atom in BN layer, respectively, while the gray and blue spheres represent the C and N atoms in g-C4N3 layer, respectively. The (1 × 1) unit cell of g-C4N3 is depicted in Fig. 1(g); the C and N atoms that are located on the hexagonal ring are referred as C1 and N1, respectively, whereas C atom located outside the ring is denoted as C2. It should be noted that the bridge adsorption site is not observed because the atoms orient themselves into the configuration shown in Fig. 1(b), regardless of the initial calculations. Table I presents the calculated total energy differences among ferromagnetic (FM) and antiferromagnetic (AFM) spin arrangements and the distances between BN and g-C4N3 layers. The results were based on the most stable lattice constant. The total energy for configuration depicted in Fig. 1(a) was set to zero as a reference energy that produced the most stable system. The interlayer distance for this configuration was 3.18 Å. Interestingly, the FM ground state was still preserved even in the BN underlayer and was independent of the adsorption sites. It is worth noting that the energy difference for configuration 1(a) between FM and non-magnetic states was 278 meV. Fig. 3 shows the total energy curve as a function of the lattice constant for the configuration Fig. 1(a) in which the ground state was realized. The solid line represents the fitting curve obtained with a parabolic polynomial function. A total energy minimum was observed at a lattice constant of 9.89 Å. The ideal monolayer of BN in a (4 × 4) unit cell has a lattice constant of 10.08 Å and that of free standing g-C4N3 in a (2 × 2) unit cell is 9.68 Å. Therefore, a newly found lattice constant of g-C4N3 was 9.89 Å, which is approximately 2% lower and larger than to those of free standing BN and g-C4N3, respectively.
We now discuss the adsorption energy. The adsorption energy (Eads) was calculated as:  , where EBN and
, where EBN and  denote the total energies of the free standing systems and
denote the total energies of the free standing systems and  represents the total energy of the hybridized system. To calculate the total energy of free standing layers, the corresponding lattice constants of (2 × 2) unit cell for g-C4N3 and (4 × 4) unit cell for BN were used. The calculated total energies of the free standing BN and g-C4N3 are 248.685 eV and 199.699 eV, respectively. The horizontal line in Fig. 2 represents the sum of the energies of the free standing systems, i.e.
represents the total energy of the hybridized system. To calculate the total energy of free standing layers, the corresponding lattice constants of (2 × 2) unit cell for g-C4N3 and (4 × 4) unit cell for BN were used. The calculated total energies of the free standing BN and g-C4N3 are 248.685 eV and 199.699 eV, respectively. The horizontal line in Fig. 2 represents the sum of the energies of the free standing systems, i.e. . Thus, the total energy of the g-C4N3/BN hybridized system should be below the horizontal line. Based on the calculated total energy curve in Fig. 3, the g-C4N3/BN hybridized system is unstable when the g-C4N3/BN structure has a lattice constant similar to that of either BN or g-C4N3 because the resulting total energy is larger than the sum of the free standing systems energies. The total energy minimum of the hybridized system was obtained at a lattice constant of 9.89 Å, which corresponds to a 2% contraction and expansion with respect to that of BN and g-C4N3, respectively. The ground state lattice constant obtained herein is comparable with the calculated average of the respective lattice constants of the free standing g-C4N3 and BN layers; the lattice changes enables adsorption of g-C4N3 layer on the BN layer. The calculated adsorption energy was 0.67 eV. Another interesting aspect is the structural behavior of the g-C4N3/BN system. According to previous theoretical prediction13, the free standing g-C4N3 surface is believed to show a (2 × 2) reconstructed geometry owing to surface buckling. However, herein, we observed that the surface reconstruction is absent on the BN layer and the g-C4N3 layer becomes flat layer with the interlayer spacings presented in Table I. We have found using Bader charge analysis20,21,22,23 that no significant charge transfer takes place between two layers. However, the van der Waals interaction between these two layers significantly influences the surface structure. Owing to this geometric effect, the lattice constant of g-C4N3/BN will be 4.945 Å in a (1 × 1) unit cell.
. Thus, the total energy of the g-C4N3/BN hybridized system should be below the horizontal line. Based on the calculated total energy curve in Fig. 3, the g-C4N3/BN hybridized system is unstable when the g-C4N3/BN structure has a lattice constant similar to that of either BN or g-C4N3 because the resulting total energy is larger than the sum of the free standing systems energies. The total energy minimum of the hybridized system was obtained at a lattice constant of 9.89 Å, which corresponds to a 2% contraction and expansion with respect to that of BN and g-C4N3, respectively. The ground state lattice constant obtained herein is comparable with the calculated average of the respective lattice constants of the free standing g-C4N3 and BN layers; the lattice changes enables adsorption of g-C4N3 layer on the BN layer. The calculated adsorption energy was 0.67 eV. Another interesting aspect is the structural behavior of the g-C4N3/BN system. According to previous theoretical prediction13, the free standing g-C4N3 surface is believed to show a (2 × 2) reconstructed geometry owing to surface buckling. However, herein, we observed that the surface reconstruction is absent on the BN layer and the g-C4N3 layer becomes flat layer with the interlayer spacings presented in Table I. We have found using Bader charge analysis20,21,22,23 that no significant charge transfer takes place between two layers. However, the van der Waals interaction between these two layers significantly influences the surface structure. Owing to this geometric effect, the lattice constant of g-C4N3/BN will be 4.945 Å in a (1 × 1) unit cell.
For the configuration depicted in Fig. 1(g), magnetic moments of N1 and C2 are 0.26 μB and 0.028 μB, respectively. The calculated total magnetic moment per unit cell is 4 μB, indicating the half metallic nature of g-C4N3. Fig. 4 shows the electronic band structure. The valence bands of majority spin states are located below the Fermi level, whereas the minority spin bands cross the Fermi level. These finding confirms that the half metallic nature of the metal free g-C4N3/BN system. Fig. 5(a) and (b) show the m-resolved density of states (m-DOS). As observed, the spin polarized state mainly originates from the px,y orbitals of the N1 atoms in the g-C4N3 layer. In contrast, no significant spin polarization was found in the pz orbital. Moreover, this result indicates that the top of the valence involves the planar px,y orbitals. Fig. 6 illustrates the calculated spin polarized charge density of g-C4N3/BN. As inferred, the planar px,y orbitals contribute to the magnetic moment. Additionally, observed spin polarization in planar orbitals indicates that the K-edge X-ray magnetic circular dichroism (XMCD) measurment, using normal incidence beam, can verify our predictions because the magnetism of g-C4N3/BN originates from the 2px,y orbital. In contrast, no XMCD signals would be detected with a grazing incidence beam because the selection Δm = ±1 is not satisfied.
In conclusion, we explored adsorption, structural and magnetic properties of g-C4N3/BN. The hybridized system had a new lattice constant, which is different from those of free standing BN and g-C4N3 layers and this lattice change was responsible for adsorption of g-C4N3 on BN layer. In addition, the surface reconstruction feature found in free standing layer disappeared due to the interaction with BN layer. The magnetic moment arises from the N atoms in g-C4N3 layer, whereas the C atoms show almost negligible spin polarization. Very interestingly, half metallic behavior is preserved even on BN layer and the half metallicity originates mainly from the 2px,y planar orbitals of N atoms in g-C4N3 layer. We suggest that this spin polarization from 2px,y orbitals can be verified with the K-edge XMCD measurement using normal incidence beam. Overall, this metal free half metallic behavior even on a supporting material will enable the development of new and intriguing properties that show potential for next generation spintronics.
References
Venkatesan, M., Fitzgerald, C. B. & Coey, J. M. D. Unexpected magnetism in a dielectric oxide. Nature 430, 630 (2004).
Kim, D., Yang, J. & Hong, J. Ultrathin half metallic N and antiferromagnetic semiconducting C layers on MgO(001). J. Phys. Condens. Matter 22, 486006 (2010).
Hong, N. H., Sakai, J., Poirot, N. & Brize, V. Room-temperature ferromagnetism observed in undoped semiconducting and insulating oxide thin films. Phys. Rev. B 73, 132404 (2006).
Kim, D., Hong, J., Park, Y. & Kim, K. The origin of oxygen vacancy induced ferromagnetism in undoped TiO2 . J. Phys. Condens. Matter 21, 195405 (2009).
Du, C. et al. Control of Magnetocrystalline Anisotropy by Epitaxial Strain in Double Perovskite Sr2FeMoO6 Films. Phys. Rev. Lett. 110, 147204 (2013).
deGroot, R. A., Mueller, F. M., van Engen, P. G. & Buschow, K. H. J. New class of materials: Half-metallic ferromagnets. Phys. Rev. Lett. 50, 2024–2027 (1983).
Yang, J., Kim, D., Hong, J. & Qian, X. Magnetism in boron nitride:Adatom and vacancy defect. Surf. Sci. 604, 1603–1607 (2010).
Si, M. S. & Xue, D. S. Magnetic properties of vacancies in a graphitic boron nitride sheet by first-principles pseudopotential calculations. Phys. Rev. B 75, 193409 (2007).
Stolbov, S. & Zuluaga, S. Sulfur doping effects on the electronic and geometric structures of graphitic carbon nitride photocatalyst: insights from first principles. J. Phys. Condens. Matter. 25, 085507 (2013).
Wei, W. & Jacob, T. Strong excitonic effects in the optical properties of graphitic carbon nitride g-C3N4 from first principles. Phys. Rev. B. 87, 085202 (2013).
Wang, X. et al. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat. Materials. 8, 76–80 (2009).
Lee, J. S., Wang, X., Luo, H. & Dai, S. Fluidic Carbon Precursors for Formation of Functional Carbon under Ambient Pressure Based on Ionic Liquids. Adv. Mater. 22, 1004–1007 (2010).
Du, A., Sanvito, S. & Smith, S. C. First-Principles Prediction of Metal-Free Magnetism and Intrinsic Half-Metallicity in Graphitic Carbon Nitride. Phys. Rev. Lett. 108, 197207 (2012).
Li, X., Zhou, J., Wang, Q., Kawazoe, Y. & Jena, P. Patterning Graphitic C-N Sheets into a Kagome Lattice for Magnetic Materials. J. Phys. Chem. Lett. 4, 259–263 (2013).
Li, X., Zhang, S. & Wang, Q. Stability and physical properties of a tri-ring based porous g-C4N3 sheet. Phys. Chem. Chem. Phys. 15, 7142–7146 (2013).
Wu, M., Wang, Q., Sun, Q. & Jena, P. Functionalized Graphitic Carbon Nitride for Efficient Energy Storage. J. Phys. Chem. C. 117, 6055–6059 (2013).
Dion, M., Rydberg, H., Schroder, E., Langreth, D. C. & Lundqvist, B. I. Van der Waals Density Functional for General Geometries. Phys. Rev. Lett. 92, 246401 (2004).
Graziano, G., Klimes, J., Fernadez-Alonso, F. & Michaelides, A. Improved description of soft layered materials with van der Waals density functional theory. J. Phys. Condens. Matter. 24, 424216 (2012).
Liu, W. et al. Benzene adsorbed on metals: Concerted effect of covalency and van der Waals bonding. Phys. Rev. B. 86, 245405 (2012).
Bader, R. F. W. Atoms in Molecules: A Quantum Theory (Oxford University Press, 1990).
Henkelman, G., Arnaldsson, A. & Jonsson, H. A fast and robust algorithm for Bader decomposition of charge density. Comput. Mater. Sci. 36, 354–360 (2006).
Sanville, E., Kenny, S. D., Smith, R. & Henkelman, G. Improved grid-based algorithm for Bader charge allocation. J. Comput. Chem. 28, 899–908 (2007).
Tang, W., Sanville, E. & Henkelman, G. A grid-based Bader analysis algorithm without lattice bias. J. Phys. Condens. Matter. 21, 084204–084207 (2009).
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea(NRF) funded by the Ministry of Education, Science and Technology(No. 2013R1A1A2006071).
Author information
Authors and Affiliations
Contributions
J.H. reviewed and wrote the manuscript. A.H. prepared figures 1–6.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Hashmi, A., Hong, J. Metal free half metallicity in 2D system: structural and magnetic properties of g-C4N3 on BN. Sci Rep 4, 4374 (2014). https://doi.org/10.1038/srep04374
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep04374
This article is cited by
-
Zigzag boron nitride nanoribbon doped with carbon atom for giant magnetoresistance and rectification behavior based nanodevices
Scientific Reports (2024)
-
Evidencing the existence of exciting half-metallicity in two-dimensional TiCl3 and VCl3 sheets
Scientific Reports (2016)
-
Anisotropic bias dependent transport property of defective phosphorene layer
Scientific Reports (2015)
-
Transparent half metallic g-C4N3 nanotubes: potential multifunctional applications for spintronics and optical devices
Scientific Reports (2014)