Summary:
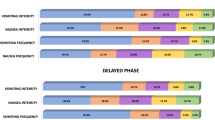
The serotonin type-3 (5-HT3) antagonists represent a significant advance in the prevention of acute nausea and vomiting (N/V) from highly emetogenic chemotherapy. We sought to determine if any differences in efficacy or adverse effects exist between two such agents, ondansetron and granisetron, during conditioning therapy for hematopoietic stem cell transplantation (HSCT). Patients were randomized to receive either ondansetron 0.15 mg/kg intravenously every 8 h or granisetron 10 μg/kg intravenously daily. Additionally, all patients received scheduled dexamethasone and lorazepam. Prophylaxis was continued until 24 h after completion of chemotherapy. Nausea and distress were measured subjectively with visual analog scales and emetic episodes were quantified. Of the 110 randomized patients, 96 were evaluable for efficacy and safety. No significant differences in efficacy were observed between the ondansetron- and granisetron-treated patients, evaluated by comparing the degree of nausea and distress, number of emetic episodes and overall control of emesis. The adverse effects were also comparable and no patients were removed from study because of severe toxicities. This trial demonstrates that ondansetron and granisetron are equally effective at preventing acute N/V associated with conditioning therapy frequently used for HSCT. The agent of choice should be based on drug acquisition cost or preference.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hesketh PJ . Comparative review of 5-HT3 receptor antagonists in the treatment of acute chemotherapy-induced nausea and vomiting. Cancer Invest 2000; 18: 163–173.
Navari R, Gandara D, Hesketh P et al. Comparative clinical trial of granisetron and ondansetron in the prophylaxis of cisplatin-induced emesis. J Clin Oncol 1995; 13: 1242–1248.
Gralla RJ, Navari RM, Hesketh PJ et al. Single-dose oral granisetron has equivalent antiemetic efficacy to intravenous ondansetron for highly emetogenic cisplatin-based chemotherapy. J Clin Oncol 1998; 16: 1568–1573.
Ruff P, Paska W, Goedhals L et al. Ondansetron compared with granisetron in the prophylaxis of cisplatin-induced acute emesis: a multicentre double-blind, randomised, parallel-group study. The Ondansetron and Granisetron Emesis Study Group. Oncology 1994; 51: 113–118.
Ondansetron versus granisetron, both combined with dexamethasone, in the prevention of cisplatin-induced emesis. Italian Group for Antiemetic Research. Ann Oncol 1995; 6: 805–810.
Audhuy B, Cappelaere P, Martin M et al. A double-blind, randomised comparison of the anti-emetic efficacy of two intravenous doses of dolasetron mesilate and granisetron in patients receiving high dose cisplatin chemotherapy. Eur J Cancer 1996; 32A: 807–813.
Hesketh P, Navari R, Grote T et al. Double-blind, randomized comparison of the antiemetic efficacy of intravenous dolasetron mesylate and intravenous ondansetron in the prevention of acute cisplatin-induced emesis in patients with cancer. Dolasetron Comparative Chemotherapy-induced Emesis Prevention Study Group. J Clin Oncol 1996; 14: 2242–2249.
Aapro MS, Bertoli L, Lordick F et al. Palonosetron (PALO) is effective in preventing acute and delayed chemotherapy-induced nausea and vomiting (CINV) in patients receiving highly emetogenic chemo-therapy (HEC). Support Care Cancer 2003; 11: 39 abstract A-17.
Lazarus HM, Bryson JC, Lemon E et al. Antiemetic efficacy and pharmacokinetic analyses of the serotonin antagonist ondansetron (GR38032F) during multiple-day chemotherapy with cisplatin prior to autologous bone marrow transplantation. J Natl Cancer Inst 1990; 82: 1776–1778.
Bosi A, Guidi S, Messori A et al. Ondansetron versus chlorpromazine for preventing emesis in bone marrow transplant recipients: a double-blind, randomized study. J Chemother 1993; 5: 191–196.
Agura ED, Brown MC, Schaffer R et al. Antiemetic efficacy and pharmacokinetics of intravenous ondansetron infusion during chemotherapy conditioning for bone marrow transplant. Bone Marrow Transplant 1995; 16: 213–222.
Barbounis V, Koumakis G, Vassilomanolakis M et al. A phase II study of ondansetron as antiemetic prophylaxis in patients receiving high-dose polychemotherapy and stem cell transplantation. Support Care Cancer 1995; 3: 301–306.
Fauser AA, Russ W, Bischoff M . Oral dolasetron mesilate (MDL 73,147EF) for the control of emesis during fractionated total-body irradiation and high-dose cyclophosphamide in patients undergoing allogeneic bone marrow transplantation. Support Care Cancer 1997; 5: 219–222.
Frakes LA, Brehm TL, Kosty MP et al. An all oral antiemetic regimen for patients undergoing high-dose chemotherapy with peripheral blood stem cell transplant. Bone Marrow Transplant 1997; 20: 473–478.
Birch R, Weaver CH, Carson K, Buckner CD . A randomized trial of once vs twice daily administration of intravenous granisetron with dexamethasone in patients receiving high-dose cyclophosphamide, thiotepa and carboplatin. Bone Marrow Transplant 1998; 22: 685–688.
Osowski CL, Dix SP, Lynn M et al. An open-label dose comparison study of ondansetron for the prevention of emesis associated with chemotherapy prior to bone marrow transplantation. Support Care Cancer 1998; 6: 511–517.
Perez EA, Tiemeier T, Solberg LA . Antiemetic therapy for high-dose chemotherapy with transplantation: report of a retrospective analysis of a 5-HT(3) regimen and literature review. Support Care Cancer 1999; 7: 413–424.
Abbott B, Ippoliti C, Hecth D et al. Granisetron (Kytril) plus dexamethasone for antiemetic control in bone marrow transplant patients receiving highly emetogenic chemotherapy with or without total body irradiation. Bone Marrow Transplant 2000; 25: 1279–1283.
Abang AM, Takemoto MH, Pham T et al. Efficacy and safety of oral granisetron versus i.v. granisetron in patients undergoing peripheral blood progenitor cell and bone marrow transplantation. AntiCancer Drugs 2000; 11: 137–142.
Ballen KK, Hesketh AM, Heyes C et al. Prospective evaluation of antiemetic outcome following high-dose chemotherapy with hematopoietic stem cell support. Bone Marrow Transplant 2001; 28: 1061–1066.
Kalaycio M, Mendez Z, Pohlman B et al. Continuous-infusion granisetron compared to ondansetron for the prevention of nausea and vomiting after high-dose chemotherapy. J Cancer Res Clin Oncol 1998; 124: 265–269.
Orchard PJ, Rogosheske J, Burns L et al. A prospective randomized trial of the anti-emetic efficacy of ondansetron and granisetron during bone marrow transplantation. Biol Blood Marrow Transplant 1999; 5: 386–393.
Bubalo J, Seelig F, Karbowicz S, Maziarz RT . Randomized open-label trial of dolasetron for the control of nausea and vomiting associated with high-dose chemotherapy with hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2001; 7: 439–445.
Southwestern Oncology Group (SWOG) toxicity grading criteria, available from URL. http://www.fda.gov/cder/cancer/toxicityframe.htm [accessed July 29, 2003].
Acknowledgements
We thank all of the BMT-unit physician assistants, nurses, staff and participating patients at the South Texas Veteran's Healthcare System, Audie L Murphy Division and University Hospital, San Antonio, Texas. Additionally, we would also like to thank Virginia Doyle, RPh, who managed the randomization, blinding, and drug preparation aspects of the study and Vanessa Garza, Pharm.D. who assisted with data entry. This study was supported, in part, by an unrestricted educational grant from SmithKline Beecham Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Walsh, T., Morris, A., Holle, L. et al. Granisetron vs ondansetron for prevention of nausea and vomiting in hematopoietic stem cell transplant patients: results of a prospective, double-blind, randomized trial. Bone Marrow Transplant 34, 963–968 (2004). https://doi.org/10.1038/sj.bmt.1704714
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.bmt.1704714
Keywords
This article is cited by
-
Palonosetron and dexamethasone for the prevention of nausea and vomiting in patients receiving allogeneic hematopoietic stem cell transplantation
Supportive Care in Cancer (2014)
-
Palonosetron (Aloxi®) and dexamethasone for the prevention of acute and delayed nausea and vomiting in patients receiving multiple-day chemotherapy
Supportive Care in Cancer (2009)
-
Nausea and vomiting with high-dose chemotherapy and stem cell rescue therapy: a review of antiemetic regimens
Bone Marrow Transplantation (2008)