Abstract
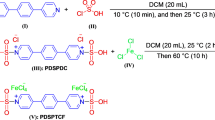
INVESTIGATIONS into the specificity of β-xylosidase required the synthesis of a number of substituted phenyl-β-D-xylosides. A very important method is the Helferich reaction in which the appropriate phenol and the fully acetylated monosaccharide are heated in the presence of an acidic catalyst. To favour the formation of xyloside-acetates with the β-anomeric configuration, p-toluene-sulphonic acid and β-tetra-O-acetyl-xylose were used1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Montgomery, E. M., Richtmeyer, N. K., and Hudson, C. S., J. Amer. Chem. Soc., 64, 690 (1942).
Vogel, A. I., A Textbook of Practical Organic Chemistry, third ed., 452 (London, Longmans, Green and Co., 1957).
Jermyn, M. A., Austral. J. Chem., 8, 403 (1955).
Loontiens, F. G., and De Bruyne, C. K., Naturwiss., 51, 359 (1964).
Thompson, A., and Wolfrom, M. L., in Methods of Carbohydrate Chemistry, 2, 215, edit. by Whistler, R. L., and Wolfrom, M. L. (New York, Academic Press, Inc., 1963).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DE BRUYNE, C., VERSELE, H. & CLAEYSSENS, M. Synthesis of Substituted Phenyl-β-D-xylo-pyranosides. Nature 205, 900 (1965). https://doi.org/10.1038/205900a0
Issue date:
DOI: https://doi.org/10.1038/205900a0