Abstract
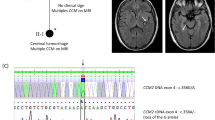
Capillary malformation (CM; ‘port-wine stain’), is a common vascular malformation affecting cutaneous capillary vessels in 0.3% of newborns. Increased incidence of lesions in first-degree relatives of these patients and several reported familial cases suggest that genetic factors may play a role in the pathogenesis of CM. We report the first genome-wide linkage analysis of familial CM. In the non-parametric linkage analysis, strong evidence of linkage (peak Z-score 6.72, P-value 0.000136) was obtained in an interval of 69 cM between markers D5S407 and D5S2098, corresponding to 5q11–5q23. Parametric linkage analysis gave a maximum combined HLOD score of 4.84 (α-value 0.67) at marker D5S2044 on 5q15, and analysis using only the linked families, defined a smaller, statistically significant locus CMC1 of 23 cM (peak LOD score 7.22) between markers D5S1962 and D5S652 corresponding to 5q13–5q15. Interesting candidate genes implicated in vascular and neural development, such as MEF2C, RASA1, and THBS4, are in this locus.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Mulliken JB, Glowacki J . Hemangiomas and vascular malformation in infants and children: A classification based on endothelial characteristics Plast Reconstr Surg 1982 69: 412–420
Jacobs AH, Walton RG . The incidence of birthmarks in the neonate Pediatrics 1976 58: 218–222
Mulliken JB, Young AE (eds). Vascular birthmarks: Hemangiomas and malformations Philadelphia: WB Saunders 1988
Pratt AG . Birthmarks in infants AMA Arch Derm Syph 1967 67: 302
Lanigan SW, Cotterill JA . Psychological disabilities amongst patients with port wine stains Br J Dermatol 1989 121: 209–215
Pasyk KA . Familial multiple lateral telangiectatic nevi (port-wine stain) Clin Genet 1992 41: 197–201
Redondo P, Vazquez-Doval F . Familial multiple nevi flammei J Am Acad Dermatol 1996 35: 769–770
van der Horst CMAM, van Eijk TGJ, de Borgie CAJM, Koster PHL, Struycken PM, Strackee SD . Hereditary port-wine stains, do they exist? Lasers Med Sci 1999 14: 238–243
Berg JN, Quaba AA, Georgantopoulou A, Porteous ME . A family with hereditary port wine stain J Med Genet 2000 37: 12
Mills CM, Lanigan SW, Hughes J, Anstey AV . Demographic study of port wine stain patients attending a laser clinic: family history, prevalence of naevus anaemicus and results of prior treatment Clin Exp Dermatol 1997 22: 166–168
Troilius A . Characterisation and treatment of patients with port wine stains with special reference to the emotional impact PhD thesis, Lund University, Lund, Sweden 1999
Aelvoet GE, Jorens PG, Roelen LM . Genetic aspects of the Klippel-Trenaunay syndrome Br J Dermatol 1992 126: 603–607
Enjolras O, Riché MC, Merland JJ . Facial port-wine stains and Sturge-Weber syndrome Pediatrics 1985 76: 48–51
Barsky SH, Rosen S, Geer DE, Noe JM . The nature and evolution of port wine stains: A computer-assisted study J Invest Dermatol 1980 74: 154–157
Finley JL, Clark RA, Colvin RB, Blackman R, Noe J, Rosen S . Immunofluorescent staining with antibodies to factor VIII, fibronectin, and collagenous basement membrane protein in normal human skin and port wine stains Arch Dermatol 1982 118: 971–975
Neumann R, Leonhartsberger H, Knobler R, Honigsmann H . Immunohistochemistry of port-wine stains and normal skin with endothelium-specific antibodies PAL-E, anti-ICAM-1, anti-ELAM-1, and anti-factor VIIIrAg Arch Dermatol 1994 130: 879–883
Smoller BR, Rosen S . Port-wine stains. A disease of altered neural modulation of blood vessels? Arch Dermatol 1986 122: 177–179
Rydh M, Malm M, Jernbeck J, Dalsgaard CJ . Ectatic blood vessels in port-wine stains lack innervation: Possible role in pathogenesis Plast Reconstr Surg 1991 87: 419–422
Oosthuyse B, Moons L, Storkebaum E et al. Deletion of the hypoxia-response element in the vascular endothelial growth factor promoter causes motor neuron degeneration Nat Genet 2001 28: 131–138
Richards B, Skoletsky J, Shuber AP et al. Multiplex PCR amplification from the CFTR gene using DNA prepared from buccal brushes/swabs Hum Mol Genet 1993 2: 159–163
Kruglyak L, Daly MJ, Reeve-Daly MP, Lander ES . Parametric and nonparametric linkage analysis: a unified multipoint approach Am J Hum Genet 1996 58: 1347–1363
Vikkula M, Boon LM, Mulliken JB, Olsen BR . Molecular basis of vascular anomalies Trends Cardiovasc Med 1998 8: 281–292
Vikkula M, Boon LM, Mulliken JB . Molecular genetics of vascular malformation Matrix Biol 2001 20: 327–335
Vikkula M, Boon LM, Carraway KL et al. Vascular dysmorphogenesis caused by an activating mutation in the receptor tyrosine kinase TIE2 Cell 1996 87: 1181–1190
Kärkkäinen MJ, Ferrell RE, Lawrence EC et al. Missense mutations interfere with VEGFR-3 signalling in primary lymphoedema Nat Genet 2000 25: 153–159
Irrthum A, Karkkainen MJ, Devriendt K, Alitalo K, Vikkula M . Congenital hereditary lymphedema caused by a mutation that inactivates VEGFR3 tyrosine kinase Am J Hum Genet 2000 67: 295–301
Johnson DW, Berg JN, Baldwin MA et al. Mutations in the activin receptor-like kinase 1 gene in hereditary haemorrhagic telangiectasia type 2 Nat Genet 1996 13: 189–195
McAllister KA, Grogg KM, Johnson DW et al. Endoglin, a TGF-beta binding protein of endothelial cells, is the gene for hereditary haemorrhagic telangiectasia type 1 Nat Genet 1994 8: 345–351
Finegold DN, Kimak MA, Lawrence EC et al. Truncating mutations in FOXC2 cause multiple lymphedema syndromes Hum Mol Genet 2001 10: 1185–1189
Laberge-le Couteulx S, Jung HH, Labauge P et al. Truncating mutations in CCM1, encoding KRIT1, cause hereditary cavernous angiomas Nat Genet 1999 23: 189–193
Sahoo T, Johnson EW, Thomas JW et al. Mutations in the gene encoding KRIT1, a Krev-1/rap1a binding protein, cause cerebral cavernous malformations (CCM1) Hum Mol Genet 1999 8: 2325–2333
Eerola I, Plate KH, Spiegel R, Boon L, Mulliken JB, Vikkula M . KRIT1 is mutated in hyperkeratotic cutaneous capillary-venous malformation associated with cerebral capillary malformation Hum Mol Genet 2000 9: 1351–1355
Brouillard P, Boon LM, Mulliken JB et al. Mutations in a novel factor glomulin are responsible for glomuvenous malformations (‘glomangiomas’) Am J Hum Genet 2002 70: 866–874
Bi W, Drake CJ, Schwarz JJ . The transcription factor MEF2C-null mouse exhibits complex vascular malformations and reduced cardiac expression of angiopoietin 1 and VEGF Dev Biol 1999 15: 255–267
Henkemeyer M, Rossi DJ, Holmyard DP et al. Vascular system defects and neuronal apoptosis in mice lacking ras GTPase-activating protein Nature 1995 377: 695–701
Greer P, Haigh J, Mbamalu G, Khoo W, Bernstein A, Pawson T . The Fps/Fes protein-tyrosine kinase promotes angiogenesis in transgenic mice Mol Cell Biol 1994 14: 6755–6763
Arber S, Caroni P . Thrombospondin-4, an extracellular matrix protein expressed in the developing and adult nervous system promotes neurite outgrowth J Cell Biol 1995 131: 1083–1094
Nakayama J, Fukuda MN, Fredette B, Ranscht B, Fukuda M . Expression cloning of a human polysialyltransferase that forms the polysialylated neural cell adhesion molecule present in embryonic brain Proc Natl Acad Sci USA 1995 92: 7031–7035
Drescher U, Kremoser C, Handwerker C, Loschinger J, Noda M, Bonhoeffer F . In vitro guidance of retinal ganglion cell axons by RAGS, a 25 kDa tectal protein related to ligands for Eph receptor tyrosine kinases Cell 1995 82: 359–370
Boye E, Yu Y, Paranya G, Mulliken JB, Olsen BR, Bischoff J . Clonality and altered behavior of endothelial cells from hemangiomas J Clin Invest 2001 107: 745–752
Berg JN, Walter JW, Thisanagayam U et al. Evidence for loss of heterozygosity of 5q in sporadic haemangiomas: Are somatic mutations involved in haemangioma formation? J Clin Pathol 2001 54: 249–252
Walter JW, Blei F, Anderson JL, Orlow SJ, Speer MC, Marchuk DA . Genetic mapping of a novel familial form of infantile hemangioma Am J Med Genet 1999 82: 77–83
Whelan AJ, Watson MS, Porter FD, Steiner RD . Klippel-Trenaunay-Weber syndrome associated with a 5:11 balanced translocation Am J Med Genet 1995 59: 492–494
Acknowledgements
The authors are grateful to all the families involved in this study. The work was supported by the Fonds Speciaux de Recherche – Université catholique de Louvain, the Belgian Federal Service for Scientific, Technical and Cultural Affairs, and the F.N.R.S (Fonds national de la recherche scientifique) (all to M Vikkula, a ‘chercheur qualifié du F.N.R.S’). I Eerola was supported by a Marie Curie Fellowship of the European Community Program Quality of Life, under contract number QLRI-CT-1999-51491. The authors thank Ms Ana Gutierrez for technical assistance and Mrs Liliana Niculescu for secretarial help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eerola, I., Boon, L., Watanabe, S. et al. Locus for susceptibility for familial capillary malformation (‘port-wine stain’) maps to 5q. Eur J Hum Genet 10, 375–380 (2002). https://doi.org/10.1038/sj.ejhg.5200817
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.ejhg.5200817
Keywords
This article is cited by
-
Theoretical and in vivo experimental investigation of laser hyperthermia for vascular dermatology mediated by liposome@Au core–shell nanoparticles
Lasers in Medical Science (2022)
-
A novel RASA1 mutation causing capillary malformation-arteriovenous malformation (CM-AVM): the first genetic clinical report in East Asia
Hereditas (2018)
-
Genome-wide analyses identify common variants associated with macular telangiectasia type 2
Nature Genetics (2017)
-
A spectrum of intracranial vascular high-flow arteriovenous shunts in RASA1 mutations
Child's Nervous System (2016)
-
Fluch des Diplomaten
Rechtsmedizin (2014)