Abstract
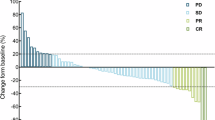
Our purpose was to determine, in patients with metastatic colorectal carcinoma treated with irinotecan single-agent after 5-FU failure, the most significant predictive parameters for tumour response, progression-free survival and toxicity. Between October 1992 and April 1995, 455 patients with 5-FU resistant metastatic colorectal carcinoma entered four consecutive phase II trials. The first two studies assessed tumour response, the other two were randomized studies which assessed the efficacy of racecadotril to prevent irinotecan-induced diarrhoea. Due to homogeneous main eligibility criterias, data from those studies could be pooled for statistical analysis. Potential clinical and biological predictive factors (PF) for toxicity, tumour growth control, e.g. response or stabilization and progression-free survival (PFS), were studied in multivariate analysis. 363 patients were evaluable for response, 432 were evaluable for PFS, 368 for neutropenia and 416 for delayed diarrhoea, respectively. Normal baseline haemoglobin level (Hb), time since diagnosis of colorectal carcinoma, grade 3 or 4 neutropenia or diarrhoea at first cycle and a low number of organs involved were the most PF for tumour growth control (P< 0.05). Significant prognostic variables for PFS were WHO Performance Status, liver and lymph-node involvement, time since diagnosis, age and CEA value (P≤ 0.02). Six groups of patients based on the number of unfavourable prognostic factors are presented. Baseline bilirubin, haemoglobin level, number of organs involved and time from diagnosis were PF for neutropenia; PS, serum creatinine, leukocyte count, time from 5-FU progression and prior abdominopelvic irradiation were PF for delayed diarrhoea (P≤ 0.05). These PF should help clinicians to anticipate for a given patient the probability to observe a response/stabilization or a toxicity. These results should also be prospectively confirmed in ongoing or future trials using irinotecan, both as a single agent and in combination with other drugs. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Advanced Colorectal Cancer Meta-analysis Project (1992) 2. Modulation of fluorouracil by leucovorin in patients with advanced colorectal cancer: evidence in terms of response rates. J Clin Oncol 10: 896–903
Advanced Colorectal Cancer Meta-analysis Project (1994) Meta-analysis of randomized trials testing the biochemical modulation of fluorouracil by methotrexate in metastatic colorectal cancer. J Clin Oncol 5: 960–969
Ahlgren JD, Trocki O and Gullo JJ (1991) Protracted infusion of 5FU with weekly low dose cisplatin as a second line therapy in patients with metastatic colorectal cancer who have failed 5-FU monotherapy. Cancer Invest 9: 27–33
Allen M, Cunningham D and Schmitt C (1998) The importance of stabilization as an endpoint in the treatment of metastatic colorectal carcinoma: recent quality of life studies. Anticancer Drugs 9: 783–790
Bertrand M, Doroshow JH, Multhauf P, Blayney DW, Carr BI and Cecchi G (1992) High dose continuous infusion folinic acid and bolus 5-fluorouracil in patients with advanced colorectal cancer: a randomized trial. J Clin Oncol 10: 904–911
Brienza S, Levi F and Valori VM (1993) Intensified (every 2 weeks) chronotherapy with 5-fluorouracil folinic acid and oxaliplatin in previously treated patients with metastatic colorectal cancer. Proc Am Soc Clin Oncol 12: 125, 620
Canal P, Gay C, Dezeuze A, Douillard JY, Bugat R, Brunet R, Adenis A, Herait P, Lokiec F and Mathieu-Boue A (1996) Pharmacokinetics and pharmacodynamics of Irinotecan during a phase II clinical trial in colorectal cancer. J Clin Oncol 14: 2688–2695
Chabot GG, Abigerges A, Catimel G, Culine S, de Forni M, Extra JM, Majoubi M, Herait P, Armand JP and Bugat R (1995) Population pharmacokinetics and pharmacodynamics of irinotecan (CPT-11) and active metabolite SN-38 during phase I trials. Ann Oncol 6: 141–151
Cunningham D, Pyrrhönen S, James RD, Punt CJ, Hickish TF, Heikkila R, Yohannesen TD, Starkkhammar H, Topham CA, Awwad L, Jacques C and Herait P (1998) Randomised trial of irinotecan plus supportive care versus supportive care alone after fluorouracil failure for patients with metastatic colorectal cancer. The Lancet 352: 1401–1418
de Gramont A, Bosset JF, Milan C, Rougier P, Bouché O, Etienne PL, Morvan F, Louvet C, Guillot T, François E and Bedenne L (1997) Randomized trial comparing monthly low-dose leucovorin and fluorouracil bolus with bimonthly high-dose leucovorin and fluorouracil bolus plus continuous infusion for advanced colorectal cancer: a french intergroup study. J Clin Oncol 15: 808–815
de Gramont A, Vignoud J, Tournigand C, Louvet C, Varette C, Raymond E, Moreau S, Le Bail N and Krulik M (1997) Oxaliplatin with high dose folinic acid and 5 fluorouracil 48 hours infusion in pretreated metastatic colorectal cancer. Eur J Cancer 33: 214–219
Glimelius B, Hoffman K, Graf W, Páhlman L and Sjödén PO (1994) Quality of life during chemotherapy in patient with symptomatic advanced colorectal cancer. Cancer 73: 556–562
Izzo J, Cvitkovic E, Zarba J, Chadjaa M, May-Levin F and Riggi M (1992) Low dose 5FU continuous infusion in advanced colorectal cancer: Clinical evidence for reversal of acquired/intrinsic resistance to 5-FU or 5-FU folinic Acid. Ann Oncol 5: 38–43
Nordic Gastrointestinal Tumor Adjuvant Project (1992) Expectancy of primary chemotherapy in patients with advanced asymptomatic colorectal cancer: a randomized trial. J Clin Oncol 10: 904–911
Pitot HC, Wender DB, O'Connell MJ, Schroeder G, Goldberg RM, Rubin J, Mailliard JA, Knost JA, Ghosh C, Kirschling PJ, Levitt R and Windschitl HE (1997) Phase II trial of irinotecan in patients with metastatic colorectal carcinoma. J Clin Oncol 15: 2910–2919
Rougier P, Bugat R, Douillard JY, Culine S, Suc E, Brunet P, Becouarn Y, Ychou M, Marty M, Extra JM, Bonneterre J, Adenis A, Seitz JF, Ganem G, Namer M, Conroy T, Negrier S, Merrouche Y, Burki F, Mousseau M, Herait P and Mahjoubi M (1997) Phase II study of irinotecan in the treatment of advanced colorectal cancer in chemotherapy-naive patients and patients pretreated with fluorouracil-based chemotherapy. J Clin Oncol 15: 251–260
Rougier P, Milan C, Lazorthes F, Fourtanier G, Partensky C, Baumel H and Faivre J (1995) Prospective study of prognostic factors in patients with unresected hepatic metastases from colorectal cancer. Br J Surg 82: 1397–1400
Rougier Ph, Van Cutsem E, Bajetta E, Niederle N, Possinger C, Labianca,, Navarro M, Moraut R, Bleigerg H, Nils J, Awad L, Herait P and Jacques C (1998) Randomized trial of irinotecan versus fluorouracil by continuous infusion after fluorouracil failure in patients with metastatic colorectal cancer. The Lancet 352: 1407–1412
Saltz L, Danenberg K and Paty P (1998) High thymidylate synthase expression does not preclude activity of CPT-11 in colorectal cancer. Proc Am Soc Clin Oncol 281a: 1210
Scheithauer W, Rosen H, Kornek GV, Sebesta C and Depisch D (1993) Randomized comparison of combination chemotherapy plus supportive care or supportive care alone in patients with metastatic colorectal cancer. BMJ 306: 752–755
Author information
Authors and Affiliations
Consortia
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Freyer, G., Rougier, P., Bugat, R. et al. Prognostic factors for tumour response, progression-free survival and toxicity in metastatic colorectal cancer patients given irinotecan (CPT-11) as second-line chemotherapy after 5FU failure. Br J Cancer 83, 431–437 (2000). https://doi.org/10.1054/bjoc.2000.1303
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1054/bjoc.2000.1303
Keywords
This article is cited by
-
Population pharmacokinetic analysis of AR-67, a lactone stable camptothecin analogue, in cancer patients with solid tumors
Investigational New Drugs (2019)
-
Acceleration of carboxylesterase-mediated activation of irinotecan to SN-38 by serum from patients with end-stage kidney disease
Cancer Chemotherapy and Pharmacology (2018)
-
Clarithromycin co-administration does not increase irinotecan (CPT-11) toxicity in colorectal cancer patients
Cancer Chemotherapy and Pharmacology (2017)
-
Cooperative inhibitory effects of uremic toxins and other serum components on OATP1B1-mediated transport of SN-38
Cancer Chemotherapy and Pharmacology (2017)
-
NKTR-102 Efficacy versus irinotecan in a mouse model of brain metastases of breast cancer
BMC Cancer (2015)