Abstract
Aim:
To search for novel inhibitors of human polo-like kinase 1 (Plk1), which plays important roles in various aspects of mitotic progression and is believed as a promising anti-cancer drug target, and further investigate the potential inhibition mechanism of active compounds against Plk1, thus developing potent anti-tumor lead compounds.
Methods:
Surface plasmon resonance (SPR) technology-based assay and enzymatic inhibition assay were used to screen Plk1 inhibitors. Sulphorhodamine B (SRB)-based assay, flow cytometry, confocal microscopy and Western blotting were used to further identify the potent Plk1 inhibitor. To investigate the inhibitory mechanism of the active compound against Plk1, enzymatic inhibition assay, SPR and yeast two-hybrid technology-based assays were used.
Results:
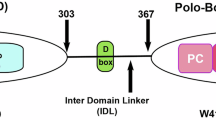
Aristolactam AIIIa was identified as a new type of Plk1 inhibitors, targeting the Polo Box domain (PBD) which is another efficient tactic for exploring Plk1 inhibitors. Further studies indicated that it could block the proliferations of HeLa, A549, HGC and the HCT-8/V cells (clinical Navelbine-resistant cancer cell), induce mitotic arrest of HeLa cells at G2/M phase with spindle abnormalities and promote apoptosis in HeLa cells. The results from SPR and yeast two-hybrid technology-based assays suggested that it could target both the catalytic domain of Plk1 (CD) and PBD and enhance the CD/PBD interaction.
Conclusion:
Our current work is expected to shed light on the potential anti-tumor mechanism of Aristolactam AIIIa, and this natural product might be possibly used as a lead compound for further developing anti-tumor drugs.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Glover DM, Hagan IM, Tavares AA . Polo-like kinases: a team that plays throughout mitosis. Genes Dev 1998; 12: 3777–87.
Hamanaka R, Maloid S, Smith MR, O'Connell CD, Longo DL, Ferris DK . Cloning and characterization of human and murine homologues of the Drosophila polo serine-threonine kinase. Cell Growth Differ 1994; 5: 249–57.
Simmons DL, Neel BG, Stevens R, Evett G, Erikson RL . Identification of an early-growth-response gene encoding a novel putative protein kinase. Mol Cell Biol 1992; 12: 4164–9.
Donohue PJ, Alberts GF, Guo Y, Winkles JA . Identification by targeted differential display of an immediate early gene encoding a putative serine/threonine kinase. J Biol Chem 1995; 270: 10351–7.
Ouyang B, Pan H, Lu L, Li J, Stambrook P, Li B, et al. Human Prk is a conserved protein serine/threonine kinase involved in regulating M phase functions. J Biol Chem 1997; 272: 28646–51.
Fode C, Motro B, Yousefi S, Heffernan M, Dennis JW . Sak, a murine protein-serine/threonine kinase that is related to the Drosophila polo kinase and involved in cell proliferation. Proc Natl Acad Sci USA 1994; 91: 6388–92.
Roshak AK, Capper EA, Imburgia C, Fornwald J, Scott G, Marshall LA . The human polo-like kinase, PLK, regulates cdc2/cyclin B through phosphorylation and activation of the cdc25C phosphatase. Cell Signal 2000; 12: 405–11.
Lane HA, Nigg EA . Antibody microinjection reveals an essential role for human polo-like kinase 1 (Plk1) in the functional maturation of mitotic centrosomes. J Cell Biol 1996; 135: 1701–13.
Sumara I, Vorlaufer E, Stukenberg PT, Kelm O, Redemann N, Nigg EA, et al. The dissociation of cohesin from chromosomes in prophase is regulated by Polo-like kinase. Mol Cell 2002; 9: 515–25.
Kotani S, Tugendreich S, Fujii M, Jorgensen PM, Watanabe N, Hoog C, et al. PKA and MPF-activated polo-like kinase regulate anaphase-promoting complex activity and mitosis progression. Mol Cell 1998; 1: 371–80.
Zhou T, Aumais JP, Liu X, Yu-Lee LY, Erikson RL . A role for Plk1 phosphorylation of NudC in cytokinesis. Dev Cell 2003; 5: 127–38.
Strebhardt K, Ullrich A . Targeting polo-like kinase 1 for cancer therapy. Nat Rev Cancer 2006; 6: 321–30.
Smith MR, Wilson ML, Hamanaka R, Chase D, Kung H, Longo DL, et al. Malignant transformation of mammalian cells initiated by constitutive expression of the polo-like kinase. Biochem Biophys Res Commun 1997; 234: 397–405.
Spänkuch-Schmitt B, Wolf G, Solbach C, Loibl S, Knecht R, Stegmüller M, et al. Downregulation of human polo-like kinase activity by antisense oligonucleotides induces growth inhibition in cancer cells. Oncogene 2002; 21: 3162–71.
Elez R, Piiper A, Kronenberger B, Kock M, Brendel M, Hermann E, et al. Tumor regression by combination antisense therapy against Plk1 and Bcl-2. Oncogene 2003; 22: 69–80.
Jang YJ, Lin CY, Ma S, Erikson RL . Functional studies on the role of the C-terminal domain of mammalian polo-like kinase. Proc Natl Acad Sci USA 2002; 99: 1984–9.
Elia AE, Rellos P, Haire LF, Chao JW, Ivins FJ, Hoepker K, et al. The molecular basis for phosphodependent substrate targeting and regulation of Plks by the Polo-box domain. Cell 2003; 115: 83–95.
Stevenson CS, Capper EA, Roshak AK, Marquez B, Eichman C, Jackson JR, et al. The identification and characterization of the marine natural product scytonemin as a novel antiproliferative pharmacophore. J Pharmacol Exp Ther 2002; 303: 858–66.
Stevenson CS, Capper EA, Roshak AK, Marquez B, Grace K, Gerwick WH, et al. Scytonemin — a marine natural product inhibitor of kinases key in hyperproliferative inflammatory diseases. Inflamm Res 2002; 51: 112–4.
Gumireddy K, Reddy MV, Cosenza SC, Boominathan R, Baker SJ, Papathi N, et al. ON01910, a non-ATP-competitive small molecule inhibitor of Plk1, is a potent anticancer agent. Cancer Cell 2005; 7: 275–86.
Reindl W, Yuan J, Krämer A, Strebhardt K, Berg T . Inhibition of polo-like kinase 1 by blocking polo-box domain-dependent protein-protein interactions. Chem Biol 2008; 15: 459–66.
Priestap HA . Seven aristololactams from Aristolochia Argentina. Phytochemistry 1985; 24: 849–52.
Couture A, Deniau E, Grandclaudon P, Rybalko-Rosen H, Léonce S, Pfeiffer B, et al. Synthesis and biological evaluation of Aristolactams. Bioorg Med Chem Lett 2002; 12: 3557–9.
Kumar V, Poonam, Prasad AK, Parmar VS . Naturally occurring Aristolactams, aristolochic acids and dioxoaporphines and their biological activities. Nat Prod Rep 2003; 20: 565–83.
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, et al. New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 1990; 82: 1107–12.
Diaz-Camino C, Risseeuw EP, Liu E, Crosby WL . A high-throughput system for two-hybrid screening based on growth curve analysis in microtiter plates. Anal Biochem 2003; 316; 171–4.
Chia YC, Chang FR, Teng CM, Wu YC . Aristolactams and dioxoaporphines from Fissistigma balansae and Fissistigma oldhamii. J Nat Prod 2000; 63: 1160–3.
Cogswell JP, Brown CE, Bisi JE, Neill SD . Dominant-negative polo-like kinase 1 induces mitotic catastrophe independent of cdc25C function. Cell Growth Differ 2000; 11: 615–23.
Sumara I, Giménez-Abián JF, Gerlich D, Hirota T, Kraft C, de la Torre C, et al. Roles of polo-like kinase 1 in the assembly of functional mitotic spindles. Curr Biol 2004; 14: 1712–22.
Soldani C, Scovassi AI . Poly (ADP-ribose) polymerase-1 cleavage during apoptosis: an update. Apoptosis 2002; 7: 321–8.
Liu X, Erikson RL . Polo-like kinase (Plk) 1 depletion induces apoptosis in cancer cells. Proc Natl Acad Sci USA 2003; 100: 5789–94.
Steegmaier M, Hoffmann M, Baum A, Lénárt P, Petronczki M, Krssák M, et al. BI 2536, a potent and selective inhibitor of polo-like kinase 1, inhibits tumor growth in vivo. Curr Biol 2007; 17: 316–22.
Lee KS, Grenfell TZ, Yarm FR, Erikson RL . Mutation of the polo-box disrupts localization and mitotic functions of the mammalian polo kinase Plk. Proc Natl Acad Sci USA 1998; 95: 9301–6.
Acknowledgements
This work was supported by the State Key Program of Basic Research of China (grants 2010CB912501, 2009CB918502), the National Natural Science Foundation of China (grant 30890044), Shanghai Basic Research Project from the Shanghai Science and Technology Commission (grant 0811141013) and the Knowledge Innovation Program of the Chinese Academy of Sciences (grant SIMM0709QN-19).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, L., Wang, X., Chen, J. et al. The natural product Aristolactam AIIIa as a new ligand targeting the polo-box domain of polo-like kinase 1 potently inhibits cancer cell proliferation. Acta Pharmacol Sin 30, 1443–1453 (2009). https://doi.org/10.1038/aps.2009.141
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/aps.2009.141
Keywords
This article is cited by
-
Polo-like kinases and acute leukemia
Oncogene (2019)
-
Enhanced cellular uptake of a TAT-conjugated peptide inhibitor targeting the polo-box domain of polo-like kinase 1
Amino Acids (2014)
-
Serendipitous alkylation of a Plk1 ligand uncovers a new binding channel
Nature Chemical Biology (2011)