Abstract
Aim:
To investigate the effects of plumbagin, a naphthoquinone derived from the medicinal plant Plumbago zeylanica, on human breast cancer cell growth and the cancer cell-induced osteolysis in the bone microenvironment of mice.
Methods:
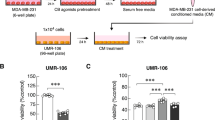
Human breast cancer cell subline MDA-MB-231SA with the ability to spread and grow in the bone was tested. The cell proliferation was determined using the CCK-8 assay. Apoptosis was detected with Annexin V/PI double-labeled flow cytometry. Red fluorescent protein-labeled MDA-MB-231SArfp cells were injected into the right tibia of female BALB/c-nu/nu mice. Three days after the inoculation, the mice were injected with plumbagin (2, 4, or 6 mg/kg, ip) 5 times per week for 7 weeks. The growth of the tumor cells was monitored using an in vivo imaging system. After the mice were sacrificed, the hind limbs were removed for radiographic and histological analyses.
Results:
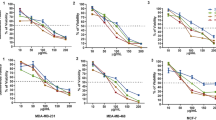
Plumbagin (2.5–20 μmol/L) concentration-dependently inhibited the cell viability and induced apoptosis of MDA-MB-231SA cells in vitro (the IC50 value of inhibition of cell viability was 14.7 μmol/L). Administration of plumbagin to breast cancer bearing mice delayed the tumor growth by 2–3 weeks and reduced the tumor volume by 44%–74%. The in vivo imaging study showed that plumbagin dose-dependently inhibited MDA-MB-231SArfp cell growth in bone microenvironment. Furthermore, X-ray images and micro-CT study demonstrated that plumbagin reduced bone erosion area and prevented a decrease in bone tissue volume. Histological studies showed that plumbagin dose-dependently inhibited the breast cancer cell growth, enhanced the cell apoptosis and reduced the number of TRAcP-positive osteoclasts.
Conclusion:
Plumbagin inhibits the cell growth and induces apoptosis in human breast cancer cells in mice bone microenvironment, leading to significant reduction in osteolytic lesions caused by the tumor cells.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Kamangar F, Dores GM, Anderson WF . Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol 2006; 24: 2137–50.
Mundy GR . Metastasis to bone: causes, consequences and therapeutic opportunities. Nat Rev Cancer 2002; 2: 584–93.
Rose A, Siegel PM . Breast cancer-derived factors facilitate osteolytic bone metastasis. Bull Cancer 2006; 93: 931–43.
Guise TA, Yin JJ, Taylor S, Kumagai Y, Dallas M, Boyce B, et al. Evidence for a causal role of parathyroid hormone-related protein in the pathogenesis of human breast cancer–mediated osteolysis. J Clin Invest 1996; 98: 1544.
Lipton A, Theriault RL, Hortobagyi GN, Simeone J, Knight RD, Mellars K, et al. Pamidronate prevents skeletal complications and is effective palliative treatment in women with breast carcinoma and osteolytic bone metastases. Cancer 2000; 88: 1082–90.
Bamias A, Kastritis E, Bamia C, Moulopoulos LA, Melakopoulos I, Bozas G, et al. Osteonecrosis of the jaw in cancer after treatment with bisphosphonates: incidence and risk factors. J Clin Oncol 2005; 23: 8580–7.
Dimopoulos MA, Kastritis E, Anagnostopoulos A, Melakopoulos I, Gika D, Moulopoulos LA, et al. Osteonecrosis of the jaw in patients with multiple myeloma treated with bisphosphonates: evidence of increased risk after treatment with zoledronic acid. haematologica 2006; 91: 968–71.
Durie BGM, Katz M, Crowley J, Woo SB, Hande K, Richardson PG, et al. Osteonecrosis of the jaw and bisphosphonates. New Engl J Med 2005; 353: 99–102.
Hoff AO, Toth BB, Altundag K, Johnson MM, Warneke CL, Hu M, et al. Frequency and risk factors associated with osteonecrosis of the jaw in cancer patients treated with intravenous bisphosphonates. J Bone Mineral Res 2008; 23: 826–36.
Choi S, Lim MH, Kim KM, Jeon BH, Song WO, Kim TW . Cordycepin-induced apoptosis and autophagy in breast cancer cells are independent of the estrogen receptor. Toxicol appl Pharmacol 2011; 257: 165–73.
Zhang Y, Zhu G, Gu S, Chen X, Hu H, Weng S . Genistein inhibits osteolytic bone metastasis and enhances bone mineral in nude mice. Environ Toxicol Pharmacol 2010; 30: 37–44.
Li Y, Bäckesjö CM, Haldosén LA, Lindgren U . Resveratrol inhibits proliferation and promotes apoptosis of osteosarcoma cells. Eur J Pharmacol 2009; 609: 13–8.
Park H, Bergeron E, Senta H, Guillemette K, Beauvais S, Blouin R, et al. Sanguinarine induces apoptosis of human osteosarcoma cells through the extrinsic and intrinsic pathways. Biochem Biophys Res Commun 2010; 399: 446–51.
Sandur SK, Ichikawa H, Sethi G, Ahn KS, Aggarwal BB . Plumbagin (5-hydroxy-2-methyl-1,4-naphthoquinone) suppresses NF-κB activation and NF-κB-regulated gene products through modulation of p65 and IκBα kinase activation, leading to potentiation of apoptosis induced by cytokine and chemotherapeutic agents. J Biol Chem 2006; 281: 17023–33.
Paiva SR, Figueiredo MR, Aragão TV, Kaplan MAC . Antimicrobial activity in vitro of plumbagin isolated from Plumbago species. Mem Inst Oswaldo Cruz 2003; 98: 959–61.
Sharma I, Gusain D, Dixit VP . Hypolipidaemic and antiatherosclerotic effects of plumbagin in rabbits. Indian J Physiol Pharmacol 1991; 35: 10–4.
Checker R, Sharma D, Sandur SK, Khanam S, Poduval T . Anti-inflammatory effects of plumbagin are mediated by inhibition of NF-κB activation in lymphocytes. Int Immunopharmacol 2009; 9: 949–58.
Manu KA, Shanmugam MK, Rajendran P, Li F, Ramachandran L, Hay HSIN, et al. Plumbagin inhibits invasion and migration of breast and gastric cancer cells by downregulating the expression of chemokine receptor CXCR4. Mol Cancer 2011; 10: 107.
Kuo PL, Hsu YL, Cho CY . Plumbagin induces G2–M arrest and autophagy by inhibiting the AKT/mammalian target of rapamycin pathway in breast cancer cells. Mol Cancer Ther 2006; 5: 3209–21.
Aziz MH, Dreckschmidt NE, Verma AK . Plumbagin, a medicinal plant-derived naphthoquinone, is a novel inhibitor of the growth and invasion of hormone-refractory prostate cancer. Cancer Res 2008; 68: 9024–32.
Sand JM, Hafeez BB, Jamal MS, Witkowsky O, Siebers EM, Fischer J, et al. Plumbagin (5-hydroxy-2-methyl-1,4-naphthoquinone), isolated from Plumbago zeylanica, inhibits ultraviolet radiation-induced development of squamous cell carcinomas. Carcinogenesis 2012; 33: 184–90.
Logothetis CJ, Lin SH . Osteoblasts in prostate cancer metastasis to bone. Nat Rev Cancer 2005; 5: 21–8.
Sung B, Oyajobi B, Aggarwal BB . Plumbagin inhibits osteoclastogenesis and reduces human breast cancer-induced osteolytic bone metastasis in mice through suppression of RANKL signaling. Mol Cancer Ther 2012; 11: 350–9.
Yoneda T, Williams PJ, Hiraga T, Niewolna M, Nishimura R . A bone-seeking clone exhibits different biological properties from the MDA-MB-231 parental human breast cancer cells and a brain-seeking clone in vivo and in vitro. J Bone Miner Res 2001; 16: 1486–95.
Price JT, Quinn JMW, Sims NA, Vieusseux J, Waldeck K, Docherty SE, et al. The heat shock protein 90 inhibitor, 17-allylamino-17-demethoxygeldanamycin, enhances osteoclast formation and potentiates bone metastasis of a human breast cancer cell line. Cancer Res 2005; 65: 4929–38.
Park HH, Lee KY, Kim SH, Lee YJ, Koh SH . L-DOPA-induced neurotoxicity is reduced by the activation of the PI3K signaling pathway. Toxicology 2009; 265: 80–6.
Zheng Y, Zhou H, Modzelewski JRK, Kalak R, Blair JM, Seibel MJ, et al. Accelerated bone resorption, due to dietary calcium deficiency, promotes breast cancer tumor growth in bone. Cancer Res 2007; 67: 9542–48.
Zheng Y, Zhou H, Brennan K, Blair JM, Modzelewski J, Seibel MJ, et al. Inhibition of bone resorption, rather than direct cytotoxicity, mediates the anti-tumour actions of ibandronate and osteoprotegerin in a murine model of breast cancer bone metastasis. Bone 2007; 40: 471.
Jiang Y, Huang Y, Cheng C, Lu W, Zhang Y, Liu X, et al. Combination of thiazolidinedione and hydralazine suppresses proliferation and induces apoptosis by PPARγ up-expression in MDA-MB-231 cells. Exp Mol Pathol 2011; 91: 768–74.
Ahmad A, Banerjee S, Wang Z, Kong D, Sarkar FH . Plumbagin-induced apoptosis of human breast cancer cells is mediated by inactivation of NF-κB and Bcl-2. J Cell Biochem 2008; 105: 1461–71.
Kawiak A, Zawacka-Pankau J, Lojkowska E . Plumbagin induces apoptosis in Her2-overexpressing breast cancer cells through the mitochondrial-mediated pathway. J Nat Prod 2012; 75: 747–51.
Aziz MH, Dreckschmidt NE, Verma AK . Plumbagin, a medicinal plant-derived naphthoquinone, is a novel inhibitor of the growth and invasion of hormone-refractory prostate cancer. Cancer Res 2008; 68: 9024–32.
Hafeez BB, Zhong W, Mustafa A, Fischer JW, Witkowsky O, Verma AK . Plumbagin inhibits prostate cancer development in TRAMP mice via targeting PKC{varepsilon}, Stat3 and neuroendocrine markers. Carcinogenesis 2012; 33: 2586–92.
Devi PU, Solomon F, Sharada A . Plumbagin, a plant naphthoquinone with antitumor and radiomodifying properties. Pharm Biol 1999; 37: 231–6.
Li Z, Xiao J, Wu X, Li W, Yang Z, Xie J, et al. Plumbagin inhibits breast tumor bone metastasis and osteolysis by modulating the tumor-bone microenvironment. Curr Mol Med 2012; 12: 967–81.
Acknowledgements
This work was supported by the grants from the National Natural Science Foundation of China (No 81172549, 81228013 and 81301531), the Shanghai Science and Technology Development Fund (No 11XD1403300 and 12140901300), the Program of Key Disciplines of Shanghai Municipal Education Commission (No J50206), and the Opening Project of Shanghai Key Laboratory of Orthopaedic Implant (KFKT2011003). The authors would like to thank Dr John PRICE for providing the MDA-MB-231SArfp cell line.
Author information
Authors and Affiliations
Corresponding author
PowerPoint slides
Rights and permissions
About this article
Cite this article
Yan, W., Wang, Ty., Fan, Qm. et al. Plumbagin attenuates cancer cell growth and osteoclast formation in the bone microenvironment of mice. Acta Pharmacol Sin 35, 124–134 (2014). https://doi.org/10.1038/aps.2013.152
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/aps.2013.152
Keywords
This article is cited by
-
Plumbagin attenuates Bleomycin-induced lung fibrosis in mice
Allergy, Asthma & Clinical Immunology (2022)
-
Phytochemicals in Breast Cancer-Induced Osteoclastogenesis and Bone Resorption: Mechanism and Future Perspective
Current Pharmacology Reports (2022)
-
Plumbagin suppresses chronic periodontitis in rats via down-regulation of TNF-α, IL-1β and IL-6 expression
Acta Pharmacologica Sinica (2017)
-
Structural simulation of adenosine phosphate via plumbagin and zoledronic acid competitively targets JNK/Erk to synergistically attenuate osteoclastogenesis in a breast cancer model
Cell Death & Disease (2016)
-
Kinsenoside screening with a microfluidic chip attenuates gouty arthritis through inactivating NF-κB signaling in macrophages and protecting endothelial cells
Cell Death & Disease (2016)