Abstract
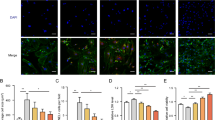
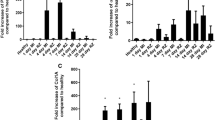
Aspirin (ASA) is a cardioprotective drug with anti-cardiac fibrosis action in vivo. This study was aimed to clarify the anti-cardiac fibrosis action of ASA and the underlying mechanisms. Two heart injury models (injection of isoproterenol and ligation of the left anterior descending branch) were used in mice to induce cardiac fibrosis. The animals were treated with ASA (10 mg·kg−1·d−1, ig) for 21 and 14 d, respectively. ASA administration significantly improved cardiac function, and ameliorated heart damage and fibrosis in the mice. The mechanisms underlying ASA's anti-fibrotic effect were further analyzed in neonatal cardiac fibroblasts (CFs) exposed to hypoxia in vitro. ASA (0.5–5 mmol/L) dose-dependently inhibited the proliferation and Akt phosphorylation in the CFs. In addition, ASA significantly inhibited CF apoptosis, and decreased the levels of apoptosis markers (cleaved caspase 3 and Parp1), which might serve as a side effect of anti-fibrotic effect of ASA. Furthermore, ASA dose-dependently inhibited the autophagy in the CFs, as evidenced by the reduced levels of autophagy marker LC3-II. The autophagy inhibitor Pepstatin A (PepA) promoted the inhibitory effect of ASA on CF proliferation, whereas the autophagy inducer rapamycin rescued ASA-caused inhibition of CF proliferation, suggesting an autophagy-dependent anti-proliferative effect of ASA. Both p38 inhibitor SB203580 and ROS scavenger N-acetyl-cysteine (NAC) significantly decreased Akt phosphorylation in CFs in the basal and hypoxic situations, but they both significantly increased LC3-II levels in the CFs. Our results suggest that an autophagy- and p38/ROS-dependent pathway mediates the anti-cardiac fibrosis effect of ASA in CFs. As PepA and SB203580 did not affect ASA-caused inhibition of CF apoptosis, the drug combination will enhance ASA's therapeutic effects.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Change history
04 March 2026
A Correction to this paper has been published: https://doi.org/10.1038/s41401-026-01780-2
References
Deb A, Ubil E . Cardiac fibroblast in development and wound healing. J Mol Cell Cardiol 2014; 70: 47–55.
Kong PP, Christia NG . Frangogiannis, The pathogenesis of cardiac fibrosis. Cell Mol Life Sci 2014; 71: 549–74.
Hankey GJ . The benefits of aspirin in early secondary stroke prevention. Lancet 2016; 388: 312–4.
Rothwell PM, Algra A, Chen Z, Diener HC, Norrving B, Mehta Z, et al. Effects of aspirin on risk and severity of early recurrent stroke after transient ischaemic attack and ischaemic stroke: time-course analysis of randomised trials. Lancet 2016; 388: 365–75.
Wang X, Lu J, Khaidakov M, Mitra S, Ding Z, Raina S, et al. Aspirin suppresses cardiac fibroblast proliferation and collagen formation through downregulation of angiotensin type 1 receptor transcription. Toxicol Appl Pharmacol 2012; 259: 346–54.
Siddesha JM, Valente AJ, Sakamuri SS, Gardner JD, Delafontaine P, Noda M, et al. Acetylsalicylic acid inhibits IL-18-induced cardiac fibroblast migration through the induction of RECK. J Cell Physiol 2014; 229: 845–55.
Nishida K, Otsu K . Autophagy during cardiac remodeling. J Mol Cell Cardiol 2016. 95: 11–8.
Yasuda O, Takemura Y, Kawamoto H, Rakugi H . Aspirin: recent developments. Cell Mol Life Sci 2008; 65: 354–8.
Hernández C, Barrachina MD, Vallecillo-Hernández J, Álvarez Á, Ortiz-Masiá D, Cosín-Roger J, et al. Aspirin-induced gastrointestinal damage is associated with an inhibition of epithelial cell autophagy. J Gastroenterol 2016; 51: 691–701.
Zhang C, Shi J, Mao SY, Xu YS, Zhang D, Feng LY, et al. Role of p38 MAPK in enhanced human cancer cells killing by the combination of aspirin and ABT-737. J Cell Mol Med 2015; 19: 408–17.
Yang JM, Rui BB, Chen C, Chen H, Xu TJ, Xu WP, et al. Acetylsalicylic acid enhances the anti-inflammatory effect of fluoxetine through inhibition of NF-kappaB, p38-MAPK and ERK1/2 activation in lipopolysaccharide-induced BV-2 microglia cells. Neuroscience 2014; 275: 296–304.
Mao ZM, Shen SM, Wan YG, Sun W, Chen HL, Huang MM, et al. Huangkui capsule attenuates renal fibrosis in diabetic nephropathy rats through regulating oxidative stress and p38MAPK/Akt pathways, compared to alpha-lipoic acid. J Ethnopharmacol 2015; 173: 256–65.
Li ZL, Shi Y, Le G, Ding Y, Zhao Q . 24-Week exposure to oxidized tyrosine induces hepatic fibrosis involving activation of the MAPK/TGF-beta1 signaling pathway in sprague-dawley rats model. Oxid Med Cell Longev 2016; 2016: 3123294.
Ou HC, Lee WJ, Wu CM, Chen JF, Sheu WH . Aspirin prevents resistin-induced endothelial dysfunction by modulating AMPK, ROS, and Akt/eNOS signaling. J Vasc Surg 2012; 55: 1104–15.
Wu Y, Zhai H, Wang Y, Li L, Wu J, Wang F, et al. Aspirin-triggered lipoxin A(4) attenuates lipopolysaccharide-induced intracellular ROS in BV2 microglia cells by inhibiting the function of NADPH oxidase. Neurochem Res 2012; 37: 1690–6.
Zhu X, Li Z, Li C, Zhang J, Zou Z, Wang J . Ginkgo biloba extract and aspirin synergistically attenuate activated platelet-induced ROS production and LOX-1 expression in human coronary artery endothelial cells. Phytomedicine 2013; 20: 114–9.
Essick EE, Ouchi N, Wilson RM, Ohashi K, Ghobrial J, Shibata R, et al. Adiponectin mediates cardioprotection in oxidative stress-induced cardiac myocyte remodeling. Am J Physiol Heart Circ Physiol 2011; 301: H984–93.
Lu H, Tian A, Wu J, Yang C, Xing R, Jia P, et al. Danshensu inhibits beta-adrenergic receptors-mediated cardiac fibrosis by ROS/p38 MAPK axis. Biol Pharm Bull 2014; 37: 961–7.
Wang YS, Li SH, Guo J, Mihic A, Wu J, Sun L, et al. Role of miR-145 in cardiac myofibroblast differentiation. J Mol Cell Cardiol 2014; 66: 94–105.
Carlson LM, Rasmuson A, Idborg H, Segerström L, Jakobsson PJ, Sveinbjörnsson B, et al. Low-dose aspirin delays an inflammatory tumor progression in vivo in a transgenic mouse model of neuroblastoma. Carcinogenesis 2013; 34: 1081–8.
Xiao J, Sheng X, Zhang X, Guo M, Ji X . Curcumin protects against myocardial infarction-induced cardiac fibrosis via SIRT1 activation in vivo and in vitro. Drug Des Devel Ther 2016; 10: 1267–77.
Chen K, Li D, Zhang X, Hermonat PL, Mehta JL . Anoxia-reoxygenation stimulates collagen type-I and MMP-1 expression in cardiac fibroblasts: modulation by the PPAR-gamma ligand pioglitazone. J Cardiovasc Pharmacol 2004; 44: 682–7.
Zhao J, He Q, Cheng Y, Zhao B, Zhang Y, Zhang S, et al. A benzoxazine derivative induces vascular endothelial cell apoptosis in the presence of fibroblast growth factor-2 by elevating NADPH oxidase activity and reactive oxygen species levels. Toxicol In Vitro 2009; 23: 1039–46.
Zhao J, Feng Y, Yan H, Chen Y, Wang J, Chua B, et al. beta-arrestin2/miR-155/GSK3beta regulates transition of 5'-azacytizine-induced Sca-1-positive cells to cardiomyocytes. J Cell Mol Med 2014; 18: 1562–70.
Zou M, Wang F, Gao R, Wu J, Ou Y, Chen X, et al. Autophagy inhibition of hsa-miR-19a-3p/19b-3p by targeting TGF-beta R II during TGF-beta1-induced fibrogenesis in human cardiac fibroblasts. Sci Rep 2016; 6: 24747.
Klionsky DJ, Abeliovich H, Agostinis P, Agrawal DK, Aliev G, Askew DS, et al. Guidelines for the use and interpretation of assays for monitoring autophagy in higher eukaryotes. Autophagy 2008; 4: 151–75.
Yue H, Li W, Liu P, Gao J, Miao J, Zhao J . Inhibition of autophagy promoted sphingosylphosphorylcholine induced cell death in non-small cell lung cancer cells. Biochem Biophys Res Commun 2014; 453: 502–7.
Shirakabe A, Ikeda Y, Sciarretta S, Zablocki DK, Sadoshima J . Aging and autophagy in the heart. Circ Res 2016; 118: 1563–76.
Ghavami S, Cunnington RH, Gupta S, Yeganeh B, Filomeno KL, Freed DH, et al. Autophagy is a regulator of TGF-beta1-induced fibrogenesis in primary human atrial myofibroblasts. Cell Death Dis 2015; 6: e1696.
Di Lisa F, Kaludercic N, Paolocci N . beta(2)-Adrenoceptors, NADPH oxidase, ROS and p38 MAPK: another 'radical' road to heart failure? Br J Pharmacol 2011; 162: 1009–11.
Sun M, Yu H, Zhang Y, Li Z, Gao W . MicroRNA-214 mediates isoproterenol-induced proliferation and collagen synthesis in cardiac fibroblasts. Sci Rep 2015; 5: 18351.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No 31671180; 31070999; 31371158).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Pp., Liu, Hh., Sun, Sh. et al. Aspirin alleviates cardiac fibrosis in mice by inhibiting autophagy. Acta Pharmacol Sin 38, 488–497 (2017). https://doi.org/10.1038/aps.2016.143
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/aps.2016.143
Keywords
This article is cited by
-
Protective effect of aspirin and gentisic acid, a plant-derived phenolic acid, on acrylamide-induced neurotoxicity by inhibiting apoptosis and autophagy
Naunyn-Schmiedeberg's Archives of Pharmacology (2025)
-
Repurposing drugs to treat cardiovascular disease in the era of precision medicine
Nature Reviews Cardiology (2022)
-
Repurposing mesalazine against cardiac fibrosis in vitro
Naunyn-Schmiedeberg's Archives of Pharmacology (2021)
-
Systemic mesalazine treatment prevents spontaneous skin fibrosis in PLK2-deficient mice
Naunyn-Schmiedeberg's Archives of Pharmacology (2021)
-
Sphingosylphosphorylcholine alleviates hypoxia-caused apoptosis in cardiac myofibroblasts via CaM/p38/STAT3 pathway
Apoptosis (2020)