Abstract
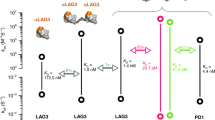
JS-001 is the first monoclonal antibody (mAb) against programmed cell death protein-1 (PD-1) approved by the China Food and Drug Administration (CFDA) into the clinical trails. To date, however, no pre-clinical pharmacological and pharmacokinetic (PK) data are available. In this study, we investigated the efficacy of JS-001 and conducted a preclinical PK study, including the monitoring of anti-drug antibodies (ADAs). We found that JS-001 specifically bound to PD-1 antigen with an EC50 of 21 nmol/L, and competently blocked the binding of PD-1 antigen to PD-L1 and PD-L2 with IC50 of 3.0 and 3.1 nmol/L, respectively. Furthermore, JS-001 displayed distinct species cross-reactivity: it could bind to the PD-1 antigen on the peripheral blood mononuclear cells (PBMCs) of humans and cynomolgus monkeys, but not to those of mice and woodchucks; the Kd values for the interaction between JS-001 and PD-1 antigens on CD8+ T cells of human and cynomolgus monkey were 2.1 nmol/L and 1.2 nmol/L, respectively. In vitro, treatment with JS-001 (0.01–10 μg/mL) dose-dependently stimulated human T cell proliferation, as well as IFN-γ and TNF-α secretion. In HBsAg-vaccinated cynomolgus monkeys, the expression of PD-1+/CD4+ and PD-1+/CD8+ was significantly elevated, intramuscular injection of JS-001 (1 and 10 mg/kg) resulted in dramatic decreases in PD-1+/CD4+ and PD-1+/CD8+ expression in a dose-dependent manner, which was supported by PD-1 receptor occupancy (RO) results. In the PK study, 18 cynomolgus monkeys treated with single, ascending doses of 1, 10, and 75 mg/kg, and another 6 cynomolgus monkeys received 10 mg/kg successive administration. The plasma clearance of JS-001 followed a linear PK profile with single administration in the 1 and 10 mg/kg groups and a non-linear PK profile in the 75 mg/kg group. In the successive 10 mg/kg administration group, no drug accumulation was observed. But the AUC from the last exposure was lower than that of the first administration, which was probably due to the production of ADAs, as demonstrated in immunogenicity study. These non-clinical data are encouraging and provide a basis for the efficacy and safety of JS-001 in clinical trials.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
McDermott DF, Atkins MB . PD-1 as a potential target in cancer therapy. Cancer Med 2013; 2: 662–73.
Velu V, Titanji K, Zhu B, Husain S, Pladevega A, Lai L, et al. Enhancing SIV-specific immunity in vivo by PD-1 blockade. Nature 2009; 458: 206–10.
Fuller MJ, Callendret B, Zhu B, Freeman GJ, Hasselschwert DL, Satterfield W, et al. Immunotherapy of chronic hepatitis C virus infection with antibodies against programmed cell death-1 (PD-1). Proc Natl Acad Sci U S A 2013; 110: 15001–6.
Richter C, Thieme S, Bandoła J, Laugsch M, Anastassiadis K, Brenner S . Generation of inducible immortalized dendritic cells with proper immune function in vitro and in vivo. PLoS One 2013; 8: e62621.
Rizvi NA, Mazières J, Planchard D, Stinchcombe TE, Dy GK, Antonia SJ, et al. Activity and safety of nivolumab, an anti-PD-1 immune checkpoint inhibitor, for patients with advanced, refractory squamous non-small-cell lung cancer (CheckMate 063): a phase 2, single-arm trial. Lancet Oncol 2015; 16: 257–65.
Brahmer JR, Drake CG, Wollner I, Powderly JD, Picus J, Sharfman WH, et al. Phase I study of single-agent anti–programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J Clin Oncol 2010; 28: 3167–75.
Gust L, D'journo XB . The use of correlation functions in thoracic surgery research. J Thorac Dis 2015; 7: E11–5.
Liu J, Zhang E, Ma Z, Wu W, Kosinska A, Zhang X, et al. Enhancing virus-specific immunity in vivo by combining therapeutic vaccination and PD-L1 blockade inchronic hepadnaviral infection. PLoS Pathog 2014; 10: e1003856.
Petrovas C, Casazza JP, Brenchley JM, Price DA, Gostick E, Adams WC, et al. PD-1 is a regulator of virus-specific CD8+ T cell survival in HIV infection. J Exp Med 2006; 203: 2281–92.
Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 2012; 366: 2443–54.
Brahmer JR, Tykodi SS, Chow LQ, Hwu WJ, Topalian SL, Hwu P, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med 2012; 366: 2455–65.
Li Y, Li F, Jiang F, Lv X, Zhang R, Lu A, et al. A mini-review for cancer immunotherapy: molecular understanding of PD-1/PD-L1 pathway & translational blockade of immune checkpoints. Int J Mol Sci 2016; 17: 1151.
Paulsen M, Janssen O . Pro- and anti-apoptotic CD95 signaling in T cells. Cell Commun Signal 2011; 9: 7.
Acknowledgements
This study was supported by National Thirteen Five Major Special Fund “Non-clinical and Clinical PK, PD, and Immunogenicity Evaluation of Biosimilar” (2015ZX09501008-006) and “Core Technology-based Non-clinical Evaluation of Biological macromolecular drugs” (2015ZX09501007-002). We thank Dr Qing-qing WANG and Zhe WANG for language assistance during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fu, J., Wang, F., Dong, Lh. et al. Preclinical evaluation of the efficacy, pharmacokinetics and immunogenicity of JS-001, a programmed cell death protein-1 (PD-1) monoclonal antibody. Acta Pharmacol Sin 38, 710–718 (2017). https://doi.org/10.1038/aps.2016.161
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/aps.2016.161
Keywords
This article is cited by
-
Efficacy and safety of lenvatinib combined with anti-PD-1 antibodies plus GEMOX chemotherapy as non-first-line systemic therapy in advanced gallbladder cancer
Cancer Immunology, Immunotherapy (2024)
-
Toripalimab combined with lenvatinib and GEMOX is a promising regimen as first-line treatment for advanced intrahepatic cholangiocarcinoma: a single-center, single-arm, phase 2 study
Signal Transduction and Targeted Therapy (2023)
-
Safety and clinical activity with an anti-PD-1 antibody JS001 in advanced melanoma or urologic cancer patients
Journal of Hematology & Oncology (2019)
-
Toripalimab: First Global Approval
Drugs (2019)