Abstract
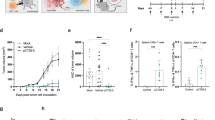
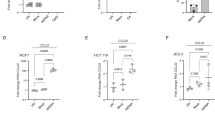
Chemokines are key regulators of both innate and adaptive immune responses. CCL4 (macrophage inflammatory protein-1β, MIP-1β) is a CC chemokine that has a broad spectrum of target cells including immature dendritic cells, which express the cognate receptor CCR5. We asked whether a plasmid encoding CCL4 is able to improve tumor protection and immune responses in a Her2/neu+ mouse tumor model. Balb/c mice were immunized twice intramuscularly with plasmid DNA on days 1 and 15. On day 25, a tumor challenge was performed with 2 × 105 syngeneic Her2/neu+ D2F2/E2 tumor cells. Different groups of mice were vaccinated with pDNA(Her2/neu) plus pDNA(CCL4), pDNA(Her2/neu), pDNA(CCL4) or mock vector alone. Our results show that CCL4 is able to (i) improve tumor protection and (ii) augment a TH1-polarized immune response against Her2/neu. Although Her2/neu-specific humoral and T-cell immune responses were comparable with that induced in previous studies using CCL19 or CCL21 as adjuvants, tumor protection conferred by CCL4 was inferior. Whether this is due to a different spectrum of (innate) immune cells, remains to be clarified. However, combination of CCL19/21 with CCL4 might be a reasonable approach in the future, particularly for DNA vaccination in Her2/neu+ breast cancer in the situation of minimal residual disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gurunathan S, Klinman DM, Seder RA . DNA vaccines: immunology, application and optimization. Annu Rev Immunol 2000; 18: 927–974.
Choo AY, Choo DK, Kim JJ, Weiner DB . DNA vaccination in immunotherapy of cancer. Cancer Res 2005; 123: 137–156.
Kutzler MA, Weiner DB . Developing DNA vaccines that call to dendritic cells. J Clin Invest 2004; 114: 1241–1244.
Rice J, Ottensmeier CH, Stevenson F . DNA vaccines: precision tools for activating Effective immunity against cancer. Nat Rev Immunol 2008; 8: 108–120.
Prud’homme G . DNA vaccination against tumors. J Gene Med 2005; 7: 3–17.
Iezzi M, Quaglino E, Amici A, Lollini P, Forni G, Cavallo F . DNA vaccination against oncoantigens.A promise. OncoImmunology 2012; 1: 316–325.
Babiuk LA, Pontarollo R, Babiuk S, Loehr B, van Drunen Littel-van den Hurk S . Induction of immune responses by DNA vaccines in large animals. Vaccine 2003; 21: 649–658.
Scheerlinck JP . Genetic adjuvants for DNA vaccines. Vaccine 2001; 19: 2647–2656.
Greenland JR, Letvin NL . Chemical adjuvants for plasmid DNA vaccines. Vaccine 2007; 25: 3731–3741.
Stewart TJ, Smyth MJ . Chemokine-chemokine receptors in cancer immunotherapy. Immunother 2009; 1: 109–127.
Bobanga ID, Petrosiute, Huang AY . Chemokines as cancer vaccine adjuvants. Vaccines 2013; 1: 444–462.
Allen SJ, Crown SE, Handel TM . Chemokine: receptor structure, interactions, and antagonism. Annu Rev Immunol 2007; 25: 787–920.
Homey B, Müller A, Zlotnik A . Chemokines: agents for the immunotherapy of cancer? Nat Rev immunol 2002; 2: 175–184.
Bromley SK, Mempel TR, Luster AD . Orchestrating the orchestrators: chemokines in control of T cell traffic. Nat Immunol 2008; 9: 970–980.
Viola A, Contento RL, Molon B . T cells and their partners: the chemokine dating agency. Trends Immunol 2006; 27: 421–427.
Eo SK, Lee S, Kumaraguru U, Rouse BT . Immunopotentiation of DNA vaccine against herpes simplex virus via co-delivery of plasmid DNA expressing CCR7 ligands. Vaccine 2001; 19: 4685–4693.
Toka FN, Gierynska M, Rouse BT . Codelivery of CCR7 ligands as molecular adjuvants enhances the protective immune response against herpes simplex virus type 1. J Virol 2003; 77: 12742–12752.
Braun SE, Chen K, Foster RG, Kim CH, Hromas R, Kaplan MH et al. The CC chemokine CK beta-11/MIP-3 beta/ELC/Exodus 3 mediates tumor rejection of murine breast cancer cells through NK cells. J Immunol 2000; 164: 4025–4031.
Hillinger S, Yang SC, Zhu L, Huang M, Duckett R, Atianzar K et al. EBV-induced molecule 1 ligand chemokine (ELC/CCL19) promotes IFN-gamma-dependent antitumor responses in a lung cancer model. J Immunol 2003; 171: 6457–6465.
Hillinger S, Yang SC, Batra RK, Striete RM, Weder W, Dubinett SM et al. CCL19 reduces tumour burden in a model of advanced lung cancer. Br J Cancer 2006; 94: 1029–1034.
Westermann J, Nguyen-Hoai T, Baldenhofer G, Höpken UE, Lipp M, Dörken B et al. CCL19 (ELC) as an adjuvant for DNA vaccination: induction of a TH1-type T-cell response and enhancement of antitumor immunity. Cancer Gene Ther 2007; 14: 523–532.
Nguyen-Hoai T, Baldenhofer G, Ahmed MS, Pham-Duc M, Gries M, Lipp M et al. CCL19 (ELC) improves TH1-polarized immune responses and protective immunity in a murine Her2/neu DNA vaccination model. J Gene Med 2012; 14: 128–137.
Nguyen-Hoai T, Baldenhofer G, Sayed Ahmed MS, Pham-Duc M, Lipp M, Dörken B et al. CCL21 (SLC) improves tumor by a DNA vaccine in a Her2/neu mouse tumor model. Cancer Gene Ther 2012; 19: 69–76.
Sharma S, Stolin M, Luo J, Strieter RM, Burdick M, Zhu LX et al. Secondary Lymphoid tissue chemokine mediates T cell-dependent antitumor responses in vivo. J Immunol 2000; 164: 4558–4563.
Sharma S, Stolina M, Zhu L, Lin Y, Batra R, Huang M et al. Secondary lymphoid organ chemokine reduces pulmonary tumor burden in spontaneous murine bronchoalveolar cell carcinoma. Cancer Res 2001; 61: 6406–6412.
Yang SC, Hillinger S, Riedl K, Zhang L, Zhu L, Huang M et al. Intratumoral administration of dendritic cells overexpressing CCL21 generates systemic antitumor responses and confers tumor immunity. Cancer Res 2004; 10: 2891–2901.
Yang SC, Batra RK, Hillinger S, Reckamp KL, Strieter RM, Dubinett SM et al. Intrapulmonary administration of CCL21 gene-modified dendritic cells reduces tumor burden in spontaneous murine bronchoalveolar cell carcinoma. Cancer Res 2006; 66: 3205–3213.
Qin H, Zhou C, Wang D, Ma W, Liang X, Lin C et al. Enhancement of antitumour immunity by a novel chemotactic antigen DNA vaccine encoding chemokines and multiepitopes of prostate-tumour-associated antigens. Immunology 2006; 117: 419–430.
Lillard JW, Singh UP, Boyaka PN, Singh S, Taub DD, McGhee JR . MIP-1a and MIP-1b differentially mediate mucosal and systemic adaptive immunity. Blood 2003; 101: 807–814.
Nguyen-Hoai T, Hohn O, Vu MD, Baldenhofer G, Sayed Ahmed MS, Dörken B et al. CCL19 as an adjuvant for intradermal gene gun immunization in a Her2/neu mouse tumor model: improved vaccine efficacy and a role for B cells as APC. Cancer Gene Ther 2012; 19: 880–887.
Lindencrona JA, Preiss S, Kammertoens T, Schuler T, Piechocki M, Wie WZ et al. CD4+T cell-mediated HER-2/neu-specific tumor rejection in the absence of B cells. Int J Cancer 2004; 109: 259–264.
Nagata Y, Furugen R, Hiasa A . Peptides derived from a wild-type murine proto-oncogene c-erbB-2/HER2/neu can induce CTL and tumor suppression in syngeneic hosts. J Immunol 1997; 159: 1336–1343.
Correa I, Plunkett T . Update on HER-2 as a target for cancer therapy: HER2/neu peptides as tumour vaccines for T cell recognition. Breast Cancer Res 2001; 3: 399–403.
Vertuani S, Sette A, Sidney J . Improved immunogenicity of an immunodominant epitope of the HER-2/neu protooncogene by alterations of MHC contact residues. J Immunol 2004; 172: 3501–3508.
Chen D, Murphy B, Sung R, Bromberg JS . Adaptive and innate immune responses to gene transfer vectors: role of cytokines and chemokines in vector function. Gene Ther 2003; 10: 991–998.
Novak L, Igoucheva O, Cho S, Alexeev V . Characterization of the CCL21-mediated melanoma-specific immune responses and in situ melanoma eradication. Mol Cancer Ther 2007; 6: 1755–1764.
Thanarajasingam U, Sanz L, Diaz R, Qiao J, Sanchez-Perez L, Kottke T et al. Delivery of CCL21 to metastatic disease improves the efficacy of adoptive T-cell therapy. Cancer Res 2007; 67: 300–308.
Song R, Liu S, Leong KW . Effects of MIP-1 alpha, MIP-3 alpha, and MIP-3 beta on the induction of HIV Gag-specific immune response with DNA vaccines. Mol Ther 2007; 15: 1007–1015.
Song JH, Kim JI, Kwon HJ, Shim DH, Parajuli N, Cuburu N et al. CCR7- CCL19/CCL21-regulated dendritic cells are responsible for effectiveness of sublingual vaccination. J Immunol 2009; 182: 6851–6860.
Song S, Liu C, Wang J, Zhang Y, You H, Wang Y et al. Vaccination with combination of Flt3L and RANTES in a DNA prime-protein boost regimen elicits strong cell mediated immunity and antitumor effect. Vaccine 2009; 27: 1111–1118.
Rainone V, Dubois G, Temchura V, Uberla K, Clivio A, Nebuloni M et al. CCL28 Induces Mucosal Homing of HIV-1-Specific IgA-Secreting Plasma Cells in Mice Immunized with HIV-1 Virus-Like Particles. PLoS One 2011; 6: e26979.
Kar UK, Srivastava MK, Andersson A, Baratelli F, Huang M, Kickhoefer V et al. A Novel CCL21-vault nanocapsule intratumoral delivery inhibits lung cancer growth. PLoS One 2011; 6: e18758.
Franciszkiewicz K, Boissonnas A, Boutet M, Combadiere C, Mami-Chouai F . Role of chemokines and chemokine receptors in shaping the effector phase of the antitumor immune response. Cancer Res 2012; 72: 6325–6332.
Igoucheva O, Jonas R, Alexeev V . Chemokine-enhanced DNA vaccination in cancer immunotherapy. Oncoimmunology 2013; 2: e26092.
Yan YH, Qi SC, Su LK, Xu QA, Fan MW . Co-delivery of ccl19 gene enhances anti-caries DNA vaccine pCIA-P immunogenicity in mice by increasing dendritic cell migration to secondary lymphoid tissues. Acta Pharmacol Sin 2013; 34: 432–440.
Hartoonian C, Sepehrizadeh Z, Tabatabai Yazdi M, Jang YS, Langroudi L, Amir Kalvanagh P et al. Enhancement of immune responses by co-delivery of CCL19/MIP-3beta chemokine plasmid with HCV core DNA/protein immunization. Hepat Mon 2014; 14: e14611.
Curcio C, Di Carlo E, Clynes R, Smyth MJ, Boggio K, Quaglino E et al. Nonredundant roles of antibody, cytokines, and perforin in the eradication of established Her-2/neu carcinomas. J Clin Invest 2003; 111: 1161–1170.
Bachmann MF, Kopf M, Marsland BJ . Chemokines: more than just road signs. Nat Rev Immunol 2006; 6: 159–164.
Wuest TR, Carr DJ . The role of chemokines during herpes simplex virus-1 infection. Front Biosci 2008; 13: 4862–4872.
Acknowledgements
This study was partially supported by a generous grant by 'Music against Cancer e.V.'
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Nguyen-Hoai, T., Pham-Duc, M., Gries, M. et al. CCL4 as an adjuvant for DNA vaccination in a Her2/neu mouse tumor model. Cancer Gene Ther 23, 162–167 (2016). https://doi.org/10.1038/cgt.2016.9
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cgt.2016.9
This article is cited by
-
The future of cancer immunotherapy: DNA vaccines leading the way
Medical Oncology (2023)
-
Cancer DNA vaccines: current preclinical and clinical developments and future perspectives
Journal of Experimental & Clinical Cancer Research (2019)
-
Recent developments in targeted imaging of CXCR4-chemokine receptor
Journal of Radioanalytical and Nuclear Chemistry (2018)
-
Synergistic effect of programmed cell death protein 1 blockade and secondary lymphoid tissue chemokine in the induction of anti-tumor immunity by a therapeutic cancer vaccine
Archives of Virology (2017)