Abstract
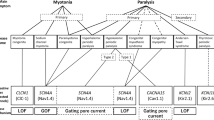
Non-dystrophic myotonias (NDMs) are caused by mutations in CLCN1 or SCN4A. The purpose of the present study was to optimize the genetic characterization of NDM in The Netherlands by analysing CLCN1 and SCN4A in tandem. All Dutch consultant neurologists and the Dutch Patient Association for Neuromuscular Diseases (Vereniging Spierziekten Nederland) were requested to refer patients with an initial diagnosis of NDM for clinical assessment and subsequent genetic analysis over a full year. Based on clinical criteria, sequencing of either CLCN1 or SCN4A was performed. When previously described mutations or novel mutations were identified in the first gene under study, the second gene was not sequenced. If no mutations were detected in the first gene, the second gene was subsequently also analysed. Underlying NDM mutations were explored in 54 families. In total, 20% (8 of 40) of our probands with suspected chloride channel myotonia showed no CLCN1 mutations but subsequent SCN4A screening revealed mutations in all of them. All 14 probands in whom SCN4A was primarily sequenced showed a mutation. In total, CLCN1 mutations were identified in 32 families (59%) and SCN4A in 22 (41%), resulting in a diagnostic yield of 100%. The yield of mutation detection was 93% with three recessive and three sporadic cases not yielding a second mutation. Among these mutations, 13 in CLCN1 and 3 in SCN4A were novel. In conclusion, the current results show that in tandem analysis of CLCN1 and SCN4A affords high-level mutation ascertainment in families with NDM.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Rüdel R, Ricker K, Lehmann-Horn F : Genotype–phenotype correlations in human skeletal muscle sodium channel diseases. Arch Neurol 1993; 50: 1241–1248.
Bryant SH, Morales-Aguilera A : Chloride conductance in normal and mytonic muscle fibres and the action of monocarboxylic aromatic acids. J Physiol 1971; 219: 367–383.
Rüdel R, Ricker K, Lehmann-Horn F : Transient weakness and altered membrane characteristic in recessive generalized myotonia (Becker). Muscle Nerve 1988; 11: 202–211.
Lipicky RJ, Bryant SH : A biophysical study of the human myotonias. In: Desmedt J (ed).: New Developments in Electromyography and Clinical Neurophysiology, Vol. 1 Basel: Karger, 1973, pp 451–563.
Koch MC, Steinmeyer K, Lorenz C et al: The skeletal muscle chloride channel in dominant and recessive human myotonia. Science 1992; 257: 797–800.
Abdalla JA, Casley WL, Cousin HK et al: Linkage of Thomsen disease to the T-cell-receptor beta (TCRB) locus on chromosome 7q35. Am J Hum Genet 1992; 51: 579–584.
Colding-Jørgensen E : Phenotypic variability in myotonia congenita. Muscle Nerve 2005; 32: 19–34.
Lehmann-Horn F, Rüdel R, Dengler R, Lorkovic H, Haass A, Ricker K : Membrane defects in paramyotonia congenita with and without myotonia in a warm environment. Muscle Nerve 1981; 4: 396–406.
Ebers GC, George Jr AL, Barchi RL et al: Paramyotonia congenita and hyperkalemic periodic paralysis are linked to the adult muscle sodium channel gene. Ann Neurol 1991; 30: 810–816.
Lerche H, Heine R, Pika U et al: Human sodium channel myotonia: slowed channel inactivation due to substitutions for a glycine within the III-IV linker. J Physiol 1993; 470: 13–22.
Ricker K, Moxley III RT, Heine R, Lehmann-Horn F : Myotonia fluctuans. A third type of muscle sodium channel disease. Arch Neurol 1994; 51: 1095–1102.
Cannon SC : Spectrum of sodium channel disturbances in the nondystrophic myotonias and periodic paralyses. Kidney Int 2000; 57: 772–779.
Meyer-Kleine C, Steinmeyer K, Ricker K, Jentsch TJ, Koch MC : Spectrum of mutations in the major human skeletal muscle chloride channel gene (CLCN1) leading to myotonia. Am J Hum Genet 1995; 57: 1325–1334.
Zhang J, George Jr AL, Griggs RC et al: Mutations in the human skeletal muscle chloride channel gene (CLCN1) associated with dominant and recessive myotonia congenita. Neurology 1996; 47: 993–998.
Sangiuolo F, Botta A, Mesoraca A et al: Identification of five new mutations and three novel polymorphisms in the muscle chloride channel gene (CLCN1) in 20 Italian patients with dominant and recessive myotonia congenita. Hum Mutat 1997; 11: 331.
Sun C, Tranebjaerg L, Torbergsen T, Holmgren G, Van Ghelue M : Spectrum of CLCN1 mutations in patients with myotonia congenita in Northern Scandinavia. Eur J Hum Genet 2001; 9: 903–909.
Plassart-Schiess E, Gervais A, Eymard B et al: Novel muscle chloride channel (CLCN1) mutations in myotonia congenita with various modes of inheritance including incomplete dominance and penetrance. Neurology 1998; 50: 1176–1179.
Lehmann-Horn F, Rüdel R : Non-dystrophic myotonias and periodic paralyses.In: Emery AEH (ed).: Diagnostic Criteria for Neuromuscular Disorders. London: Royal Society of Medicine Press, 1997, pp 31–36.
Miller SA, Dykes DD, Polesky HF : A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 1988; 16: 1215.
Ginjaar HB, van der Kooi AJ, Ceelie H et al: Sarcoglycanopathies in Dutch patients with autosomal recessive limb girdle muscular dystrophy. J Neurol 2000; 247: 524–529.
Lorenz C, Meyer-Kleine C, Steinmeyer K, Koch MC, Jentsch TJ : Genomic organization of the human muscle chloride channel CIC-1 and analysis of novel mutations leading to Becker-type myotonia. Hum Mol Genet 1994; 3: 941–946.
Sloan-Brown K, George Jr AL : Inheritance of three distinct muscle chloride channel gene (CLCN1) mutations in a single recessive myotonia congenita family. Neurology 1997; 48: 542–543.
George Jr AL, Sloan-Brown K, Fenichel GM, Mitchell GA, Spiegel R, Pascuzzi RM : Nonsense and missense mutations of the muscle chloride channel gene in patients with myotonia congenita. Hum Mol Genet 1994; 3: 2071–2072.
Kubisch C, Schmidt-Rose T, Fontaine B, Bretag AH, Jentsch TJ : ClC-1 chloride channel mutations in myotonia congenita: variable penetrance of mutations shifting the voltage dependence. Hum Mol Genet 1998; 7: 1753–1760.
Meyer-Kleine C, Ricker K, Otto M, Koch MC : A recurrent 14 bp deletion in the CLCN1 gene associated with generalized myotonia (Becker). Hum Mol Genet 1994; 3: 1015–1016.
Steinmeyer K, Lorenz C, Pusch M, Koch MC, Jentsch TJ : Multimeric structure of ClC-1 chloride channel revealed by mutations in dominant myotonia congenita (Thomsen). EMBO J 1994; 13: 737–743.
Pusch M : Myotonia caused by mutations in the muscle chloride channel gene CLCN1. Hum Mutat 2002; 19: 423–434.
Papponen H, Toppinen T, Baumann P et al: Founder mutations and the high prevalence of myotonia congenita in Northern Finland. Neurology 1999; 53: 297–302.
Rosenfeld J, Sloan-Brown K, George Jr AL : A novel muscle sodium channel mutation causes painful congenital myotonia. Ann Neurol 1997; 42: 811–814.
Sugiura Y, Aoki T, Sugiyama Y, Hida C, Ogata M, Yamamoto T : Temperature-sensitive sodium channelopathy with heat-induced myotonia and cold-induced paralysis. Neurology 2000; 54: 2179–2181.
Ptácek LJ, Tawil R, Griggs RC et al: Sodium channel mutations in acetazolamide-responsive myotonia congenita, paramyotonia congenita and hyperkalemic periodic paralysis. Neurology 1994; 44: 1500–1503.
McClatchey AI, Van den Bergh P, Pericak-Vance MA et al: Temperature-sensitive mutations in the III-IV cytoplasmic loop region of the skeletal muscle sodium channel gene in paramyotonia congenita. Cell 1992; 68: 769–774.
Yamada T, Ochi H, Hara H, Yoshimura T, Kobayashi T : A skeletal muscle sodium channel mutation in a Japanese family with paramyotonia congenita. J Neurol Sci 1995; 133: 192–193.
Ptácek LJ, George Jr AL, Barchi RL et al: Mutations in an S4 segment of the adult skeletal muscle sodium channel cause paramyotonia congenita. Neuron 1992; 8: 891–897.
Bulman DE : Phenotype variation and newcomers in ion channel disorders. Hum Mol Genet 1997; 6: 1679–1685.
Wu FF, Ryan A, Devaney J et al: Novel CLCN1 mutations with unique clinical and electrophysiological consequences. Brain 2002; 125: 2392–2407.
Grunnet M, Jespersen T, Colding-Jørgensen E et al: Characterization of two new dominant ClC-1 channel mutations associated with myotonia. Muscle Nerve 2003; 28: 722–732.
Colding-Jørgensen E, Dunø M, Schwartz M, Vissing J : Decrement of compound muscle action potential is related to mutation type in myotonia congenita. Muscle Nerve 2003; 27: 449–455.
Estévez R, Pusch M, Ferrer-Costa C, Orozco M, Jentsch TJ : Functional and structural conservations of CBS domains from CLC chloride channels. J Physiol 2004; 557: 363–378.
Tsujino A, Maertens C, Ohno K et al: Myasthenic syndrome caused by mutation of the SCN4A sodium channel. Proc Natl Acad Sci USA 2003; 100: 7377–7382.
Bendahhou S, Cummins TR, Kula RW, Fu YH, Ptácek LJ : Impairment of slow inactivation as a common mechanism for periodic paralysis in DIIS4-S5. Neurology 2002; 58: 1266–1272.
Simpson BJ, Height TA, Rychkov GY et al: Characterization of three myotonia-associated mutations of the CLCN1 chloride channel gene via heterologous expression. Hum Mutat 2004; 24: 185.
Bouhours M, Sternberg D, Davoine CS et al: Functional characterization and cold sensitivity of T1313A, a new mutation of the skeletal muscle sodium channel causing paramyotonia congenita in humans. J Physiol 2004; 554: 635–647.
Jurkat-Rott K, Lehmann-Horn F : Muscle channelopathies and critical points in functional and genetic studies. J Clin Invest 2005; 115: 2000–2009.
Dutzler R, Campbell EB, Cadene M, Chait BT, MacKinnon R : X-ray structure of a ClC chloride channel at 3.0 Å reveals the molecular basis of anion selectivity. Nature 2002; 415: 287–294.
Acknowledgements
We thank the VSN for their cooperation in this study, and Linda Vijfhuizen and Naima Lamzira for their expert technical assistance and G van Zonneveld for illustrating figures. This study was supported by Prinses Beatrix Fonds (MAR04-0118), The Netherlands.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trip, J., Drost, G., Verbove, D. et al. In tandem analysis of CLCN1 and SCN4A greatly enhances mutation detection in families with non-dystrophic myotonia. Eur J Hum Genet 16, 921–929 (2008). https://doi.org/10.1038/ejhg.2008.39
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ejhg.2008.39
Keywords
This article is cited by
-
Community data-driven approach to identify pathogenic founder variants for pan-ethnic carrier screening panels
Human Genomics (2023)
-
Non-dystrophic myotonias: clinical and mutation spectrum of 70 German patients
Journal of Neurology (2021)
-
Association of Three Different Mutations in the CLCN1 Gene Modulating the Phenotype in a Consanguineous Family with Myotonia Congenita
Journal of Molecular Neuroscience (2021)
-
Up-regulation of voltage-gated sodium channels by peptides mimicking S4-S5 linkers reveals a variation of the ligand-receptor mechanism
Scientific Reports (2020)
-
Clinical exome sequencing in neuromuscular diseases: an experience from Turkey
Neurological Sciences (2020)