Abstract
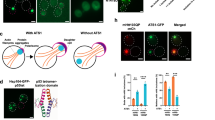
Huntington's disease is caused by CAG trinucleotide expansions in the gene encoding huntingtin. N- terminal fragments of huntingtin with polyglutamine produce aggregates in the endosome-lysosomal system, where proteolytic fragments of huntingtin is generated. Heat shock protein 70 (HSP70) prevents the formation of protein aggregates, but the effect of HSP70 on the huntingtin in the endosome-lysosomal system is unknown. This study was to determine whether HSP70 alters the distribution of huntingtin in endosome-lysosomal system. HSP70 expressing stable cells (NIH/3T3 or cerebral hybrid cell line A1) were generated, and mutant [(CAG)100] huntingtin was transiently overexpressed. Analysis of subcellular distribution by immnuocytochemistry or proteolysis cleavage by Western blotting was performed. 18 CAG repeat wild type [WT; (CAG)18] huntingtin was used as a control. Cells with huntingtin showed patterns of endosome- lysosomal accumulation, from a 'dispersed vacuole (DV)' type into a coalescent 'perinuclear vacuole (PV)' type over time. In WT huntingtin, HSP70 increased the cells with the PV types that enhanced the proteolytic cleavage of huntingtin. However, HSP70 reduced cells of the DV and PV types expressing mutant huntingtin, that result in less proteolysis than that of control. In addition, intranuclear inclusions were formed only in mutant cells, which was not affected by HSP70. These results suggest that HSP70 alters the distribution of huntingtin in the endosome-lysosomal system, and that this contributes to huntingtin proteolysis.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Kang, B., Ahn, JY., Kim, M. et al. Heat shock protein 70 alters the endosome-lysosomal localization of huntingtin. Exp Mol Med 39, 38–46 (2007). https://doi.org/10.1038/emm.2007.5
Published:
Issue date:
DOI: https://doi.org/10.1038/emm.2007.5