Abstract
Purpose
To analyze the relation between ophthalmologic and motor changes in spinocerebellar ataxia type 7 (SCA7).
Patients and methods
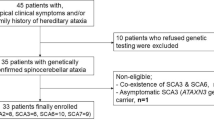
This was a case series study. Sixteen SCA7 patients underwent a comprehensive ophthalmic examination, including ocular extrinsic motility testing, color vision test, and optical coherence tomography of the optic nerve and macula. Changes in the corneal endothelium, electroretinographic patterns, and a complete neurologic evaluation using the Scale for the Assessment and Rating of Ataxia (SARA) were evaluated. Correlations of endothelial cell density (ECD) with number of CAG repetitions and the SARA scores were estimated.
Results
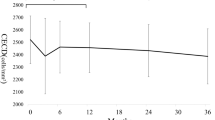
All patients showed various degrees of visual impairment mainly due to macular deterioration. Notably, they also presented decreased ECD. Pairwise correlations of ECD with number of CAG repeats and severity of motor symptoms quantified with the SARA scores were inverse (r=−0.46, P=0.083 and r=−0.64, P=0.009, respectively). Further analyses indicated an average ECD decrease of 48 cells/mm2 (P=0.006) per unit of change on the number of CAG repeats, and of 75 cells/mm2 (P=0.001) per unit of change on the SARA scores.
Conclusions
The results agree with previous ophthalmological findings regarding the widespread effect of SCA7 mutation on the patient’s visual system. However, the results also show a significant negative correlation of decreased ECD with both CAG repetitions and SARA scores. This suggests that motor systems could degenerate in parallel with visual systems, although more research is needed to determine whether the degeneration is caused by the same mechanisms.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Rub U, Schols L, Paulson H, Auburger G, Kermer P, Jen JC et al. Clinical features, neurogenetics and neuropathology of the polyglutamine spinocerebellar ataxias type 1, 2, 3, 6 and 7. Prog Neurobiol 2013; 104: 38–66.
Paulson HL . The spinocerebellar ataxias. J Neuroophthalmol 2009; 29 (3): 227–237.
Teive HA, Munhoz RP, Arruda WO, Lopes-Cendes I, Raskin S, Werneck LC et al. Spinocerebellar ataxias: genotype-phenotype correlations in 104 Brazilian families. Clinics (Sao Paulo) 2012; 67 (5): 443–449.
Michalik A, Martin JJ, Van Broeckhoven C . Spinocerebellar ataxia type 7 associated with pigmentary retinal dystrophy. Eur J Hum Genet 2004; 12 (1): 2–15.
Hernandez-Castillo C, Alcauter S, Galvez V, Barrios F, Yescas P, Ochoa A et al. Disruption of visual and motor connectivity in spinocerebellar ataxia type 7. Mov Disord 2013; 28 (12): 1708–1716.
Enevoldson TP, Sanders MD, Harding AE . Autosomal dominant cerebellar ataxia with pigmentary macular dystrophy. A clinical and genetic study of eight families. Brain 1994; 117 (Pt 3): 445–460.
Gouw LG, Kaplan CD, Haines JH, Digre KB, Rutledge SL, Matilla A et al. Retinal degeneration characterizes a spinocerebellar ataxia mapping to chromosome 3p. Nat Genet 1995; 10 (1): 89–93.
Lebre AS, Brice A . Spinocerebellar ataxia 7 (SCA7). Cytogenet Genome Res 2003; 100 (1-4): 154–163.
Hugosson T, Granse L, Ponjavic V, Andreasson S . Macular dysfunction and morphology in spinocerebellar ataxia type 7 (SCA 7). Ophthalmic Genet 2009; 30 (1): 1–6.
Manrique RK, Noval S, Aguilar-Amat MJ, Arpa J, Rosa I, Contreras I . Ophthalmic features of spinocerebellar ataxia type 7. J Neuroophthalmol 2009; 29 (3): 174–179.
Garcia-Velazquez LE, Canizales-Quinteros S, Romero-Hidalgo S, Ochoa-Morales A, Martinez-Ruano L, Marquez-Luna C et al. Founder effect and ancestral origin of the spinocerebellar ataxia type 7 (SCA7) mutation in Mexican families. Neurogenetics 2014; 15 (1): 13–17.
Schmitz-Hubsch T, du Montcel ST, Baliko L, Berciano J, Boesch S, Depondt C et al. Scale for the assessment and rating of ataxia: development of a new clinical scale. Neurology 2006; 66 (11): 1717–1720.
Molina-Rey D, Gomez A . Evaluación por décadas de edad del comportamiento de las células endoteliales corneales en población mexicana. Rev Mex Oftalmol 2005; 79 (2): 93–100.
Stevanin G, David G, Durr A, Giunti P, Benomar A, Abada-Bendib M et al. Multiple origins of the spinocerebellar ataxia 7 (SCA7) mutation revealed by linkage disequilibrium studies with closely flanking markers, including an intragenic polymorphism (G3145TG/A3145TG). Eur J Hum Genet 1999; 7 (8): 889–896.
Magana JJ, Gomez R, Maldonado-Rodriguez M, Velazquez-Perez L, Tapia-Guerrero YS, Cortes H et al. Origin of the spinocerebellar ataxia type 7 gene mutation in Mexican population. Cerebellum 2013; 12 (6): 902–905.
Garden GA, La Spada AR . Molecular pathogenesis and cellular pathology of spinocerebellar ataxia type 7 neurodegeneration. Cerebellum 2008; 7 (2): 138–149.
Smith DC, Atadzhanov M, Mwaba M, Greenberg LJ . Evidence for a common founder effect amongst South African and Zambian individuals with Spinocerebellar ataxia type 7. J Neurol Sci 2015; 354 (1-2): 75–78.
Martin JJ . Spinocerebellar ataxia type 7. Handb Clin Neurol 2012; 103: 475–491.
Martin J, Van Regemorter N, Del-Favero J, Lofgren A, Van Broeckhoven C . Spinocerebellar ataxia type 7 (SCA7) - correlations between phenotype and genotype in one large Belgian family. J Neurol Sci 1999; 168 (1): 37–46.
Velazquez-Perez L, Rodriguez-Labrada R, Cruz-Rivas EM, Fernandez-Ruiz J, Vaca-Palomares I, Lilia-Campins J et al. Comprehensive study of early features in spinocerebellar ataxia 2: delineating the prodromal stage of the disease. Cerebellum 2014; 13 (5): 568–579.
Larson E, Fyfe I, Morton AJ, Monckton DG . Age-, tissue- and lenght-dependent bidirectional somatic CAG-CTG repeat instability in an allelic series of R6/2 Huntington disease mice. Neurobiol Dis 2015; 76: 98–111.
Olejniczak M, Urbanek MO, Krzyzosiak WJ . The role of the immune system in triplet repeat expansion diseases. Mediat Inflamm 2015; 2015: 873860.
Soliman AZ, Xing C, Radwan SH, Gong X, Mootha VV . Correlation of severity of Fuchs endothelial corneal dystrophy with triplet repeat expansion in TCF4. JAMA Ophthalmol 2015; 133: 1386–1391.
Mootha VV, Hussain I, Cunnusamy K, Graham E, Gong X, Neelam S et al. TCF4 triplet repeat expansion and nuclear RNA foci in Fuchs’ endothelial corneal dystrophy. Invest Ophthalmol Vis Sci 2015; 56 (3): 2003–2011.
Aleman TS, Cideciyan AV, Volpe NJ, Stevanin G, Brice A, Jacobson SG . Spinocerebellar ataxia type 7 (SCA7) shows a cone-rod dystrophy phenotype. Exp Eye Res 2002; 74 (6): 737–745.
Miller RC, Tewari A, Miller JA, Garbern J, Van Stavern GP . Neuro-ophthalmologic features of spinocerebellar ataxia type 7. J Neuroophthalmol 2009; 29 (3): 180–186.
Chen S, Peng GH, Wang X, Smith AC, Grote SK, Sopher BL et al. Interference of Crx-dependent transcription by ataxin-7 involves interaction between the glutamine regions and requires the ataxin-7 carboxy-terminal region for nuclear localization. Hum Mol Genet 2004; 13 (1): 53–67.
Williams AJ, Paulson HL . Polyglutamine neurodegeneration: protein misfolding revisited. Trends Neurosci 2008; 31 (10): 521–528.
Durr A . Autosomal dominant cerebellar ataxias: polyglutamine expansions and beyond. Lancet Neurol 2010; 9 (9): 885–894.
Takahashi T, Katada S, Onodera O . Polyglutamine diseases: where does toxicity come from? what is toxicity? where are we going? J Mol Cell Biol 2010; 2 (4): 180–191.
Yvert G, Lindenberg KS, Picaud S, Landwehrmeyer GB, Sahel JA, Mandel JL . Expanded polyglutamines induce neurodegeneration and trans-neuronal alterations in cerebellum and retina of SCA7 transgenic mice. Hum Mol Genet 2000; 9 (17): 2491–2506.
Acknowledgements
Partially funded by DGAPA-PAPIIT IN221413 CONACYT 0220871 and Fund Research of Instituto de Oftalmologia Conde de Valenciana. The funding organizations had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Campos-Romo, A., Graue-Hernandez, E., Pedro-Aguilar, L. et al. Ophthalmic features of spinocerebellar ataxia type 7. Eye 32, 120–127 (2018). https://doi.org/10.1038/eye.2017.135
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/eye.2017.135
This article is cited by
-
Long-Term Follow-Up before and during Riluzole Treatment in Six Patients from Two Families with Spinocerebellar Ataxia Type 7
The Cerebellum (2024)
-
Clinical value of vestibulo-ocular reflex in the differentiation of spinocerebellar ataxias
Scientific Reports (2023)
-
A Proposal for Classification of Retinal Degeneration in Spinocerebellar Ataxia Type 7
The Cerebellum (2021)
-
Longitudinal Analysis of the Relation Between Clinical Impairment and Gray Matter Degeneration in Spinocerebellar Ataxia Type 7 Patients
The Cerebellum (2021)
-
Molecular Targets and Therapeutic Strategies in Spinocerebellar Ataxia Type 7
Neurotherapeutics (2019)