Abstract
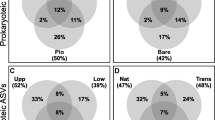
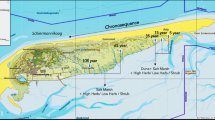
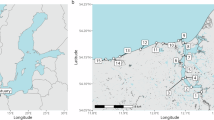
A primary focus among microbial ecologists in recent years has been to understand controls on the distribution of microorganisms in various habitats. Much less attention has been paid to the way that environmental disturbance interacts with processes that regulate bacterial community composition. We determined how human disturbance affected the distribution and community structure of salt marsh sediment bacteria by using denaturing gradient gel electrophoresis of 16S rRNA in five different habitats in each of four salt marshes located in northeastern Massachusetts, USA. Two of the four marsh creeks were experimentally enriched 15 × above background by the addition of nitrogen and phosphorus fertilizers for two or more growing seasons. Our results indicate that extrinsic factors acting at broad scales do not influence the distribution of salt marsh sediment bacteria. Intrinsic factors, controlled by local-scale environmental heterogeneity, do play a role in structuring these sediment microbial communities, although nutrient enrichment did not have a consequential effect on the microbial community in most marsh habitats. Only in one habitat, a region of the marsh creek wall that is heavily colonized by filamentous algae, did we see any effect of fertilization on the microbial community structure. When similar habitats were compared among marshes, there was considerable convergence in the microbial community composition during the growing season. Environmental factors that correlated best with microbial community composition varied with habitat, suggesting that habitat-specific intrinsic forces are primarily responsible for maintaining microbial diversity in salt marsh sediments.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Bass-Becking LGM . (1934). Geobiologie of Inleiding tot de Milieukunde. WP Van Stockum and Zoon: The Hague, the Netherlands.
Blum LK, Roberts MS, Garland JL, Mills AL . (2004). Distribution of microbial communities associated with the dominant high marsh plants and sediments of the United States east coast. Microb Ecol 48: 375–388.
Bowen JL, Crump BC, Deegan LA, Hobbie JE . (2009). Increased supply of ambient nitrogen has minimal effect on salt marsh bacterial production. Limnol Oceanogr 54: 713–722.
Buesing N, Gessner MO . (2003). Incorporation of radiolabeled leucine into protein to estimate bacterial production in plant litter, sediment, epiphytic biofilms, and water samples. Microb Ecol 45: 291–301.
Cho JC, Tiedje JM . (2000). Biogeography and degree of endemicity of fluorescent Pseudomonas strains in soil. Appl Environ Microbiol 66: 5448–5456.
Chrost RJ, Siuda W . (2006). Microbial production, utilization, and enzymatic degradation of organic matter in the upper trophogenic layer in the pelagial zone of lakes along a eutrophication gradient. Limnol Oceanogr 51: 749–762.
Clarke KR . (1993). Non-parametric multivariate analysis of changes in community structure. Aust J Ecol 18: 117–143.
Clarke KR, Warwick RM . (2001). Changes in Marine Communities: An Approach to Statistical Analysis and Interpretation, 2nd edn. Primer-E Ltd.: Plymouth, Marine Laboratory, Plymouth UK, 177pp.
Córdova-Kreylos AL, Cao Y, Green PG, Hwang HM, Kuivila KM, LaMontagne MG et al. (2006). Diversity, composition, and geographical distribution of microbial communities in California salt marsh sediments. Appl Environ Microbiol 72: 3357–3366.
Crump BC, Adams HE, Hobbie JE, Kling GW . (2007). Biogeography of bacterioplankton in lakes and streams of an arctic tundra catchment. Ecology 88: 1365–1478.
Crump BC, Hobbie JE . (2005). Synchrony and seasonality in bacterioplankton communities of two temperate rivers. Limnol Oceanogr 50: 1718–1729.
Crump BC, Kling GW, Bahr M, Hobbie JE . (2003). Bacterioplankton community shifts in an arctic lake correlate with seasonal changes in organic matter source. Appl Environ Microbiol 69: 2253–2268.
Deegan LA, Bowen JL, Drake D, Fleeger JW, Friedrichs CT, Galván CT et al. (2007). Susceptibility of salt marshes to nutrient enrichment and predator removal. Ecol Appl 17: S42–S63.
Drake DC, Peterson BJ, Deegan LA, Harris LA, Miller EE, Warren RS . (2008). Plant nitrogen dynamics in fertilized and natural New England salt marshes: a paired 15N tracer study. Mar Ecol Prog Ser 354: 35–46.
EPA (2002). Mid-Atlantic Integrated Assessment (MAIA) Estuaries 1997–1998. Summary Report. U.S. Environmental Protection Agency. Office of Research and Development. National Health and Environmental Effects Research Laboratory, Atlantic Ecology Division. Narragansett, RI 02882. 115 pp.
Fierer N, Jackson RB . (2006). The diversity and biogeography of soil bacterial communities. Proc Natl Acad Sci USA 103: 626–631.
Fierer N, Morse JL, Berthrong ST, Bernhardt ES, Jackson RB . (2007). Environmental controls on landscape-scale biogeography of stream bacterial communities. Ecology 88: 2162–2173.
Franklin RB, Blum LK, McComb AC, Mills AL . (2002). A geostatistical analysis of small-scale spatial variability in bacterial abundance and community structure in salt marsh creek bank sediments. FEMS Microbiol Ecol 42: 71–80.
Fuhrman JA, Hewson I, Schwalbach MS, Steele JA, Brown MV, Naeem S . (2006). Annually reoccurring bacterial communities are predictable from ocean conditions. Proc Natl Acad Sci USA 103: 13104–13109.
Green J, Bohannan BJM . (2006). Spatial scaling of microbial biodiversity. Trends Ecol Evol 21: 501–507.
Hewson I, Jacobson/Meyers ME, Fuhrman JA . (2007). Diversity and biogeography of bacterial assemblages in surface sediments across the San Pedro Basin, Southern California Borderlands. Environ Microbiol 9: 923–933.
Horner-Devine MC, Lage M, Hughes JB, Bohannan BJM . (2004). A taxa–area relationship for bacteria. Nature 432: 750–1753.
Hughes Martiny JB, Bohannan BJM, Brown JH, Colwell RK, Fuhrman JA, Green JL et al. (2006). Microbial biogeography: putting microorganisms on the map. Nat Rev Microbiol 4: 102–112.
Johnson DS, Fleeger JW . (2009). Weak response of saltmarsh infauna to ecosystem-wide nutrient enrichment and fish predator removal: A four year study. J Exp Mar Biol Ecol 373: 35–44.
Kan J, Crump BC, Wang K, Chen F . (2007). Bacterioplankton community in Chesapeake Bay: predictable or random assemblages. Limnol Oceanogr 51: 2157–2169.
Kent AD, Yannarell AC, Rusak JA, Triplett EW, McMahon KD . (2007). Synchrony in aquatic microbial community dynamics. ISME J 1: 38–47.
Legendre P, Legendre L . (1988). Numerical Ecology. Elsevier: Amsterdam, The Netherlands.
Lorenzen GL . (1967). Determination of chlorophyll and phaeo-pigments; spectrophotometric equations. Limnol Oceanogr 12: 343–346.
Magalhaes CM, Joye SB, Moreira RM, Wiebe WJ, Bordelo AA . (2005). Effect of salinity and inorganic nitrogen concentrations on nitrification and denitrification rates in intertidal sediments and rocky biofilms of the Douro River estuary, Portugal. Wat Res 39: 1783–1794.
Papke RT, Ramsing NB, Bateson MM, Ward DM . (2003). Geographical isolation in hot spring cyanobacteria. Trends Ecol Evol 8: 86–90.
Ramette A . (2007). Multivariate analyses in microbial ecology. FEMS Microbiol Ecol 62: 142–160.
Ramette A, Tiedje JM . (2007). Multiscale responses of microbial life to spatial distance and environmental heterogeneity in a patchy ecosystem. Proc Natl Acad Sci USA 104: 2761–2766.
Rees GN, Baldwin DS, Watson GO, Perryman S, Nielsen DL . (2004). Ordination and significance testing of microbial community composition derived from terminal restriction fragment length polymorphisms: application of multivariate statistics. Antonie van Leeuwenhoek 86: 339–347.
Sarda R, Valiela I, Foreman K . (1996). Decadal shifts in a salt marsh macroinfaunal community in response to long-term experimental nutrient enrichment. J Exp Mar Biol Ecol 205: 63–81.
Stepanauskas R, Moran MA, Bergamaschi BA, Hollibaugh JT . (2003). Covariance of bacterioplankton composition and environmental variables in a temperate delta system. Aquat Microb Ecol 31: 85–98.
Sullivan MJ, Currin CA . (2000). Community structure and functional dynamics of benthic microalgae in salt marshes. In: Weinstein MP, Kreeger DA (eds). Concepts and Controversies in Tidal Marsh Ecology. Kluwer Academic Publishers: Dordrecht, The Netherlands, pp 81–106.
Urakawa H, Maki H, Kawabata S, Fujimara T, Ando H, Kawai T et al. (2006). Abundance and population structure of ammonia oxidizing bacteria that inhabit canal sediments receiving effluent from municipal wastewater treatment plants. Appl Environ Microbiol 72: 6845–6850.
Valiela I, Howes B, Howarth R, Gibin A, Foreman K, Teal JM et al. (1982). Regulation of primary production in a salt marsh ecosystem. In: Gopal B, Turner RE, Wetzel RG, Whigham DF (eds). Wetlands: Ecology and Management. International Scientific Publications: Jaipur, India, pp 152–168.
Valiela I, Teal J . (1974). Nutrient limitation in salt marsh vegetation. In: Reimold RJ, Queen WH (eds). Ecology of Halophytes. Academic Press: New York, USA, pp. 547–563.
Valiela I, Teal JM, Allen SD, Van Etten R, Goehringer D, Volkmann S . (1985). Decomposition in salt marsh ecosystems: the phases and major factors affecting disappearance of above-ground organic matter. J Exp Mar Biol Ecol 89: 29–54.
Van Raalte CD, Valiela I, Carpenter EJ, Teal JM . (1974). Inhibition of nitrogen fixation in salt marshes measured by acetylene reduction. Estuar Coast Mar Sci 2: 301–305.
Van Raalte CD, Valiela I, Teal JM . (1976). Production of epibenthic algae: light and nutrient limitation. Limnol Oceanogr 21: 862–872.
Woodcock S, Curtis TP, Head IM, Lunn KM, Sloan WT . (2006). Taxa–area relationships for microbes: the unsampled and the unseen. Ecol Lett 9: 805–812.
Acknowledgements
Funding for this research came from a National Science Foundation Postdoctoral Fellowship in Microbial Biology to JLB. Funding for the marsh fertilization experiment (TIDE) came from The National Science Foundation (grants DEB 0213767, OCE 9726921 and OCE 0423565). We thank J Michael Johnson, Christian Picard, Deanne Drake, Kari A Galván, D Samuel Johnson, Lynsey E Lemay and Erin Miller for maintaining the enrichment experiment and for field assistance. Two anonymous reviewers greatly improved the quality of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bowen, J., Crump, B., Deegan, L. et al. Salt marsh sediment bacteria: their distribution and response to external nutrient inputs. ISME J 3, 924–934 (2009). https://doi.org/10.1038/ismej.2009.44
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ismej.2009.44
Keywords
This article is cited by
-
An analysis of Great Salt Lake Winogradsky columns
Journal of Oceanology and Limnology (2023)
-
Geochemical properties of blue carbon sediments through an elevation gradient: study of an anthropogenically impacted coastal lagoon
Biogeochemistry (2023)
-
Biogeochemical properties of blue carbon sediments influence the distribution and monomer composition of bacterial polyhydroxyalkanoates (PHA)
Biogeochemistry (2023)
-
Vegetation Zonation Predicts Soil Carbon Mineralization and Microbial Communities in Southern New England Salt Marshes
Estuaries and Coasts (2022)
-
Biogeography and Diversity of Freshwater Bacteria on a River Catchment Scale
Microbial Ecology (2019)