Abstract
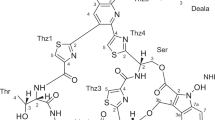
Thiazomycin is a novel thiazolyl peptide closely related to nocathiacin I. It was isolated from Amycolatopsis fastidiosa by chemical and biological screening. Thiazomycin showed highly potent bactericidal activity against Gram-positive pathogens (MIC range 0.002~0.064 μg/ml) and did not show cross-resistance to clinically relevant antibiotic classes such as β-lactams, vancomycin, oxazolidinone and quinolones. It was highly efficacious against Staphylococcus aureus infection in mice exhibiting an ED99 value of 0.15 mg/kg by subcutaneous administration. It inhibited bacterial growth by selective inhibition of protein synthesis and it was thought to interact with L11 protein and 23S rRNA of the 50S ribosome. Structurally, it possesses an oxazolidine ring in the amino-sugar residue that provides further opportunities for selective chemical modifications that are not feasible with other thiazolyl peptides. More importantly such a modification can potentially lead to semi-synthetic compounds that overcome problems that have hampered clinical development of this class of compounds. Despite its positive attributes, emergence of an unacceptable frequency of resistance poses significant challenges for further development of thiazomycin and this class of molecules for therapeutic use.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Walsh CT . Antibiotics: Actions, Origin, Resistance. ASM Press, Washington DC, ( 2003)
Singh SB, Barrett JF . Empirical antibacterial drug discovery—foundation in natural products. Biochem Pharmacol 71: 1006–1015 ( 2006)
Singh SB, Jayasuriya H, Ondeyka JG, Herath KB, Zhang C, Zink DL, Tsou NN, Ball RG, Basilio A, Genilloud O, Diez MT, Vicente F, Pelaez F, Young K, Wang J . Isolation, structure, and absolute stereochemistry of platensimycin, a broad spectrum antibiotic discovered using an antisense differential sensitivity strategy. J Am Chem Soc 128: 11916–11920 and 15547 ( 2006)
Wang J, Soisson SM, Young K, Shoop W, Kodali S, Galgoci A, Painter R, Parthasarathy G, Tang YS, Cummings R, Ha S, Dorso K, Motyl M, Jayasuriya H, Ondeyka J, Herath K, Zhang C, Hernandez L, Allocco J, Basilio A, Tormo JR, Genilloud O, Vicente F, Pelaez F, Colwell L, Lee SH, Michael B, Felcetto T, Gill C, Silver LL, Hermes JD, Bartizal K, Barrett J, Schmatz D, Becker JW, Cully D, Singh SB . Platensimycin is a selective FabF inhibitor with potent antibiotic properties. Nature 441: 358–361 ( 2006)
Wang J, Kodali S, Lee SH, Galgoci A, Painter R, Dorso K, Racine F, Motyl M, Hernandez L, Tinney E, Colletti S, Herath K, Cummings R, Salazar O, Gonzalez I, Basilio A, Vicente F, Genilloud O, Pelaez F, Jayasuriya H, Young K, Cully D, Singh SB . Platencin is a dual fabf and fabh inhibitor with potent in vivo antibiotic properties. Proc Natl Acad Sci USA 104: 7612–7616 ( 2007)
Jayasuriya H, Herath KB, Zhang C, Zink DL, Basilio A, Genilloud O, Diez MT, Vicente F, Gonzalez I, Salazar O, Pelaez F, Cummings R, Ha S, Wang J, Singh SB . Isolation and structure of platencin: A novel FabH and FabF dual inhibitor with potent broad spectrum antibiotic activity produced by Streptomyces platensis MA7339. Angew Chem Int Ed 46: 4684–4688 ( 2007)
Bagley MC, Dale JW, Merritt EA, Xiong X . Thiopeptide antibiotics. Chem Rev 105: 685–714 ( 2005)
Jayasuriya H, Herath K, Ondeyka J, Zhang C, Zink D, Brower M, Gailliot FP, Liesch G, Ushio M, Burgess B, Russotti G, Walker A, Hesse M, Seeley A, Junker B, Connors N, Masurekar P, Barrett JF, Singh SB . Isolation and structure elucidation of thiazomycin—A potent thiazolyl peptide antibiotic from Amycolatopsis fastidiosa. J. Antibiot 60: 554–564 ( 2007)
Connolly TP, Regueiro-Ren A, Leet JE, Springer DM, Goodrich J, Huang XS, Pucci MJ, Clark JM, Bronson JJ, Ueda Y . Chemical conversion of nocathiacin I to nocathiacin II and a lactone analogue of glycothiohexide alpha. J Nat Prod 68: 550–553 ( 2005)
Constantine KL, Mueller L, Huang S, Abid S, Lam KS, Li W, Leet JE . Conformation and absolute configuration of nocathiacin I determined by NMR spectroscopy and chiral capillary electrophoresis. J Am Chem Soc 124: 7284–7285 ( 2002)
Hrnciar P, Ueda Y, Huang S, Leet JE, Bronson JJ . Synthesis of novel nocathiacin-class antibiotics. Condensation of glycolaldehyde with primary amides and tandem reductive amination of amadori-rearranged 2-oxoethyl intermediates. J Org Chem 67: 8789–8793 ( 2002)
Li W, Leet JE, Ax HA, Gustavson DR, Brown DM, Turner L, Brown K, Clark J, Yang H, Fung-Tomc J, Lam KS . Nocathiacins, new thiazolyl peptide antibiotics from Nocardia sp. I. Taxonomy, fermentation and biological activities. J Antibiot 56: 226–231 ( 2003)
Naidu BN, Sorenson ME, Zhang Y, Kim OK, Matiskella JD, Wichtowski JA, Connolly TP, Li W, Lam KS, Bronson JJ, Pucci MJ, Clark JM, Ueda Y . Nocathiacin I analogues: synthesis, in vitro and in vivo biological activity of novel semi-synthetic thiazolyl peptide antibiotics. Bioorg Med Chem Lett 14: 5573–5577 ( 2004)
Naidu BN, Sorenson ME, Hudyma T, Zheng X, Zhang Y, Bronson JJ, Pucci MJ, Clark JM, Ueda Y . Synthesis and antibacterial activity of O-substituted nocathiacin I derivatives. Bioorg Med Chem Lett 14: 3743–3746 ( 2004)
Naidu BN, Sorenson ME, Bronson JJ, Pucci MJ, Clark JM, Ueda Y . Synthesis, in vitro, and in vivo antibacterial activity of nocathiacin I thiol-Michael adducts. Bioorg Med Chem Lett 15: 2069–2072 ( 2005)
Naidu BN, Sorenson ME, Matiskella JD, Li W, Sausker JB, Zhang Y, Connolly TP, Lam KS, Bronson JJ, Pucci MJ, Yang H, Ueda Y . Synthesis and antibacterial activity of nocathiacin I analogues. Bioorg Med Chem Lett 16: 3545–3549 ( 2006)
Pucci MJ, Bronson JJ, Barrett JF, DenBleyker KL, Discotto LF, Fung-Tomc JC, Ueda Y . Antimicrobial evaluation of nocathiacins, a thiazole peptide class of antibiotics. Antimicrob Agents Chemother 48: 3697–3701 ( 2004)
Regueiro-Ren A, Ueda Y . Mild method for cleavage of dehydroalanine units: highly efficient conversion of nocathiacin I to nocathiacin IV. J Org Chem 67: 8699–8702 ( 2002)
Regueiro-Ren A, Naidu BN, Zheng X, Hudyma TW, Connolly TP, Matiskella JD, Zhang Y, Kim OK, Sorenson ME, Pucci M, Clark J, Bronson JJ, Ueda Y . Novel semi-synthetic nocathiacin antibiotics: synthesis and antibacterial activity of bis- and mono-O-alkylated derivatives. Bioorg Med Chem Lett 14: 171–175 ( 2004)
Onishi HR, Pelak BA, Gerckens LS, Silver LL, Kahan FM, Chen MH, Patchett AA, Galloway SM, Hyland SA, Anderson MS, Raetz CR . Antibacterial agents that inhibit lipid A biosynthesis. Science 274: 980–982 ( 1996)
Wang J, Galgoci A, Kodali S, Herath KB, Jayasuriya H, Dorso K, Vicente F, Gonzalez A, Cully D, Bramhill D, Singh S . Discovery of a small molecule that inhibits cell division by blocking FtsZ, a novel therapeutic target of antibiotics. J Biol Chem 278: 44424–44428 ( 2003)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, S., Occi, J., Jayasuriya, H. et al. Antibacterial Evaluations of Thiazomycin. J Antibiot 60, 565–571 (2007). https://doi.org/10.1038/ja.2007.71
Received:
Accepted:
Issue date:
DOI: https://doi.org/10.1038/ja.2007.71
Keywords
This article is cited by
-
Antibacterial Alternatives to a Dying Antibiotic Pipeline
Current Treatment Options in Infectious Diseases (2023)
-
Natural thiopeptides as a privileged scaffold for drug discovery and therapeutic development
Medicinal Chemistry Research (2019)
-
Thiazomycin, nocathiacin and analogs show strong activity against clinical strains of drug-resistant Mycobacterium tuberculosis
The Journal of Antibiotics (2017)
-
Occurrence, distribution, dereplication and efficient discovery of thiazolyl peptides by sensitive-resistant pair screening
The Journal of Antibiotics (2013)
-
Antibiotic discovery in the twenty-first century: current trends and future perspectives
The Journal of Antibiotics (2010)