Abstract
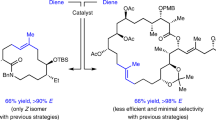
An alternative and improved total synthesis of incednam, the aglycon of the 24-membered macrolactam glycoside antibiotic incednine, was accomplished. The synthesis was realized via construction of the 24-membered macrocycle using intramolecular ring-closing olefin metathesis reaction as a key step.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Futamura, Y. et al. Discovery of incednine as a potent modulator of the anti-apoptotic function of Bcl-xL from microbial origin. J. Am. Chem. Soc. 130, 1822–1823 (2008).
Tsujimoto, Y., Finger, L. R., Yunis, J., Nowell, P. C. & Croce, C. M. Cloning of the chromosome breakpoint of neoplastic B cells with the t(14;18) chromosome translocation. Science 226, 1097–1099 (1984).
Reed, J. C., Cuddy, M., Slabiak, T., Croce, C. M. & Nowell, P. C. Oncogenic potential of bcl-2 demonstrated by gene transfer. Nature 336, 259–261 (1988).
Gross, A., McDonnell, J. M. & Korsmeyer, S. J. BCL-2 family members and the mitochondria in apoptosis. Genes Dev. 13, 1899–1911 (1999).
Ohtani, T. et al. Total synthesis of incednam, the aglycon of incednine. Org. Lett. 12, 5068–5071 (2010).
Grubbs, R. H. Olefin metathesis. Tetrahedron 60, 7117–7140 (2004).
Gradillas, A. & Pérez-Castells, J. Macrocyclization by ring-closing metathesis in the total synthesis of natural products: reaction conditions and limitations. Angew. Chem. Int. Ed. 45, 6086–6101 (2006).
Nicolaou, K. C., Bulger, P. G. & Sarlah, D. Metathesis reactions in total synthesis. Angew. Chem. Int. Ed. 44, 4490–4527 (2005).
Betzer, J. -F., Delaloge, F., Muller, B., Pancrazi, A. & Prunet, J. Radical hydrostannylation, Pd(0)-catalyzed hydrostannylation, stannylcupration of propargyl alcohols and enynols: regio- and stereoselectivities. J. Org. Chem. 62, 7768–7780 (1997).
Michels, T. D., Rhee, J. U. & Vanderwal, C. D. Synthesis of δ-tributylstannyl-α,β,γ,δ-unsaturated aldehydes from pyridines. Org. Lett. 10, 4787–4790 (2008).
Staudinger, H. & Meyer, J. New organic compounds of phosphorus. III. Phosphinemethylene derivatives and phosphinimines. Helv. Chim. Acta 2, 635–646 (1919).
Stuckwisch, C. G. Azomethine ylides, azomethine imines, and iminophosphoranes in organic syntheses. Synthesis 469–483 (1973).
Han, X., Stoltz, B. M. & Corey, E. J. Cuprous chloride accelerated Stille reactions. A general and effective coupling system for sterically congested substrates and for enantioselective synthesis. J. Am. Chem. Soc. 121, 7600–7605 (1999).
Schwab, P., France, M. B., Ziller, J. W. & Grubbs, R. H. A series of well-defined metathesis catalysts–synthesis of [RuCl2(:CHR’)(PR3)2] and its reactions. Angew. Chem. Int. Ed. 34, 2039–2041 (1995).
Scholl, M., Trnka, T. M., Morgan, J. P. & Grubbs, R. H. Increased ring closing metathesis activity of ruthenium-based olefin metathesis catalysts coordinated with imidazolin-2-ylidene ligands. Tetrahedron Lett. 40, 2247–2250 (1999).
Kingsbury, J. S., Harrity, J. P. A., Bonitatebus, P. J. Jr & Hoveyda, A. H. A recyclable Ru-based metathesis catalyst. J. Am. Chem. Soc 121, 791–799 (1999).
Garber, S. B., Kingsbury, J. S., Gray, B. L. & Hoveyda, A. H. Efficient and recyclable monomeric and dendritic Ru-based metathesis catalysts. J. Am. Chem. Soc. 122, 8168–8179 (2000).
Grela, K., Harutyunyan, S. & Michrowska, A. A highly efficient ruthenium catalyst for metathesis reactions. Angew. Chem. Int. Ed. 41, 4038–4040 (2002).
Bieniek, M., Michrowska, A., Gułajski, Ł. & Grela, K. A practical larger scale preparation of second-generation Hoveyda-type catalysts. Organometallics 26, 1096–1099 (2007).
Ohtani, T., Sakai, S., Takada, A., Takahashi, D. & Toshima, K. Efficient and stereoselective synthesis of the disaccharide fragment of incednine. Org. Lett. 13, 6126–6129 (2011).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takada, A., Uda, K., Ohtani, T. et al. Improved total synthesis of incednam. J Antibiot 66, 155–159 (2013). https://doi.org/10.1038/ja.2013.4
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ja.2013.4