Abstract
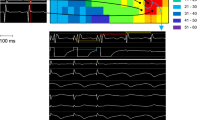
Bradycardia is a trigger of ventricular arrhythmias in patients with arrhythmia including Brugada syndrome and long QT syndrome. The HCN4 channel controls the heart rate, and its mutations predispose to inherited sick sinus syndrome and long QT syndrome associated with bradycardia. We found a 4 base-insertion at the splice donor site of the HCN4 gene in a patient with idiopathic ventricular tachycardia, which was supposed to generate a truncated channel. To investigate the role of the HCN4 channel in ventricular arrhythmia, we introduced a ventricular action potential of If channel produced by HCN4 in a computer simulation model and found that the If channel generated a leaky outward current during the plateau phase of ventricular action potential. Currents through the If channel were suggested to contribute to the shortening of the action potential duration and the prevention of early after-depolarization in bradycardia. These observations suggested that the HCN4 channel played a preventive role in triggering bradycardia-induced ventricular arrhythmias.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Accession codes
References
Ueda, K., Nakamura, K., Hayashi, T., Inagaki, N., Takahashi, M., Arimura, T. et al. Functional characterization of a trafficking-defective HCN4 mutation, D553N, associated with cardiac arrhythmia. J. Biol. Chem. 279, 27194–27198 (2004).
Schulze-Bahr, E., Neu, A., Friederich, P., Kaupp, UB., Breithardt, G., Pongs, O. et al. Pacemaker channel dysfunction in a patient with sinus node disease. J. Clin. Invest. 111, 1537–1545 (2003).
Wang, Q., Curran, M. E., Splawski, I., Burn, T. C., Millholland, J. M., VanRaay, T. J. et al. Positional cloning of a novel potassium channel gene: KVLQT1 mutations cause cardiac arrhythmias. Nat. Genet. 12, 17–23 (1996).
Curran, M. E., Splawski, I., Timothy, K. W., Vincent, G. M., Green, E. D. & Keating, M. T. A molecular basis for cardiac arrhythmia: HERG mutations cause long QT syndrome. Cell 80, 795–803 (1995).
Wang, Q., Shen, J., Splawski, I., Atkinson, D., Li, Z., Robinson, J. L. et al. SCN5A mutations associated with an inherited cardiac arrhythmia, long QT syndrome. Cell 80, 805–811 (1995).
Splawski, I., Tristani-Firouzi, M., Lehmann, M. H., Sanguinetti, M. C. & Keating, M. T. Mutations in the hminK gene cause long QT syndrome and suppress IKs function. Nat. Genet. 17, 338–340 (1996).
Abbott, G. W., Sesti, F., Splawski, I., Buck, M. E., Lehmann, M. H., Timothy, K. W. et al. MiRP1 forms IKr potassium channels with HERG and is associated with cardiac arrhythmia. Cell 97, 175–187 (1997).
Tristani-Firouzi, M., Jensen, J. L., Donaldson, M. R., Sansone, V., Meola, G., Hahn, A. et al. Functional and clinical characterization of KCNJ2 mutations associated with LQT7 (Andersen syndrome). J. Clin. Invest. 110, 381–388 (2002).
Splawski, I., Timothy, K. W., Sharpe, L. M., Decher, N., Kumar, P., Bloise, R. et al. Ca(V)1.2 calcium channel dysfunction causes a multisystem disorder including arrhythmia and autism. Cell 119, 19–31 (2004).
Brugada, R., Hong, K., Dumaine, R., Cordeiro, J., Gaita, F., Borggrefe, M. et al. Sudden death associated with short-QT syndrome linked to mutations in HERG. Circulation 109, 30–35 (2004).
Bellocq, C., van Ginneken, A. C., Bezzina, C. R., Alders, M., Escande, D., Mannens, M. M. et al. Mutation in the KCNQ1 gene leading to the short QT-interval syndrome. Circulation 109, 2394–2397 (2004).
Chen, Y. H., Xu, S. J., Bendahhou, S., Wang, X. L., Wang, Y., Xu, W. Y. et al. KCNQ1 gain-of-function mutation in familial atrial fibrillation. Science 299, 251–254 (2003).
Yang, Y., Xia, M., Jin, Q., Bendahhou, S., Shi, J., Chen, Y. et al. Identification of a KCNE2 gain-of-function mutation in patients with familial atrial fibrillation. Am. J. Hum. Genet. 75, 899–905 (2004).
Chen, Q., Kirsch, G. E., Zhang, D., Brugada, R., Brugada, J., Brugada, P. et al. Genetic basis and molecular mechanism for idiopathic ventricular fibrillation. Nature 392, 293–296 (1998).
Schott, J. J., Alshinawi, C., Kyndt, F., Probst, V., Hoorntje, T. M., Hulsbeek, M. et al. Cardiac conduction defects associate with mutations in SCN5A. Nat. Genet. 23, 20–21 (1999).
Priori, S. G., Napolitano, C., Tiso, N., Memmi, M., Vignati, G., Bloise, R. et al. Mutations in the cardiac ryanodine receptor gene (hRyR2) underlie catecholaminergic polymorphic ventricular tachycardia. Circulation 103, 196–200 (2001).
Marban, E. Cardiac channelopathies. Nature 415, 213–218 (2002).
Moosmang, S., Stieber, J., Zong, X., Biel, M., Hofmann, F. & Ludwig, A. Cellular expression and functional characterization of four hyperpolarization-activated pacemaker channels in cardiac and neuronal tissues. Eur. J. Biochem. 268, 1646–1652 (2001).
Westenskow, P., Splawski, I., Timothy, K. W., Keating, M. T. & Sanguinetti, M. C. Compound mutations: a common cause of severe long-QT syndrome. Circulation 109, 1834–1841 (2004).
Nishi, H., Kimura, A., Harada, H., Toshima, H. & Sasazuki, T. Novel missense mutation in cardiac beta myosin heavy chain gene found in a Japanese patient with hypertrophic cardiomyopathy. Biochem. Biophys. Res. Commun. 188, 379–387 (1992).
Wang, Q., Li, Z., Shen, J. & Keating, M. T. Genomic organization of the human SCN5A gene encoding the cardiac sodium channel. Genomics 34, 9–16 (1996).
Splawski, I., Shen, J., Timothy, K. W., Vincent, G. M., Lehmann, M. H. & Keating, M. T. Genomic structure of three long QT syndrome genes: KVLQT1, HERG, and KCNE1. Genomics 51, 86–97 (1998).
Plaster, N. M., Tawil, R., Tristani-Firouzi, M., Canun, S., Bendahhou, S., Tsunoda, A. et al. Mutations in Kir2.1 cause the developmental and episodic electrical phenotypes of Andersen's syndrome. Cell 105, 511–519 (2001).
Chen, J., Mitcheson, J. S., Tristani-Firouzi, M., Lin, M. & Sanguinetti, M. C. The S4-S5 linker couples voltage sensing and activation of pacemaker channels. Proc. Natl. Acad. Sci. 98, 11277–11282 (2001).
Priebe, L. & Beuckelmann, D. J. Simulation study of cellular electric properties in heart failure. Circ. Res. 82, 1206–1223 (1998).
ten Tusscher, K. H., Noble, D., Noble, P. J. & Panfilov, A. V. A model for human ventricular tissue. Am. J. Physiol. Heart Circ. Physiol. 286, H1573–H1589 (2004).
Luo, C. H. & Rudy, Y. A dynamic model of the cardiac ventricular action potential: I. Simulations of ionic currents and concentration changes. Circ. Res. 74, 1071–1096 (1994).
Cerbai, E., Sartiani, L., de Paoli, P., Pino, R., Maccherini, M., Bizzarri, F. et al. The properties of the pacemaker current I(F)in human ventricular myocytes are modulated by cardiac disease. J. Mol. Cell. Cardiol. 33, 441–448 (2001).
Kaupp, U. B. & Seifert, R. Molecular diversity of pacemaker ion channels. Annu. Rev. Physiol. 63, 235–257 (2001).
Brugada, P., Geelen, P., Brugada, R., Mont, L. & Brugada, J. Prognostic value of electrophysiologic investigations in Brugada syndrome. J. Cardiovasc. Electrophysiol. 12, 1004–1007 (2001).
Acknowledgements
We thank Drs J Yamada, T Miura, M Kanamoto, T Tamura, N Miya, Y Horie, H Sakurada, S Ogawa, M Nishizaki, T Sakamoto, T Ueyama, K Hirao and J Nitta for their contributions in collecting blood samples and clinical records from the patients with BrS and LQTS. This study was supported in part by Grant-in-Aids from the Ministry of Education, Culture, Sports, Science and Technology, Japan and a research grant from the Ministry of Health, Labour and Welfare, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ueda, K., Hirano, Y., Higashiuesato, Y. et al. Role of HCN4 channel in preventing ventricular arrhythmia. J Hum Genet 54, 115–121 (2009). https://doi.org/10.1038/jhg.2008.16
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/jhg.2008.16
Keywords
This article is cited by
-
Atrial involvement and progression of sinus node dysfunction in non-senile patients: evidences from electroanatomic mapping and long-term follow-up
Journal of Interventional Cardiac Electrophysiology (2021)
-
Characterization of drug binding within the HCN1 channel pore
Scientific Reports (2019)
-
Disease-linked mutations alter the stoichiometries of HCN-KCNE2 complexes
Scientific Reports (2019)
-
Pathogenesis and management of Brugada syndrome
Nature Reviews Cardiology (2016)
-
Genetics of Brugada syndrome
Journal of Human Genetics (2016)