Abstract
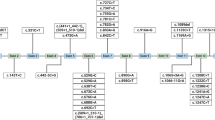
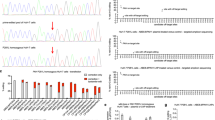
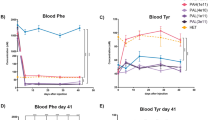
The aims of our research were to define the genotype–phenotype correlations of mutations in the phenylalanine hydroxylase (PAH) gene that cause phenylketonuria (PKU) among the Israeli population. The mutation spectrum of the PAH gene in PKU patients in Israel is described, along with a discussion on genotype–phenotype correlations. By using polymerase chain reaction/denaturing high-performance liquid chromatography (PCR/dHPLC) and DNA sequencing, we screened all exons of the PAH gene in 180 unrelated patients with four different PKU phenotypes [classic PKU, moderate PKU, mild PKU, and mild hyperphenylalaninemia (MHP)]. In 63.2% of patient genotypes, the metabolic phenotype could be predicted, though evidence is also found for both phenotypic inconsistencies among subjects with more than one type of mutation in the PAH gene. Data analysis revealed that about 25% of patients could participate in the future in (6R)-l-erythro-5, 6, 7, 8-tetrahydrobiopterin (BH4) treatment trials according to their mutation genotypes. This study enables us to construct a national database in Israel that will serve as a valuable tool for genetic counseling and a prognostic evaluation of future cases of PKU.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Avigad S, Cohen BE, Bauer S, Schwartz G, Frydman M, Woo SL, Niny Y, Shiloh Y (1990) A single origin of phenylketonuria in Yemenite Jews. Nature 344:168–170
Avigad S, Kleiman S, Weinstein M, Cohen BE, Schwartz G, Woo SL, Shiloh Y (1991) Compound heterozygosity in nonphenylketonuria hyperphenylalanemia: the contribution of mutations for classical phenylketonuria. Am J Hum Genet 49:393–399
Blau N (2003) BIOPKU: international database of patients and mutations causing tetrahydrobiopterin-responsive. HPA/PKU. http://www.bh4.org
Blau N, Erlandsen H (2004) The metabolic and molecular bases of tetrahydrobiopterin-responsive phenylalanine hydroxylase deficiency. Mol Genet Metab 82:101–111
Bercovich D, Beaudet AL (2003) Denaturing high-performance liquid chromatography for the detection of mutations and polymorphisms in UBE3A. Genet Test 7:189–194
Bercovich D, Elimelech A, Yardeni T, Korem S, Zlotogora J, Gal N, Goldstein N, Vilensky B, Segev R, Avraham S, Loewenthal R, Schwartz G, Anikster Y (2008) A mutation analysis of the phenylalanine hydroxylase (PAH) gene in the Israeli population. Ann Hum Genet AHG-07-0124 (in press)
DiLella AG, Kwok SC, Ledley FD, Marvit J, Woo SL (1986) Molecular structure and polymorphic map of the human phenylalanine hydroxylase gene. Biochemistry 25:743–749
Erlandsen H, Stevens RC (1999) The structural basis of phenylketonuria. Mol Genet Metab 68:103–125
Erlandsen H, Pey AL, Gamez A, Perez B, Desviat LR, Aguado C, Koch R, Surendran S, Tyring S, Matalon R, Scriver CR, Ugarte M, Martinez A, Stevens RC (2004) Correction of kinetic and stability defects by tetrahydrobiopterin in phenylketonuria patients with certain phenylalanine hydroxylase mutations. Proc Natl Acad Sci USA 101:16903–16908
Guldberg P, Henriksen KF, Guttler F (1993a) Molecular analysis of phenylketonuria in Denmark: 99% of the mutations detected by denaturing gradient gel electrophoresis. Genomics 17:141–146
Guldberg P, Romano V, Ceratto N, Bosco P, Ciuna M, Indelicato A, Mollica F, Meli C, Giovannini M, Riva E et al (1993b) Mutational spectrum of phenylalanine hydroxylase deficiency in Sicily: implications for diagnosis of hyperphenylalaninemia in southern Europe. Hum Mol Genet 2:1703–1707
Guldberg P, Rey F, Zschocke J, Romano V, Francois B, Michiels L, Ullrich K, Hoffmann GF, Burgard P, Schmidt H et al (1998) A European multicenter study of phenylalanine hydroxylase deficiency: classification of 105 mutations and a general system for genotype-based prediction of metabolic phenotype. Am J Hum Genet 63:71–79
Güttler F, Lou H (1990) Phenylketonuria and hyperphenylalaninemia. In: Fernandes J, Saudubray JM, Tada K (eds) Inborn metabolic disease: diagnosis and treatment. Springer, Berlin, pp 161–174
Kayaalp E, Treacy E, Waters PJ, Byck S, Nowacki P, Scriver CR (1997) Human phenylalanine hydroxylase mutations and hyperphenylalaninemia phenotypes: a meta-analysis of genotype–phenotype correlations. Am J Hum Genet 61:1309–1317
Kleiman S, Bernstein J, Schwartz G, Eisensmith RC, Woo SL, Shiloh Y (1992) A defective splice site at the phenylalanine hydroxylase gene in phenylketonuria and benign hyperphenylalaninemia among Palestinian Arabs. Hum Mutat 1:340–343
Kleiman S, Avigad S, Vanagaite L, Shmuelevitz A, David M, Eisensmith RC, Brand N, Schwartz G, Rey F, Munnich A et al (1994) Origins of hyperphenylalaninemia in Israel. Eur J Hum Genet 2:24–34
Kozak L, Blazkova M, Kuhrova V, Pijackova A, Ruzickova S, St’astna S (1997) Mutation and haplotype analysis of phenylalanine hydroxylase alleles in classical PKU patients from the Czech Republic: identification of four novel mutations. J Med Genet 34:893–898
Leuzzi V, Carducci C, Carducci C, Chiarotti F, Artiola C, Giovanniello A, Antonozzi I (2006) The spectrum of phenylalanine variations under tetrahydrobiopterin load in subjects affected by phenylalanine hydroxylase deficiency. J Inherit Metab Dis 29:38–46
Mallolas J, Vilaseca MA, Campistol J, Lambruschini N, Cambra FJ, Estivill X, Mila M (1999) Mutational spectrum of phenylalanine hydroxylase deficiency in the population resident in Catalonia: genotype–phenotype correlation. Hum Genet 105:468–473
O’Donnell KA, O’Neill C, Tighe O, Bertorelle G, Naughten E, Mayne PD, Croke DT (2002) The mutation spectrum of hyperphenylalaninaemia in the Republic of Ireland: the population history of the Irish revisited. Eur J Hum Genet 10:530–538
Okano Y, Eisensmith RC, Guttler F, Lichter-Konecki U, Konecki DS, Trefz FK, Dasovich M, Wang T, Henriksen K, Lou H et al (1991) Molecular basis of phenotypic heterogeneity in phenylketonuria. N Engl J Med 324:1232–1238
Scriver CR, Eisensmith RC, Woo SLC (1995) The hyperphenylalaninemias. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The metabolic and molecular basis of inherited disease, vol 1. McGraw-Hill, New York, pp 1015–1077
Song F, Qu YJ, Zhang T, Jin YW, Wang H, Zheng XY (2005) Phenylketonuria mutations in Northern China. Mol Genet Metab 86(1):107–118
Shlush LI, Behar DM, Zelazny A, Keller N, Lupski JR, Beaudet AL, Bercovich D (2002) Molecular epidemiological analysis of the changing nature of a meningococcal outbreak following a vaccination campaign. J Clin Microbiol 40:3565–3571
Waters PJ (2003) How PAH gene mutations cause hyper-phenylalaninemia and why mechanism matters: insights from in vitro expression. Hum Mutat 21:357–369
Weinstein M, Eisensmith RC, Abadie V, Avigad S, Lyonnet S, Schwartz G, Munnich A, Woo SL, Shiloh Y (1993) A missense mutation, S349P, completely inactivates phenylalanine hydroxylase in north African Jews with phenylketonuria. Hum Genet 90:645–649
Zschocke J, Hoffmann GF (1999) Phenylketonuria mutations in Germany. Hum Genet 104:390–398
Zschocke J (2003) Phenylketonuria mutations in Europe. Hum Mutat 21:345–356
Acknowledgments
The study was supported in part by grants from The Elsa and Leo Abramson Fund and The Rivka Nacht Fund for Research in Neurology. This study was part of a master’s degree at Tel Aviv University (AE). We also thank the Israeli PKU Family Foundation for their support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bercovich, D., Elimelech, A., Zlotogora, J. et al. Genotype–phenotype correlations analysis of mutations in the phenylalanine hydroxylase (PAH) gene. J Hum Genet 53, 407–418 (2008). https://doi.org/10.1007/s10038-008-0264-4
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1007/s10038-008-0264-4