Abstract
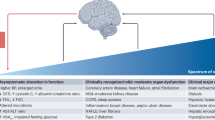
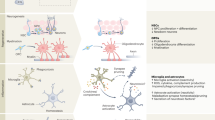
Although systemic diseases take the biggest toll on human health and well-being, increasingly, a failing brain is the arbiter of a death preceded by a gradual loss of the essence of being. Ageing, which is fundamental to neurodegeneration and dementia, affects every organ in the body and seems to be encoded partly in a blood-based signature. Indeed, factors in the circulation have been shown to modulate ageing and to rejuvenate numerous organs, including the brain. The discovery of such factors, the identification of their origins and a deeper understanding of their functions is ushering in a new era in ageing and dementia research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
United Nations Department of Economic and Social Affairs, Population Division. World Population Ageing 2015. Report ST/ESA/SER.A/390 http://www.un.org/en/development/desa/population/publications/pdf/ageing/WPA2015_Report.pdf (United Nations, 2015).
Elobeid, A., Libard, S., Leino, M., Popova, S. N. & Alafuzoff, I. Altered proteins in the aging brain. J. Neuropathol. Exp. Neurol. 75, 316–325 (2016). A comprehensive study and review of the literature describing the prevalence of protein aggregates in cognitively unimpaired aged brains.
Mrak, R. E., Griffin, S. T. & Graham, D. I. Aging-associated changes in human brain. J. Neuropathol. Exp. Neurol. 56, 1269–1275 (1997).
Brunk, U. T. & Terman, A. The mitochondrial-lysosomal axis theory of aging: accumulation of damaged mitochondria as a result of imperfect autophagocytosis. Eur. J. Biochem. 269, 1996–2002 (2002).
Safaiyan, S. et al. Age-related myelin degradation burdens the clearance function of microglia during aging. Nature Neurosci. 19, 995–998 (2016). An elegant demonstration of how age-related myelin breakdown results in the accumulation of microglial lipofuscin and cell dysfunction.
Nixon, R. A., Cataldo, A. M. & Mathews, P. M. The endosomal-lysosomal system of neurons in Alzheimer's disease pathogenesis: a review. Neurochem. Res. 25, 1161–1172 (2000).
Nixon, R. A. et al. Extensive involvement of autophagy in Alzheimer disease: an immuno-electron microscopy study. J. Neuropathol. Exp. Neurol. 64, 113–122 (2005).
Menzies, F. M., Fleming, A. & Rubinsztein, D. C. Compromised autophagy and neurodegenerative diseases. Nature Rev. Neurosci. 16, 345–357 (2015).
Pickford, F. et al. The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid β accumulation in mice. J. Clin. Invest. 118, 2190–2199 (2008).
Nilsson, P. et al. Aβ secretion and plaque formation depend on autophagy. Cell Rep. 5, 61–69 (2013).
Caccamo, A. et al. mTOR regulates tau phosphorylation and degradation: implications for Alzheimer's disease and other tauopathies. Aging Cell 12, 370–380 (2013).
Ash, P. E. A., Vanderweyde, T. E., Youmans, K. L., Apicco, D. J. & Wolozin, B. Pathological stress granules in Alzheimer's disease. Brain Res. 1584, 52–58 (2014).
Vanderweyde, T. et al. Contrasting pathology of the stress granule proteins TIA-1 and G3BP in tauopathies. J. Neurosci. 32, 8270–8283 (2012).
Kawas, C. H. et al. Multiple pathologies are common and related to dementia in the oldest-old: The 90+ Study. Neurology 85, 535–542 (2015).
Dubois, B. et al. Preclinical Alzheimer's disease: definition, natural history, and diagnostic criteria. Alzheimers Dement. 12, 292–323 (2016).
Jack, C. R. et al. Suspected non-Alzheimer disease pathophysiology — concept and controversy. Nature Rev. Neurol. 12, 117–124 (2016).
Landau, S. M., Horng, A., Fero, A. & Jagust, W. J. Amyloid negativity in patients with clinically diagnosed Alzheimer disease and MCI. Neurology 86, 1377–1385 (2016).
Small, S. A., Schobel, S. A., Buxton, R. B., Witter, M. P. & Barnes, C. A. A pathophysiological framework of hippocampal dysfunction in ageing and disease. Nature Rev. Neurosci. 12, 585–601 (2011).
Herskind, A. M. et al. The heritability of human longevity: a population-based study of 2872 Danish twin pairs born 1870–1900. Hum. Genet. 97, 319–323 (1996).
vB Hjelmborg, J. et al. Genetic influence on human lifespan and longevity. Hum. Genet. 119, 312–321 (2006).
Shadyab, A. H. & LaCroix, A. Z. Genetic factors associated with longevity: a review of recent findings. Ageing Res. Rev. 19, 1–7 (2015).
Pilling, L. C. et al. Human longevity is influenced by many genetic variants: evidence from 75,000 UK Biobank participants. Aging 8, 547–560 (2016).
Newman, A. B. & Murabito, J. M. The epidemiology of longevity and exceptional survival. Epidemiol. Rev. 35, 181–197 (2013).
Erikson, G. A. et al. Whole-genome sequencing of a healthy aging cohort. Cell 165, 1002–1011 (2016).
Matteini, A. M. et al. GWAS analysis of handgrip and lower body strength in older adults in the CHARGE consortium. Aging Cell 15, 792–800 (2016).
Putin, E. et al. Deep biomarkers of human aging: application of deep neural networks to biomarker development. Aging 8, 1021–1033 (2016).
Zeng, Y. et al. Novel loci and pathways significantly associated with longevity. Sci. Rep. 6, 21243 (2016).
Sebastiani, P. et al. Meta-analysis of genetic variants associated with human exceptional longevity. Aging 5, 653–661 (2013).
Podtelezhnikov, A. A. et al. Molecular insights into the pathogenesis of Alzheimer's disease and its relationship to normal aging. PLoS ONE 6, e29610 (2011). The first large-scale analysis of transcriptional brain networks in ageing people and those with AD, and the discovery of an accelerated ageing profile in AD.
Hernandez, D. G. et al. Distinct DNA methylation changes highly correlated with chronological age in the human brain. Hum. Mol. Genet. 20, 1164–1172 (2011).
Horvath, S. DNA methylation age of human tissues and cell types. Genome Biol. 14, R115 (2013); erratum 16, 96 (2015). Description of an 'epigenetic clock' that correlates with tissue ageing and shows acceleration in cancer.
Horvath, S. & Ritz, B. R. Increased epigenetic age and granulocyte counts in the blood of Parkinson's disease patients. Aging 7, 1130–1142 (2015).
Lunnon, K. et al. Methylomic profiling implicates cortical deregulation of ANK1 in Alzheimer's disease. Nature Neurosci. 17, 1164–1170 (2014).
De Jager, P. L. et al. Alzheimer's disease: early alterations in brain DNA methylation at ANK1, BIN1, RHBDF2 and other loci. Nature Neurosci. 17, 1156–1163 (2014). A large-scale genome-wide DNA methylation study of a neurodegenerative disease.
Lord, J. & Cruchaga, C. The epigenetic landscape of Alzheimer's disease. Nature Neurosci. 17, 1138–1140 (2014).
Gjoneska, E. et al. Conserved epigenomic signals in mice and humans reveal immune basis of Alzheimer's disease. Nature 518, 365–369 (2015).
Satoh, J.-I., Asahina, N., Kitano, S. & Kino, Y. A comprehensive profile of ChIP-Seq-based PU.1/Spi1 target genes in microglia. Gene Regul. Syst. Bio. 8, 127–139 (2014).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. The hallmarks of aging. Cell 153, 1194–1217 (2013).
Ray, S. et al. Classification and prediction of clinical Alzheimer's diagnosis based on plasma signaling proteins. Nature Med. 13, 1359–1362 (2007).
Menni, C. et al. Circulating proteomic signatures of chronological age. J. Gerontol. A Biol. 70, 809–816 (2015).
Baird, G. S. et al. Age-dependent changes in the cerebrospinal fluid proteome by slow off-rate modified aptamer array. Am. J. Pathol. 180, 446–456 (2012).
Hu, W. T. et al. Plasma multianalyte profiling in mild cognitive impairment and Alzheimer disease. Neurology 79, 897–905 (2012); erratum 79, 1935 (2012).
Johnstone, D., Milward, E. A., Berretta, R. & Moscato, P. Multivariate protein signatures of pre-clinical Alzheimer's disease in the Alzheimer's Disease Neuroimaging Initiative (ADNI) Plasma Proteome Dataset. PLoS ONE 7, e34341 (2012).
Hye, A. et al. Plasma proteins predict conversion to dementia from prodromal disease. Alzheimers Dement. 10, 799–807 (2014).
Soares, H. D. et al. Plasma biomarkers associated with the apolipoprotein E genotype and Alzheimer disease. Arch. Neurol. 69, 1310–1317 (2012).
Britschgi, M. et al. Modeling of pathological traits in Alzheimer's disease based on systemic extracellular signaling proteome. Mol. Cell. Proteomics 10, M111.008862 (2011).
Kiddle, S. J. et al. Plasma based markers of [11C] PiB-PET brain amyloid burden. PLoS ONE 7, e44260 (2012).
Sattlecker, M. et al. Alzheimer's disease biomarker discovery using SOMAscan multiplexed protein technology. Alzheimers Dement. 10, 724–734 (2014).
Jaeger, P. A. et al. Network-driven plasma proteomics expose molecular changes in the Alzheimer's brain. Mol. Neurodegener. 11, 31 (2016); erratum 11, 42 (2016).
Villeda, S. A. et al. The ageing systemic milieu negatively regulates neurogenesis and cognitive function. Nature 477, 90–94 (2011). The first demonstration of the effects of circulatory blood factors on brain ageing and cognitive function and the first systematic treatment of ageing with plasma injections.
Baruch, K. et al. CNS-specific immunity at the choroid plexus shifts toward destructive Th2 inflammation in brain aging. Proc. Natl Acad. of Sci. USA 110, 2264–2269 (2013).
Vasudevan, A. R. Eotaxin and obesity. J. Clin. Endocrinol. Metab. 91, 256–261 (2006).
Choi, K. M. et al. Effect of exercise training on plasma visfatin and eotaxin levels. Eur. J. Endocrinol. 157, 437–442 (2007).
Smith, L. K. et al. β2-microglobulin is a systemic pro-aging factor that impairs cognitive function and neurogenesis. Nature Med. 21, 932–937 (2015).
Shatz, C. J. MHC class I: an unexpected role in neuronal plasticity. Neuron 64, 40–45 (2009).
Franceschi, C. & Campisi, J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J. Gerontol. A Biol. 69, S4–S9 (2014).
Coppé, J.-P. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 6, 2853–68 (2008).
Salminen, A. et al. Astrocytes in the aging brain express characteristics of senescence-associated secretory phenotype. Eur. J. Neurosci. 34, 3–11 (2011).
Grabert, K. et al. Microglial brain region-dependent diversity and selective regional sensitivities to aging. Nature Neurosci. 19, 504–516 (2016).
Mosher, K. I. & Wyss-Coray, T. Microglial dysfunction in brain aging and Alzheimer's disease. Biochem. Pharmacol. 88, 594–604 (2014).
Lalli, M. A. et al. Whole-genome sequencing suggests a chemokine gene cluster that modifies age at onset in familial Alzheimer's disease. Mol. Psychiatry 20, 1294–1300 (2015).
Heneka, M. T. et al. Neuroinflammation in Alzheimer's disease. Lancet Neurol. 14, 388–405 (2015).
Lucin, K. M. & Wyss-Coray, T. Immune activation in brain aging and neurodegeneration: too much or too little? Neuron 64, 110–122 (2009).
McGeer, P. L., McGeer, E., Rogers, J. & Sibley, J. Anti-inflammatory drugs and Alzheimer disease. Lancet 335, 1037 (1990).
Côté, S. et al. Nonsteroidal anti-inflammatory drug use and the risk of cognitive impairment and Alzheimer's disease. Alzheimers Dement. 8, 219–226 (2012).
Vlad, S. C., Miller, D. R., Kowall, N. W. & Felson, D. T. Protective effects of NSAIDs on the development of Alzheimer disease. Neurology 70, 1672–1677 (2008).
Manthripragada, A. D. et al. Non-steroidal anti-inflammatory drug use and the risk of Parkinson's disease. Neuroepidemiology 36, 155–161 (2011).
Holmes, C., Cunningham, C., Zotova, E., Culliford, D. & Perry, V. H. Proinflammatory cytokines, sickness behavior, and Alzheimer disease. Neurology 77, 212–218 (2011).
Holmes, C. et al. Systemic inflammation and disease progression in Alzheimer disease. Neurology 73, 768–774 (2009).
Zhang, B. et al. Integrated systems approach identifies genetic nodes and networks in late-onset Alzheimer's disease. Cell 153, 707–720 (2013).
Linnartz, B. & Neumann, H. Microglial activatory (immunoreceptor tyrosine-based activation motif)- and inhibitory (immunoreceptor tyrosine-based inhibition motif)-signaling receptors for recognition of the neuronal glycocalyx. Glia 61, 37–46 (2013).
Jonsson, T. et al. Variant of TREM2 associated with the risk of Alzheimer's disease. N. Engl. J. Med. 368, 107–116 (2013).
Guerreiro, R. et al. TREM2 variants in Alzheimer's disease. N. Engl. J. Med. 368, 117–127 (2013).
Naj, A. C. et al. Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer's disease. Nature Genet. 43, 436–441 (2011).
Hollingworth, P. et al. Common variants at ABCA7, MS4A6A/MS4A4E, EPHA1, CD33 and CD2AP are associated with Alzheimer's disease. Nature Genet. 43, 429–435 (2011).
Lambert, J. C. et al. Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer's disease. Nature Genet. 45, 1452–1458 (2013).
Spencer, B. & Masliah, E. Immunotherapy for Alzheimer's disease: past, present and future. Front. Aging Neurosci. 6, 114 (2014).
Conboy, I. M. et al. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature 433, 760–764 (2005). This landmark study provides the first molecular evidence that heterochronic parabiosis can rejuvenate and reverse stem cell ageing in numerous tissues.
Ruckh, J. M. et al. Rejuvenation of regeneration in the aging central nervous system. Cell Stem Cell 10, 96–103 (2012).
Villeda, S. A. et al. Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice. Nature Med. 20, 659–663 (2014). The first report to show that systemic administration of plasma from young mice can reverse cognitive deficits in aged mice.
Katsimpardi, L. et al. Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors. Science 344, 630–634 (2014).
Salpeter, S. J. et al. Systemic regulation of the age-related decline of pancreatic β-cell replication. Diabetes 62, 2843–2848 (2013).
Loffredo, F. S. et al. Growth differentiation factor 11 is a circulating factor that reverses age-related cardiac hypertrophy. Cell 153, 828–839 (2013).
Baht, G. S. et al. Exposure to a youthful circulation rejuvenates bone repair through modulation of β-catenin. Nature Commun. 6, 7131 (2015); erratum 6, 7761 (2015).
Sinha, M. et al. Restoring systemic GDF11 levels reverses age-related dysfunction in mouse skeletal muscle. Science 344, 649–652 (2014).
Conboy, M. J., Conboy, I. M. & Rando, T. A. Heterochronic parabiosis: historical perspective and methodological considerations for studies of aging and longevity. Aging Cell 12, 525–530 (2013).
Castellano, J. M., Kirby, E. D. & Wyss-Coray, T. Blood-borne revitalization of the aged brain. JAMA Neurol. 72, 1191–1194 (2015).
Kim, M. J., Miller, C. M., Shadrach, J. L., Wagers, A. J. & Serwold, T. Young, proliferative thymic epithelial cells engraft and function in aging thymuses. J. Immunol. 194, 4784–4795 (2015).
Middeldorp, J. et al. Preclinical assessment of young blood plasma for Alzheimer disease. JAMA Neurol. http://dx.doi.org/10.1001/jamaneurol.2016.3185 (2016).
Sonntag, W. E., Ramsey, M. & Carter, C. S. Growth hormone and insulin-like growth factor-1 (IGF-1) and their influence on cognitive aging. Ageing Res. Rev. 4, 195–212 (2005).
Trejo, J. L., Carro, E. & Torres-Aleman, I. Circulating insulin-like growth factor I mediates exercise-induced increases in the number of new neurons in the adult hippocampus. J. Neurosci. 21, 1628–1634 (2001).
Stern, S. A., Kohtz, A. S., Pollonini, G. & Alberini, C. M. Enhancement of memories by systemic administration of insulin-like growth factor II. Neuropsychopharmacol. 39, 2179–2190 (2014).
Friedlander, A. L. et al. One year of insulin-like growth factor I treatment does not affect bone density, body composition, or psychological measures in postmenopausal women. J. Clin. Endocrinol. Metab. 86, 1496–1503 (2001).
Baker, L. D. et al. Effects of growth hormone-releasing hormone on cognitive function in adults with mild cognitive impairment and healthy older adults. Arch. Neurol. 69, 1420–1429 (2012).
Zhang, G. et al. Hypothalamic programming of systemic ageing involving IKK-β, NF-κB and GnRH. Nature 497, 211–216 (2013). Provides genetic evidence that the hypothalamus controls age-related inflammatory changes in the periphery.
Walker, R. G. et al. Biochemistry and biology of GDF11 and myostatin: similarities, differences, and questions for future investigation. Circ. Res. 118, 1125–1141 (2016).
Schafer, M. J. et al. Quantification of GDF11 and myostatin in human aging and cardiovascular disease. Cell Metab. 23, 1207–1215 (2016).
Acknowledgements
I would like to thank T. Montine at Stanford University for his critical reading of the manuscript. This work was supported by the US Department of Veterans Affairs and the US National Institute on Aging (AG045034).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
T.W.-C. is scientific adviser to and founder of Alkahest Inc., a company developing blood-based treatments to increase health span.
Additional information
Reprints and permissions information is available at www.nature.com/reprints.
Reviewer Information Nature thanks F. Gage, M. Mattson and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Rights and permissions
About this article
Cite this article
Wyss-Coray, T. Ageing, neurodegeneration and brain rejuvenation. Nature 539, 180–186 (2016). https://doi.org/10.1038/nature20411
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/nature20411


Majid Ali
ECT and EBR Therapies for Drug-Resistant Major Depression
The words ?Making Connections? begin the text on the cover of Nature of 10 November, 2016. In the first article of Nature Insight in it, Wyss-Coray writes: ?Indeed, factors in the circulation have been shown to modulate ageing and to rejuvenate numerous organs, including the brain.?(ref. 1). Concerning brain rejuvenation, I offer some comments on the continuing use of the term electroconvulsive therapy (ECT) to treat major depression which is refractory to multiple drug regimens.
In 1961, as a third-year medical student at King Edward Medical College, Lahore, Pakistan, on my bicycle I took my 12-year-old sister, Majida Begum, for ECT treatments. During the procedure, the psychiatrist asked me to firmly hold the back of her head between my hands. On the return bicycle trips, she said nothing about ECT, nor did I.
ECT was first used in 1938. Ever since, the issues concerning consent for treatment have drawn much attention. A 2008 national Scottish audit found that that 23% of patients who received the treatment were incapable of giving informed consent (ref. 2) A 2005 British review found that only about half of patients felt they were given sufficient information about ECT and its adverse effects (ref. 3).
Many patients with major depression not responding to multiple drug regimens ask me for advice about ECT. Our conversation proceeds something like the following:
?My psychiatrist has recommended ECT treatment. I?m in terror of it. What do you think?? the patient asks.
?It is considered the most effective treatment for severe depression which does not respond to antidepressant drugs. I suggest an informed second opinion before you take ECT,? I reply.
?You do know what ECT is, don?t you?? the patient asks tersely.
?Yes, I do.?
?What does ECT stand for?? the patient asks rhetorically.
?ECT is short for electroconvulsive therapy,? I respond.
?Those words terrorize me,? he protests.
?Well, well ?,? I search for the right words.
?Well what? Is this what you want me to do? Convulse to get better? Is that your idea of healing?? he fires his questions with rising frustration.
I look at him in silence, wondering how I myself might react to the possibility of receiving ECT. In earlier years, I had not heard the expressions like ?re-booting the brain,? and ?clearing the cobwebs of the brain? for explaining how ECT works.
One day, I saw a patient who was palpably tormented by intense suffering of unrelenting depression. He engaged me in a similar conversation. I tried to plumb the depth of his suffering for some moments, then stammered, wishing to say the following:
We can call ECT energy therapy. That is what ECT really is. It is energy that filters and re-directs cellular cross-talk in the brain. Really, that is all what ECT is ? an energy therapy, just like electronic devices for pain control. It is energy that first puts you to sleep. You do not feel any pain. You wake up just as you do after sleep. Honestly, that is all what ECT is.
As words rolled from my lips, I saw the face of patient soften somewhat. We looked at each other in silence for long moments. Encouraged I continued. This healing energy works by silencing hurtful voices in the brain, switching off cycles of fear and panic, so to speak. Obliterating deep despair of falling through black bottomless abyss of depression.
The patient looks at me intently and then takes some deep breaths. I continue. Years ago, my younger sister received this form of energy therapy. She did not complain of pain, may be a little headache sometimes. Nothing more. Honestly, nothing more. I stop. Again, we look at each other, searching for the meanings of the words.
?Energy healing, eh!? he says with doubt.
?Yes, energy therapy.? I reply matter-of-factly.
?Energy healing to change cellular cross-talk in the brain, as you put it.?
?Yes. Yes. It is energy that rejuvenates the brain, in a manner of speaking.?
?It is really that simple, is it?? he asks, tensing up again.
?I can complicate it if you want,? I try to lighten up the conversation. ?We could call it brain re-birthing. Or may be just energy brain rejuvenation.?
Holistic View of Neurotransmission and Depression
In the same Nature Insight, Canter and colleagues write the following: ?Emerging functional data are also shifting the aetiological focus of the disease from a neuron-centric view to an integrated outlook that acknowledges the synergistic functions of the different cell types of the brain. Consistent with this holistic view, numerous disrupted processes are interconnected in AD and interact to provoke a cycle of dysfunction as the disease progresses.? (ref. 4)
The above words succinctly state my case for the science and philosophy of holism in understanding and treating depression (ref. 5), in addition to the case of Alzheimer?s disease, for which the above-cited paragraph was written. So began the thinking which led me to coin the term ?energetic brain rejuvenation therapy? - the ?EBR therapy,? for short. I add that in my mind, brain rejuvenation means functional restoration of oxygen signaling (ref. 6), insulin homeostasis (ref. 7), brain cell membrane redox equilibrium (ref. 8), and generation of neuronal membrane action potential that releases neurotransmitters from vesicles which store them. In my view, all of this cannot be achieved without integratively addressing relevant issues of nutrition, metabolism, gut microbiota, hepatic detoxification pathways, and the matters of fears, anger, and anxiety. (ref.5-8).
References
1.	Wyss-Coray. Ageing, neurodegeneration and brain rejuvenation. Nature 2016;539:180.
2.	Ferguson G, et al. (2009) The Scottish ECT Accreditation Network (SEAN) Report 2009. Retreived 2010.05.24.
3.	Rose D, Wykes T, Bindman J, et al. Information, Consent, and perceived Coercion: Patient?s perspective on Ectroconvulsive Therapy. British Journal of Psychiatry. 2005;186:54-59.
4.	Canter RG, Penney J, Tsai L-H. The road to restoring neural circuits for the treatment of Alzheimer's disease. Nature. 2016; 539, 187?196.
5.	Ali M. Depression ? An Overview. http://drali1.org/depressio....
6.	Ali M. Oxygen and Aging. (2nd ed.) New York, Canary 21 Press. 2004.
7.	Ali M. The Dysox Model of Diabetes and De-Diabetization Potential. Townsend Letter-The examiner of Alternative Medicine. 2007; 286:137-145.
8.	Ali M. Oxidative coagulopathy in environmental illness. Environmental Management and Health. 2000;11:175-191.
Har Katcher
When I first published the basic method of heterochronic plasma exchange (Katche HL "Studies that shed a new light on aging", Biochemistry (Moscow) 2013, and before Wyss-Cory signed for a patent), it was on a different basis from what Wyss-Coray and the Conboys, Amy Wagers are trying to accomplish – finding 'the' factor or factors that blood-borne aging depends on – when it is clear that concentration effects are important and a host of different molecules, only some of which are proteins maybe be signalling or effector molecules of aging processes. The most obvious examples are the exosome encapsulated miRNAs that have been found to be taken up by other cells, changing the behavior of the recipient cells, or the stable long, non-coding RNAs found in the blood (lncRNAs). I believe this is reductionism at its worst – taking apart the goose to discover the source of golden eggs. If young plasma is sufficient to revitalize many vital tissues (and young epithelial tissues are not hard to obtain- or produce) then it should be a matter of engineering to reproduce this technique and to maximize it, as unlike heterochronic parabiosis, plasma exchange is a medically approved technology more than seventy years old, that can replace virtually all of the blood plasma of an old person with young plasma. If significant rejuvenation of rats and mice can be obtained, when according to size differences an old rate would only get something like 1/3 of its plasma replaced by young plasma, imagine what the replacement of 90-something percent might bring? The caveat is that the plasma must be unchanged with regards to unknown parameters or at least as close as we can get to 'virgin' plasma.