Abstract
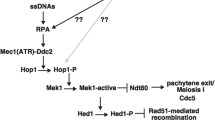
Replication protein A (RPA) is a highly conserved single-stranded DNA–binding protein involved in DNA replication, recombination and repair. We show here that RPA is present at the telomeres of the budding yeast Saccharomyces cerevisiae, with a maximal association in S phase. A truncation of the N-terminal region of Rfa2p (associated with the rfa2Δ40 mutated allele) results in severe telomere shortening caused by a defect in the in vivo regulation of telomerase activity. Cells carrying rfa2Δ40 show impaired binding of the protein Est1p, which is required for telomerase action. In addition, normal telomere length can be restored by expressing a Cdc13-Est1p hybrid protein. These findings indicate that RPA activates telomerase by loading Est1p onto telomeres during S phase. We propose a model of in vivo telomerase action that involves synergistic action of RPA and Cdc13p at the G-rich 3′ overhang of telomeric DNA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Longhese, M.P., Plevani, P. & Lucchini, G. Replication factor A is required in vivo for DNA replication, repair, and recombination. Mol. Cell. Biol. 14, 7884–7890 (1994).
Wold, M.S. Replication protein A: a heterotrimeric, single-stranded DNA-binding protein required for eukaryotic DNA metabolism. Annu. Rev. Biochem. 66, 61–92 (1997).
Smith, J., Zou, H. & Rothstein, R. Characterization of genetic interactions with RFA1: the role of RPA in DNA replication and telomere maintenance. Biochimie 82, 71–78 (2000).
Brill, S.J. & Stillman, B. Replication factor-A from Saccharomyces cerevisiae is encoded by three essential genes coordinately expressed at S phase. Genes Dev. 5, 1589–1600 (1991).
Brush, G.S., Morrow, D.M., Hieter, P. & Kelly, T.J. The ATM homologue MEC1 is required for phosphorylation of replication protein A in yeast. Proc. Natl. Acad. Sci. USA 93, 15075–15080 (1996).
Brush, G.S. & Kelly, T.J. Phosphorylation of the replication protein A large subunit in the Saccharomyces cerevisiae checkpoint response. Nucleic Acids Res. 19, 3725–3732 (2000).
Brush, G.S., Clifford, D.M., Marinco, S.M. & Bartrand, A.J. Replication protein A is sequentially phosphorylated during meiosis. Nucleic Acids Res. 29, 4808–4817 (2001).
Schramke, V. et al. The set1Δ mutation unveils a novel signaling pathway relayed by the Rad53-dependent hyperphosphorylation of replication protein A that leads to transcriptional activation of repair genes. Genes Dev. 15, 1845–1858 (2001).
McEachern, M.J., Krauskopf, A. & Blackburn, EH. Telomeres and their control. Annu. Rev. Genet. 34, 331–358 (2000).
Nugent, C.I. & Lundblad, V. The telomerase reverse transcriptase: components and regulation. Genes Dev. 12, 1073–1085 (1998).
Price, C.M. Synthesis of the telomeric C-strand. A review. Biochemistry 62, 1216–1223 (1997).
Wellinger, R.J., Wolf, A.J. & Zakian, V.A. Saccharomyces telomeres acquire single-strand TG1-3 tails late in S phase. Cell 72, 51–60 (1993).
Lin, J.J. & Zakian, V.A. The Saccharomyces CDC13 protein is a single-strand TG1-3 telomeric DNA-binding protein in vitro that affects telomere behavior in vivo. Proc. Natl. Acad. Sci. USA 93, 13760–13765 (1996).
Evans, S.K. & Lundblad, V. Est1 and Cdc13 as comediators of telomerase access. Science 286, 117–120 (1999).
Pennock, E., Buckley, K. & Lundblad, V. Cdc13 delivers separate complexes to the telomere for end protection and replication. Cell 104, 387–396 (2001).
Marcand, S., Brevet, V. & Gilson, E. Progressive cis-inhibition of telomerase upon telomere elongation. EMBO J. 18, 3509–3519 (1999).
Diede, S.J. & Gottschling, D.E. Telomerase-mediated telomere addition in vivo requires DNA primase and DNA polymerases α and δ. Cell 99, 723–733 (1999).
Taggart, A.K., Teng, S.C. & Zakian, V.A. Est1p as a cell cycle-regulated activator of telomere-bound telomerase. Science 297, 1023–1026 (2002).
Smith, C.D., Smith, D.L., DeRisi, J.L. & Blackburn, E.H. Telomeric protein distributions and remodeling through the cell cycle in Saccharomyces cerevisiae. Mol. Biol. Cell. 14, 556–570 (2003).
Bourns, B.D., Alexander, M.K., Smith, A.M. & Zakian, V.A. Sir proteins, Rif proteins, and Cdc13p bind Saccharomyces telomeres in vivo. Mol. Cell. Biol. 18, 5600–5608 (1998).
Gravel, S., Larrivee, M., Labrecque, P. & Wellinger, R.J. Yeast Ku as a regulator of chromosomal DNA end structure. Science 280, 741–744 (1998).
Huffman, K.E., Levene, S.D., Tesmer, V.M., Shay, J.W. & Wright, W.E. Telomere shortening is proportional to the size of the G-rich telomeric 3′-overhang. J. Biol. Chem. 275, 19719–19722 (2000).
Greenwell, P.W. et al. TEL1, a gene involved in controlling telomere length in S. cerevisiae, is homologous to the human ataxia telangiectasia gene. Cell 82, 823–829 (1995).
Craven, R.J. & Petes, T.D. Dependence of the regulation of telomere length on the type of subtelomeric repeat in the yeast Saccharomyces cerevisiae. Genetics 152, 1531–1541 (1999).
Peterson, S.E. et al. The function of a stem-loop in telomerase RNA is linked to the DNA repair protein Ku. Nat. Genet. 27, 64–67 (2001).
Polotnianka, R.M., Li, J. & Lustig, A.J. The yeast Ku heterodimer is essential for protection of the telomere against nucleolytic and recombinational activities. Curr. Biol. 8, 831–834 (1998).
Dornreiter, I. et al. Interaction of DNA polymerase α-primase with cellular replication protein A and SV40-T antigen. EMBO J. 11, 769–776 (1992).
Longhese, M.P., Neecke, H., Paciotti, V., Lucchini, G. & Plevani, P. The 70 kDa subunit of replication protein A is required for the G1/S and intra-S DNA damage checkpoints in budding yeast. Nucleic Acids Res. 24, 3533–3537 (1996).
Ritchie, K.B. & Petes, T.D. The Mre11p/Rad50p/Xrs2p complex and the Tel1p function in a single pathway for telomere maintenance in yeast. Genetics 155, 475–479 (2000).
Chan, S.W., Chang, J., Prescott, J. & Blackburn, E.H. Altering telomere structure allows telomerase to act in yeast lacking ATM kinases. Curr. Biol. 11, 1240–1250 (2001).
Tsukamoto, Y., Taggart, A.K. & Zakian, V.A. The role of the Mre11-Rad50-Xrs2 complex in telomerase- mediated lengthening of Saccharomyces cerevisiae telomeres. Curr. Biol. 11, 1328–1335 (2001).
Maniar, H.S., Wilson, R. & Brill, S.J. Roles of replication protein-A subunits 2 and 3 in DNA replication fork movement in Saccharomyces cerevisiae. Genetics 145, 891–902 (1997).
Mallory, J.C. et al. Amino acid changes in Xrs2p, Dun1p, and Rfa2p that remove the preferred targets of the ATM family of protein kinases do not affect DNA repair or telomere length in Saccharomyces cerevisiae. DNA Repair 2, 1041–1064 (2003).
Reichenbach, P. et al. A human homolog of yeast est1 associates with telomerase and uncaps chromosome ends when overexpressed. Curr. Biol. 13, 568–574 (2003).
Snow, B.E. et al. Functional conservation of the telomerase protein est1p in humans. Curr. Biol. 13, 698–704 (2003).
Tanaka, T. & Nasmyth, K. Association of RPA with chromosomal replication origins requires an Mcm protein, and is regulated by Rad53, and cyclin- and Dbf4-dependent kinases. EMBO J. 17, 5182–5191 (1998).
Martin, S.G., Laroche, T., Suka, N., Grunstein, M. & Gasser, S.M. Relocalization of telomeric Ku and SIR proteins in response to DNA strand breaks in yeast. Cell 97, 621–633 (1999).
Acknowledgements
We thank S. Brill for antibodies to Rfa2p, V. Lundblad for providing plasmid pVL1120 and plasmids used to Myc-tag Cdc13p and Est2p, and V. Zakian for reagents for the one-hybrid assay for telomeric proteins. Work in V.G.'s laboratory was supported by l'Association pour la Recherche sur le Cancer and by la Fondation pour la Recherche, work in E.G.'s laboratory by the Ligue Nationale contre le Cancer and work in M.P.L.'s laboratory by the Associazione Italiana per la Ricerca sul Cancro and by Telethon-Italy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Schramke, V., Luciano, P., Brevet, V. et al. RPA regulates telomerase action by providing Est1p access to chromosome ends. Nat Genet 36, 46–54 (2004). https://doi.org/10.1038/ng1284
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ng1284
This article is cited by
-
New insights into the mechanism of RPA in preserving genome stability
Genome Instability & Disease (2022)
-
RPA-coated single-stranded DNA as a platform for post-translational modifications in the DNA damage response
Cell Research (2015)
-
Human single-stranded DNA binding proteins are essential for maintaining genomic stability
BMC Molecular Biology (2013)
-
DNA-end capping by the budding yeast transcription factor and subtelomeric binding protein Tbf1
The EMBO Journal (2012)
-
RPA facilitates telomerase activity at chromosome ends in budding and fission yeasts
The EMBO Journal (2012)