Abstract
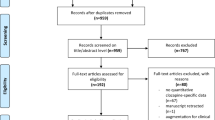
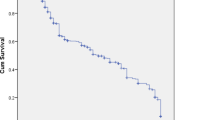
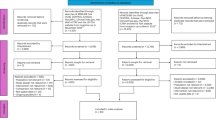
The large numbers of partial clozapine responders represent a major therapeutic challenge. Unfortunately, there are no clear data to support how best to treat these patients. This study examines the efficacy and safety of adjunctive risperidone in a well-defined treatment-resistant population optimally treated with clozapine. A total of 69 inpatients and outpatients with DSM-IV schizophrenia or schizoaffective disorder entered a 16-week double-blind, placebo-controlled, randomized clinical trial. Of them, 33 participants were randomized to risperidone and 36 were randomized to placebo. There was no significant group difference in the predefined response criteria. There were modest group differences for Brief Psychiatric Rating Scale (BPRS) positive symptoms, which were significant in the completer analysis (F=5.70; df=1, 70.3; p=0.02; ES=0.27) but not the intent-to-treat (ITT) analyses (F=3.01; df=1, 77.5; p=0.09; ES=0.19). A similar pattern was found for the BPRS total score, with the completer analysis showing a significant improvement in the risperidone group (F=5.21; df=1, 64.9; p=0.03; ES=0.27), whereas the ITT analysis was not significant (F=3.52; df=1, 71.3; p=0.06; ES=0.22). In addition, there was a small, but significant, group difference for negative symptoms, as measured by the SANS total score, which favored the risperidone group (F=5.67; df=1, 78.7; p=0.02; ES=0.24). There were no significant group differences on safety measures, including neuropsychological test and extrapyramidal symptom scores. A significant elevation of prolactin in the risperidone group was observed. The study results suggest that adjunctive risperidone may have a modest benefit for treatment-resistant clozapine patients. The study results are discussed in the context of previous double-blind studies of adjunctive risperidone. (clinicaltrials.gov, trial number: NCT00056498).
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Anil Yağcioğlu AE, Kivircik Akdede BB, Turget TI, Tümüklü M, Yazici MK, Alptekin K et al (2005). A double-blind controlled study of adjunctive treatment with risperidone in schizophrenic patients partially responsive to clozapine: efficacy and safety. J Clin Psychopharmacol 66: 63–72.
Awad AG (1993). Subjective response to neuroleptics in schizophrenia. Schizophr Bull 19: 609–618.
Barnes TRE (1989). A rating scale for drug-induced akathisia. Br J Psychiatry 154: 672–676.
Beaulieu JM, Gainetdinov RR, Caron MG (2007). The Akt-GSK-3 signaling cascade in the actions of dopamine. Trends Pharmacol Sci 28: 166–172.
Benedict R (1997). Brief Visuospatial Memory Test—Revised. Psychological Assessment Resources: Odessa, FL.
Benjamini Y, Hochberg Y (1995). Controlling the false discovery rate: a new and powerful approach to multiple testing. J R Stat Soc, Ser B 57: 1289–1300.
Benton AL, Hamsher DdeS (1989). Multilingual Aphasia Examination. AJA Associates: Iowa City, IO.
Brandt J (1991). The Hopkins verbal learning test: development of a new verbal memory test with six equivalent forms. Clin Neuropsychol 5: 125–142.
Buchanan RW, Breier A, Kirkpatrick B, Ball P, Carpenter Jr WT (1998). Positive and negative symptom response to clozapine in schizophrenic patients with and without the deficit syndrome. Am J Psychiatry 155: 751–760.
Buchanan RW, Javitt DC, Marder SR, Schooler NR, Gold JM, McMahon RP et al (2007). The cognitive and negative symptoms in schizophrenia trial (CONSIST): the efficacy of glutamatergic agents for negative symptoms and cognitive impairments. Am J Psychiatry 164: 1593–1602.
Cochran SM, McKerchar CE, Morris BJ, Pratt JA (2002). Induction of differential patterns of local cerebral glucose metabolism and immediate-early genes by acute clozapine and haloperidol. Neuropharmacology 43: 394–407.
Conover WJ, Salsburg DS (1988). Locally most powerful tests for detecting treatment effects when only a subset of patients are expected to ‘respond’ to treatment. Biometrics 44: 189–196.
Cornblatt BA, Keilp JG (1994). Impaired attention, genetics, and the pathophysiology of schizophrenia. Schizophr Bull 20: 31–46.
Correll CU, Rummel-Kluge C, Corves C, Kane JM, Leucht S (2009). Antipsychotic combinations vs monotherapy in schizophrenia: a meta-analysis of randomized controlled trials. Schizophr Bull 35: 443–457.
Feinman RD (2009). Intention-to-treat. What is the question? Nutr Metab 6: 1.
First MB, Spitzer RL, Gibbon M, Williams J (1997). Structural Clinical Interview for DSM-IV Axis Disorders (SCID-IV). Biometrics Research Department, New York State Psychiatric Institute: New York, NY.
Freudenreich O, Henderson DC, Walsh JP, Culhane MA, Goff DC (2007). Risperidone augmentation for schizophrenia partially responsive to clozapine: a double-blind, placebo-controlled trial. Schizophr Res 92: 90–94.
Gold JM, Carpenter C, Randolph C, Goldberg TE, Weinberger DR (1997). Auditory working memory and Wisconsin Card Sorting Test performance in schizophrenia. Arch Gen Psychiatry 54: 159–165.
Guy W (1976). ECDEU Assessment Manual for Psychopharmacology. US Department of Health and Human Services publication (ADM) 76-338: Rockville, MD. pp. 534–535.
Hawk AB, Carpenter WT, Strauss JS (1975). Diagnostic criteria and five-year outcome in schizophrenia: a report from the International Pilot Study of Schizophrenia. Arch Gen Psychiatry 32: 343–347.
Heaton RK, Chelune GJ, Talley JL, Kay GG, Curtiss G (1993). Wisconsin Card Sorting Test, Manual. Psychological Assessment Resources: Odessa, FL.
Hershey T, Craft S, Glauser TA, Hale S (1998). Short-term and long-term memory in early temporal lobe dysfunction. Neuropsychology 12: 52–64.
Holcomb HH, Cascella NG, Thaker GK, Medoff DR, Dannals RF, Tamminga CA (1996). Functional sites of neuroleptic drug action in the human brain: PET/FDG studies with and without haloperidol. Am J Psychiatry 153: 41–49.
Honer WG, Thornton AE, Chen EY, Chan RC, Wong JO, Bergmann A, et al, Clozapine and Risperidone Enhancement (CARE) Study Group (2006). Clozapine alone versus clozapine and risperidone with refractory schizophrenia. N Engl J Med 354: 472–482.
Josiassen RC, Ashok J, Kohegyi E, Stokes S, Dadvand M, Paing WW et al (2005). Clozapine augmented with risperidone in the treatment of schizophrenia: a randomized double blind placebo controlled trial. Am J Psychiatry 162: 130–136.
Kane J, Honigfeld G, Singer J, Meltzer H, and the Clozarial Collaborative Study Group (1988). Clozapine for the treatment-resistant schizophrenia: a double-blind comparison with chlorpromazine. Arch Gen Psychiatry 45: 789–796.
Kane JM, Marder SR, Schooler NR, Wirshing WC, Umbricth D, Baker RW et al (2001). Clozapine and haloperidol in moderately refractory schizophrenia: a 6-month randomized and double-blind comparison. Arch Gen Psychiatry 58: 965–972.
Kapur S, Remington G, Jones C, Wilson A, DaSilva J, Houle S et al (1996). High levels of dopamine D2 receptor occupancy with low-dose haloperidol treatment: a PET study. Am J Psychiatry 153: 948–950.
Kapur S, Zipursky RB, Remington G (1999). Clinical and theoretical implications of 5-HT2 and D2 receptor occupancy of clozapine, risperidone, and olanzapine in schizophrenia. Am J Psychiatry 156: 286–293.
Kivircik Akdede BB, Anil Yağcioğlu AE, Alptekin K, Turgut TI, Tumuklu M, Yazici MK et al (2006). A double-blind study of combination of clozapine with risperidone in patients with schizophrenia: effects on cognition. J Clin Psychiatry 67: 1912–1919.
Kronig MH, Munne RH, Szymanski S, Safferman AZ, Pollack S, Cooper T et al (1995). Plasma clozapine levels and clinical response for treatment-refractory schizophrenic patients. Am J Psychiatry 152: 179–182.
Lahti AC, Holcomb HH, Weiler MA, Medoff DR, Tamminga CA (2003). Functional effects of antipsychotic drugs: comparing clozapine with haloperidol. Biol Psychiatry 53: 601–608.
Lewis SW, Barnes TR, Davies L, Murrary RM, Dunn G, Hayhurst KP et al (2006). Randomized controlled trial of effect of prescription of clozapine versus other second-generation antipsychotic drugs in resistant schizophrenia. Schizophr Bull 32: 715–723.
Lezak MD (2004). Neuropsychological Assessment. 4th edn, Oxford University Press: New York, NY.
Liang KY, Zeger S (1986). Longitudinal data analysis using generalized linear models. Biometrika 73: 13–22.
MacGibbon GA, Lawlor PA, Bravo R, Dragunow M (1994). Clozapine and haloperidol produce a differential pattern of immediate early gene expression in rat caudate-putamen, nucleus accumbens, lateral septum and islands of Calleja. Brain Res Mol Brain Res 23: 21–32.
Mantel N (1963). Chi-square tests with one degree of freedom: extensions of the Mantel-Haenszel procedure. J Am Stat Assoc 58: 690–700.
Masri B, Salahpour A, Didriksen M, Ghisi V, Beaulieu JM, Gainetdinov RR et al (2008). Antagonism of dopamine D2 receptor/β-arrestin 2 interaction is a common property of clinically effective antipsychotics. Proc Natl Acad Sci USA 105: 13656–13661.
Matthews C, Klove H (1964). Instruction Manual for the Adult Neuropsychological Battery. University of Wisconsin Medical School: Madison, WI.
McEvoy JP, Lieberman JA, Stroup TS, Dvais SM (2006). Effectiveness of clozapine versus olanzapine, quetiapine, and risperidone in patients with chronic schizophrenia who did not respond to prior atypical antipsychotic treatment. Am J Psychiatry 163: 600–610.
McMahon RP, Arndt S, Conley RR (2005). More powerful two-sample tests for differences in repeated measures of adverse effects in psychiatric trials when only some patients may be at risk. Stat Med 24: 11–21.
McMahon RP, Kelly DL, Boggs DL, Li L, Hu Q, Davis JM et al (2008). Feasibility of reducing the duration of placebo-controlled trials in schizophrenia research. Schizophr Bull 34: 292–301.
Nguyen TV, Kosofsky BE, Birnbaum R, Cohen BM, Hyman SE (1992). Differential expression of c-Fos and Zif268 in rat striatum after haloperidol, clozapine, and amphetamine. Proc Natl Acad Sci USA 89: 4270–4274.
Overall JE, Gorham DR (1962). The brief psychiatric rating scale. Psychol Rep 10: 799–812.
Paton C, Whittington C, Barnes TR (2007). Augmentation with a second antipsychotic in patients with schizophrenia who partially respond to clozapine: a meta-analysis. J Clin Psychopharmacol 27: 198–204.
Pepe MS, Whitaker RC, Seidel K (1999). Estimating and comparing univariate associations with application to the prediction of adult obesity. Stat Med 18: 163–173.
Perry PJ, Miller DD, Arndt SV, Cadoret RJ (1991). Clozapine and nonclozapine plasma concentrations and clinical response of treatment-refractory schizophrenic patients. Am J Psychiatry 148: 231–235.
Reitan RM (1958). Validity of the trail-making test as an indication of organic brain damage. Percept Mot Skills 8: 271–276.
Rosenheck R, Cramer J, Xu W, Thomas J, Henderson W, Frisman L et al (1998). A comparison of clozapine and haloperidol in hospitalized patients with refractory schizophrenia. Department of Veterans Affairs Cooperative Study Group on Clozapine in Refractory Schizophrenia. N Engl J Med 337: 809–815.
Shiloh R, Zemishlany Z, Aizenberg D, Radwan M, Schwartz B, Dorfman-Etrog P et al (1997). Sulpiride augmentation in people with schizophrenia partially responsive to clozapine. A double-blind, placebo-controlled study. Br J Psychiatry 171: 569–573.
Simpson GM, Angus JWS (1970). A rating scale for extrapyramidal side effects. Acta Psychiatr Scand 212: 11–19.
Acknowledgements
The study was supported by the NIMH grant R01 MH45074-11, Clozapine Treatment of Schizophrenic Patients (PI: RWB); NIMH grant P30 MH06850, Advanced Center for Intervention and Services Research Grant (PI: RWB); and the University of Maryland General Clinical Research Center grant M01 RR 16500, General Clinical Research Centers Program, National Center for Research Resources, NIH. Double-blind medications were provided by Ortho McNeil Janssen Scientific Affairs, LLC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Robert R Conley is a full-time employee and stockholder of Eli Lilly, and this study was completed while he was at the MPRC. James M Gold has been in the advisory boards of Astra-Zeneca and Pfizer, has served as a consultant for Glaxo-Smith-Kline, and has received royalty payments for the Brief Assessment of Cognition in Schizophrenia. Deanna L Kelly has been in the advisory boards of Solvay, Bristol Myers Squibb, and Janssen. Robert W Buchanan has been a DSMB member for Cephalon, Otsuka, and Pfizer; has served as a consultant for Abbott, Cypress Bioscience, Glaxo-Smith-Kline, Merck, Organon, Sanofi-Aventis, Solvay, and Wyeth; and has been in the advisory boards of Astra-Zeneca, Pfizer, and Roche. All the remaining authors have no competing interests or financial support to disclose.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Weiner, E., Conley, R., Ball, M. et al. Adjunctive Risperidone for Partially Responsive People with Schizophrenia Treated with Clozapine. Neuropsychopharmacol 35, 2274–2283 (2010). https://doi.org/10.1038/npp.2010.101
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/npp.2010.101
Keywords
This article is cited by
-
Treatment of Clozapine Nonresponders
Current Treatment Options in Psychiatry (2019)
-
Betahistine effects on weight-related measures in patients treated with antipsychotic medications: a double-blind placebo-controlled study
Psychopharmacology (2018)
-
Serum galectin-3, but not galectin-1, levels are elevated in schizophrenia: implications for the role of inflammation
Psychopharmacology (2017)
-
Nutzen und Risiken der Polypharmazie
NeuroTransmitter (2012)