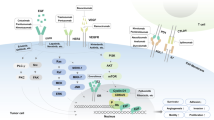
Abstract
The plethora of novel molecular-targeted agents (MTAs) has provided an opportunity to selectively target pathways involved in carcinogenesis and tumour progression. Combination strategies of MTAs are being used to inhibit multiple aberrant pathways in the hope of optimizing antitumour efficacy and to prevent development of resistance. While the selection of specific agents in a given combination has been based on biological considerations (including the role of the putative targets in cancer) and the interactions of the agents used in combination, there has been little exploration of the possible enhanced toxicity of combinations resulting from alterations in multiple signalling pathways in normal cell biology. Owing to the complex networks and crosstalk that govern normal and tumour cell proliferation, inhibiting multiple pathways with MTA combinations can result in unpredictable disturbances in normal physiology. This Review focuses on the main toxicities and the lack of tolerability of some common MTA combinations, particularly where evidence of enhanced toxicity compared to either agent alone is documented or there is development of unexpected toxicity. Toxicities caused by MTA combinations highlight the need to introduce new preclinical testing paradigms early in the drug development process for the assessment of chronic toxicities resulting from such combinations.
Key Points
-
There is increasing interest in combining molecular-targeted agents (MTAs) because of the growing understanding of the complexity and heterogeneity of the signalling networks controlling solid tumour growth
-
While numerous combination trials of MTAs that target select dysregulated pathways have been conducted, there has been little exploration of the molecular vulnerability of normal tissues to these combinations
-
For combinations using inhibitors of VEGF, EGFR, mTOR, and HER2 pathways, effective tolerable doses have been difficult to deliver owing to overlapping toxicities, enhanced single-agent toxicity, and/or unexpected toxicities
-
There is an urgent need for strategies to evaluate the consequences of multitarget inhibition in tumours and normal tissues
-
Development of biomarkers of efficacy and toxicity of MTA combinations, and for preclinical pharmacokinetic and pharmacodynamic modelling to guide schedules and dose adjustments are needed
-
Such biomarkers will help determine the recommended phase II doses based on acute and chronic toxicities associated with the use of MTA combinations
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Chapman, P. B. et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 364, 2507–2516 (2011).
Swanton, C. Intratumor heterogeneity: evolution through space and time. Cancer Res. 72, 4875–4882 (2012).
Loque, J. S. & Morrison, D. K. Complexity in the signalling network: insights from the use of targeted inhibitors in cancer therapy. Genes Dev. 26, 641–650 (2012).
Humphrey, R. W. et al. Opportunities and challenges in the development of experimental drug combinations for cancer. J. Natl Cancer Inst. 103, 1222–1226 (2011).
Kummar, S. et al. Drug development in oncology: classical cytotoxics and molecularly targeted agents. Br. J. Clin. Pharmacol. 62, 15–26 (2006).
Feldman, D. R. et al. Phase I trial of bevacizumab plus escalated doses of sunitinib in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 27, 1432–1439 (2009).
DePrimo, S. E. et al. Circulating protein biomarkers of pharmacodynamic activity of sunitinib in patients with metastatic renal cell carcinoma: modulation of VEGF and VEGF-related proteins. J. Transl. Med. 5, 32 (2007).
Rini, B. I. et al. A phase I study of sunitinib plus bevacizumab in advanced solid tumors. Clin. Cancer Res. 15, 6277–6283 (2009).
Yang, J. C. et al. A randomized trial of bevacizumab, an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N. Engl. J. Med. 349, 427–434 (2003).
Burger, R. A., Sill, M. W., Monk, B. J., Greer, B. E. & Sorosky, J. I. Phase II trial of bevacizumab in persistent or recurrent epithelial ovarian cancer or primary peritoneal cancer: a Gynecologic Oncology Group Study. J. Clin. Oncol. 25, 5165–5171 (2007).
Friedman, H. S. et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J. Clin. Oncol. 27, 4733–4740 (2009).
Monk, B. J. et al. Phase II trial of bevacizumab in the treatment of persistent or recurrent squamous cell carcinoma of the cervix: a gynecologic oncology group study. J. Clin. Oncol. 27, 1069–1074 (2009).
Bukowski, R. M. et al. Randomized phase II study of erlotinib combined with bevacizumab compared with bevacizumab alone in metastatic renal cell cancer. J. Clin. Oncol. 25, 4536–4541 (2007).
Barrios, C. H. et al. Phase II trial of continuous once-daily dosing of sunitinib as first-line treatment in patients with metastatic renal cell carcinoma. Cancer 118, 1252–1259 (2012).
Mahmood, S. T. et al. Phase II study of sunitinib malate, a multi-targeted tyrosine kinase inhibitor in patients with relapsed or refractory soft tissue sarcomas. Focus on 3 prevalent histologies: leiomyosarcoma, liposarcoma, and malignant fibrous histiocytoma. Int. J. Cancer 129, 1963–1969 (2011).
Barrios, C. H. et al. Phase III randomized trial of sunitinib versus capecitabine in patients with previously treated HER2-negative advanced breast cancer. Breast Cancer Res. Treat. 121, 121–131 (2010).
Raymond, E. et al. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N. Engl. J. Med. 364, 501–513 (2011).
Motzer, R. J. et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N. Engl. J. Med. 356, 115–124 (2007).
Kulke, M. H. et al. Activity of sunitinib in patients with advanced neuroendocrine tumors. J. Clin. Oncol. 26, 3403–3410 (2008).
Rini, B. I. et al. Toxicity of sunitinib plus bevacizumab in renal cell carcinoma. J. Clin. Oncol. 28, e284–e285; author reply e286–e287 (2010).
Azad, N. S. et al. Combination targeted therapy with sorafenib and bevacizumab results in enhanced toxicity and antitumor activity. J. Clin. Oncol. 26, 3709–3714 (2008).
Sosman, J. A. et al. Updated results of phase I trial of sorafenib (S) and bevacizumab (B) in patients with metastatic renal cell cancer (mRCC) [abstract]. J. Clin. Oncol. 26 (Suppl.), a5011 (2008).
Lee, J. M. et al. Combination therapy: intermittent sorafenib with bevacizumab yields activity and decreased toxicity. Br. J. Cancer 102, 495–499 (2010).
Escudier, B. et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N. Engl. J. Med. 356, 125–134 (2007).
Llovet, J. M. et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 359, 378–390 (2008).
Procopio, G. et al. Sorafenib with interleukin-2 vs sorafenib alone in metastatic renal cell carcinoma: the ROSORC trial. Br. J. Cancer 104, 1256–1261 (2011).
Grignani, G. et al. A phase II trial of sorafenib in relapsed and unresectable high-grade osteosarcoma after failure of standard multimodal therapy: an Italian Sarcoma Group study. Ann. Oncol. 23, 508–516 (2012).
Rini, B. I. et al. Comparative effectiveness of axitinib versus sorafenib in advanced renal cell carcinoma (AXIS): a randomised phase 3 trial. Lancet 378, 1931–1939 (2011).
Mahalingam, D. et al. Phase II study evaluating the efficacy, safety, and pharmacodynamic correlative study of dual antiangiogenic inhibition using bevacizumab (B) in combination with sorafenib (S) in patients (pts) with advanced malignant melanoma [abstract]. J. Clin. Oncol. 28 (Suppl.), e19008 (2010).
Galanis, E. et al. NCCTG phase II trial of bevacizumab in combination with sorafenib in recurrent GBM [abstract]. J. Clin. Oncol. 28 (Suppl.), a2018 (2010).
Tortora, G., Ciardiello, F. & Gasparini, G. Combined targeting of EGFR-dependent and VEGF-dependent pathways: rationale, preclinical studies and clinical applications. Nat. Clin. Pract. Oncol. 5, 521–530 (2008).
Herbst, R. S. et al. Phase I/II trial evaluating the anti-vascular endothelial growth factor monoclonal antibody bevacizumab in combination with the HER-1/epidermal growth factor receptor tyrosine kinase inhibitor erlotinib for patients with recurrent non-small-cell lung cancer. J. Clin. Oncol. 23, 2544–2555 (2005).
Ko, A. H. et al. A phase II study of bevacizumab plus erlotinib for gemcitabine-refractory metastatic pancreatic cancer. Cancer Chemother. Pharmacol. 66, 1051–1057 (2010).
Hainsworth, J. D. et al. Treatment of metastatic renal cell carcinoma with a combination of bevacizumab and erlotinib. J. Clin. Oncol. 23, 7889–7896 (2005).
Cohen, E. E. et al. Erlotinib and bevacizumab in patients with recurrent or metastatic squamous-cell carcinoma of the head and neck: a phase I/II study. Lancet Oncol. 10, 247–257 (2009).
Dickler, M. N. et al. A phase II trial of erlotinib in combination with bevacizumab in patients with metastatic breast cancer. Clin. Cancer Res. 14, 7878–7883 (2008).
Herbst, R. S. et al. Phase II study of efficacy and safety of bevacizumab in combination with chemotherapy or erlotinib compared with chemotherapy alone for treatment of recurrent or refractory non small-cell lung cancer. J. Clin. Oncol. 25, 4743–4750 (2007).
Thomas, M. B. et al. Phase II trial of the combination of bevacizumab and erlotinib in patients who have advanced hepatocellular carcinoma. J. Clin. Oncol. 27, 843–850 (2009).
Jackman, D. M. et al. Erlotinib plus bevacizumab in previously treated patients with malignant pleural mesothelioma. Cancer 113, 808–814 (2008).
Hainsworth, J. D. et al. Phase II trial of bevacizumab and erlotinib in carcinomas of unknown primary site: the Minnie Pearl Cancer Research Network. J. Clin. Oncol. 25, 1747–1752 (2007).
Dingemans, A. M. et al. First-line erlotinib and bevacizumab in patients with locally advanced and/or metastatic non-small-cell lung cancer: a phase II study including molecular imaging. Ann. Oncol. 22, 559–566 (2011).
Lubner, S. J. et al. Report of a multicenter phase II trial testing a combination of biweekly bevacizumab and daily erlotinib in patients with unresectable biliary cancer: a phase II Consortium study. J. Clin. Oncol. 28, 3491–3497 (2010).
Rohrberg, K. S. et al. Phase II trial of erlotinib and bevacizumab in patients with advanced upper gastrointestinal cancers. Acta. Oncol. 51, 234–242 (2012).
Philip, P. A. et al. Phase 2 study of bevacizumab plus erlotinib in patients with advanced hepatocellular cancer. Cancer 118, 2424–2430 (2012).
Herbst, R. S. et al. Efficacy of bevacizumab plus erlotinib versus erlotinib alone in advanced non-small-cell lung cancer after failure of standard first-line chemotherapy (BeTa): a double-blind, placebo-controlled, phase 3 trial. Lancet 377, 1846–1854 (2011).
Ko, A. H. et al. Phase II, randomized, open-label study of cetuximab and bevacizumab alone or in combination with fixed-dose rate (FDR) gemcitabine as first-line therapy for patients with metastatic adenocarcinoma of the pancreas (MPC) [abstract]. 2009 ASCO Gastrointestinal Cancers Symposium (abstract 183) (2009). http://www.asco.org/ASCOv2/Meetings/Abstracts?&vmview=abst_detail_view&confID=63&abstractID=10368
Saltz, L. B. et al. Randomized phase II trial of cetuximab, bevacizumab, and irinotecan compared with cetuximab and bevacizumab alone in irinotecan-refractory colorectal cancer: the BOND-2 study. J. Clin. Oncol. 25, 4557–4561 (2007).
Tol, J. et al. Chemotherapy, bevacizumab, and cetuximab in metastatic colorectal cancer. N. Engl. J. Med. 360, 563–572 (2009).
Sobrero, A. F. et al. EPIC: phase III trial of cetuximab plus irinotecan after fluoropyrimidine and oxaliplatin failure in patients with metastatic colorectal cancer. J. Clin. Oncol. 26, 2311–2319 (2008).
Bokemeyer, C. et al. Fluorouracil, leucovorin, and oxaliplatin with and without cetuximab in the first-line treatment of metastatic colorectal cancer. J. Clin. Oncol. 27, 663–671 (2009).
Van Cutsem, E. et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N. Engl. J. Med. 360, 1408–1417 (2009).
Pirker, R. et al. Cetuximab plus chemotherapy in patients with advanced non-small-cell lung cancer (FLEX): an open-label randomised phase III trial. Lancet 373, 1525–1531 (2009).
Lynch, T. J. et al. Cetuximab and first-line taxane/carboplatin chemotherapy in advanced non-small-cell lung cancer: results of the randomized multicenter phase III trial BMS099. J. Clin. Oncol. 28, 911–917 (2010).
Philip, P. A. et al. Phase III study comparing gemcitabine plus cetuximab versus gemcitabine in patients with advanced pancreatic adenocarcinoma: Southwest Oncology Group–directed intergroup trial S0205. J. Clin. Oncol. 28, 3605–3610 (2010).
Duran, I. et al. Phase I targeted combination trial of sorafenib and erlotinib in patients with advanced solid tumors. Clin. Cancer Res. 13, 4849–4857 (2007).
Quintela-Fandino, M. et al. Phase I combination of sorafenib and erlotinib therapy in solid tumors: safety, pharmacokinetic, and pharmacodynamic evaluation from an expansion cohort. Mol. Cancer Ther. 9, 751–760 (2010).
Lind, J. S. et al. A multicenter phase II study of erlotinib and sorafenib in chemotherapy-naive patients with advanced non-small cell lung cancer. Clin. Cancer Res. 16, 3078–3087 (2010).
Gridelli, C. et al. Sorafenib in combination with erlotinib or with gemcitabine in elderly patients with advanced non-small-cell lung cancer: a randomized phase II study. Ann. Oncol. 22, 1528–1534 (2011).
Spigel, D. R. et al. Randomized, double-blind, placebo-controlled, phase II trial of sorafenib and erlotinib or erlotinib alone in previously treated advanced non-small-cell lung cancer. J. Clin. Oncol. 29, 2582–2589 (2011).
Cho, B. et al. A multicenter phase II study of sorafenib in combination with erlotinib in patients with advanced non-small cell lung cancer (NSCLC) [abstract]. J. Clin. Oncol. 28 (Suppl.), a7547 (2010).
Prados, M. et al. Phase I/II study of sorefenib and erlotinib for patients with recurrent glioblastoma (GBM) (NABTC 05–02) [abstract]. J. Clin. Oncol. 27 (Suppl.), a2005 (2009).
Shepherd, F. A. et al. Erlotinib in previously treated non-small-cell lung cancer. N. Engl. J. Med. 353, 123–132 (2005).
Cappuzzo, F. et al. Erlotinib as maintenance treatment in advanced non-small-cell lung cancer: a multicentre, randomised, placebo-controlled phase 3 study. Lancet Oncol. 11, 521–529 (2010).
Natale, R. B. et al. Phase III trial of vandetanib compared with erlotinib in patients with previously treated advanced non-small-cell lung cancer. J. Clin. Oncol. 29, 1059–1066 (2011).
Ryan, C. W. et al. A dose-escalation phase II study of sunitinib (S) plus erlotinib (E) in advanced renal carcinoma (RCC) [abstract]. Proc. Am. Soc. Clin. Oncol. (Genitourinary Cancers Symposium) 2008 (abstract 361) (2008). http://www.asco.org/ASCOv2/Meetings/Abstracts?&vmview=abst_detail_view&confID=54&abstractID=20029.
Ryan, C. W. et al. A phase II study of sunitinib (S) plus erlotinib (E) in advanced renal carcinoma (RCC) [abstract]. J. Clin. Oncol. 28 (Suppl.), a4528 (2010).
O'Mahar, S. E. et al. Phase I study of sunitinib and erlotinib in advanced nonsquamous non-small cell lung cancer. J. Thorac. Oncol. 6, 951–953 (2011).
Adjei, A. A. et al. Phase I trial of sorafenib in combination with gefitinib in patients with refractory or recurrent non-small cell lung cancer. Clin. Cancer Res. 13, 2684–2691 (2007).
Motzer, R. J. et al. Phase I/II trial of sunitinib plus gefitinib in patients with metastatic renal cell carcinoma. Am. J. Clin. Oncol. 33, 614–618 (2010).
Thatcher, N. et al. Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: results from a randomised, placebo-controlled, multicentre study (Iressa Survival Evaluation in Lung Cancer). Lancet 366, 1527–1537 (2005).
Crinò, L. et al. Gefitinib versus vinorelbine in chemotherapy-naive elderly patients with advanced non-small-cell lung cancer (INVITE): a randomized, phase II study. J. Clin. Oncol. 26, 4253–4260 (2008).
Stewart, J. S. et al. Phase III study of gefitinib compared with intravenous methotrexate for recurrent squamous cell carcinoma of the head and neck [corrected]. J. Clin. Oncol. 27, 1864–1871 (2009).
Dejonge, M. et al. A phase I, open-label study of the safety and pharmacokinetics (PK) of pazopanib (P) and lapatinib (L) administered concurrently [abstract]. J. Clin. Oncol. 24 (18 Suppl.), a3088 (2006).
Monk, B. J. et al. Phase II, open-label study of pazopanib or lapatinib monotherapy compared with pazopanib plus lapatinib combination therapy in patients with advanced and recurrent cervical cancer. J. Clin. Oncol. 22, 3562–3569 (2010).
Slamon, D. et al. Randomized study of pazopanib + lapatinib vs. lapatinib alone in patients with HER2-positive advanced or metastatic breast cancer [abstract]. J. Clin. Oncol. 26 (Suppl.), a1016 (2008).
Land, S. C. & Tee, A. R. Hypoxia-inducible factor 1 alpha is regulated by the mammalian target of rapamycin (mTOR) via an mTOR signaling motif. J. Biol. Chem. 282, 20534–20543 (2007).
Del Bufalo, D. et al. Antiangiogenic potential of the mammalian target of rapamycin inhibitor temsirolimus. Cancer Res. 66, 5549–5554 (2006).
Zafar, Y. et al. Preliminary results of a phase I study of bevacizumab (BV) in combination with everolimus (E) in patients with advanced solid tumors [abstract]. J. Clin. Oncol. 24 (Suppl.), a3097 (2006).
Hainsworth, J. D. et al. Phase II trial of bevacizumab and everolimus in patients with advanced renal cell carcinoma. J. Clin. Oncol. 28, 2131–2136 (2010).
Hainsworth, J. D. et al. Bevacizumab and everolimus in the treatment of patients with metastatic melanoma: a phase 2 trial of the Sarah Cannon Oncology Research Consortium. Cancer 116, 4122–4129 (2010).
Altomare, I. et al. A phase II trial of bevacizumab plus everolimus for patients with refractory metastatic colorectal cancer. Oncologist 16, 1131–1137 (2011).
Motzer, R. J. et al. Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet 372, 449–456 (2008).
Yao, J. C. et al. Everolimus for advanced pancreatic neuroendocrine tumors. N. Engl. J. Med. 364, 514–523 (2011).
Negrier, S. et al. Temsirolimus and bevacizumab, or sunitinib, or interferon alfa and bevacizumab for patients with advanced renal cell carcinoma (TORAVA): a randomised phase 2 trial. Lancet Oncol. 12, 673–680 (2011).
Escudier, B. J. et al. Can. the combination of temsirolimus and bevacizumab improve the treatment of metastatic renal cell carcinoma (mRCC)? Results of the randomized TORAVA phase II trial [abstract]. J. Clin. Oncol. 28 (Suppl.), a4516 (2010).
Hudes, G. et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N. Engl. J. Med. 356, 2271–2281 (2007).
Bellmunt, J., Szczylik, C., Feingold, J., Strahs, A. & Berkenblit, A. Temsirolimus safety profile and management of toxic effects in patients with advanced renal cell carcinoma and poor prognostic features. Ann. Oncol. 19, 1387–1392 (2008).
Cohen, E. E. et al. A phase I study of sirolimus and bevacizumab in patients with advanced malignancies. Eur. J. Cancer 47, 1484–1489 (2011).
Patnaik, A. et al. A phase I, pharmacokinetic and pharmacodynamic study of sorafenib (S), a multi-targeted kinase inhibitor in combination with temsirolimus (T), an mTOR inhibitor in patients with advanced solid malignancies [abstract]. J. Clin. Oncol. 25 (Suppl.), a3512 (2007).
Wen, P. Y. et al. Phase I/II study of sorafenib and temsirolimus for patients with recurrent glioblastoma (GBM) (NABTC 05–02) [abstract]. J. Clin. Oncol. 27 (Suppl.), a2006 (2009).
Davies, M. A. et al. Phase I study of the combination of sorafenib and temsirolimus in patients with metastatic melanoma. Clin. Cancer Res. 18, 1120–1128 (2012).
Margolin, K. A. et al. Randomized phase II trial of sorafenib with temsirolimus or tipifarnib in untreated metastatic melanoma (S0438). Clin. Cancer Res. 18, 1129–1137 (2012).
Finn, R. S. et al. Phase I study of everolimus in combination with sorafenib in patients with advanced hepatocellular carcinoma (HCC) [abstract]. J. Clin. Oncol. 29 (Suppl.), a4074 (2011).
Harzstark, A. L. et al. A phase 1 study of everolimus and sorafenib for metastatic clear cell renal cell carcinoma. Cancer 117, 4194–4200 (2011).
Amato, R. J., Flaherty, A. L. & Stepankiw, M. Phase I trial of everolimus plus sorafenib for patients with advanced renal cell cancer. Clin. Genitourin. Cancer 10, 26–31 (2012).
Desar, I. M., Timmer-Bonte, J. N., Burger, D. M., van der Graaf, W. T. & van Herpen, C. M. A phase I dose-escalation study to evaluate safety and tolerability of sorafenib combined with sirolimus in patients with advanced solid cancer. Br. J. Cancer 103, 1637–1643 (2010).
Patel, P. H., Senico, P. L., Curiel, R. E. & Motzer, R. J. Phase I study combining treatment with temsirolimus and sunitinib malate in patients with advanced renal cell carcinoma. Clin. Genitourin. Cancer 7, 24–27 (2009).
Li, J. et al. Phase I study of sunitinib in combination with sirolimus in adults with advanced refractory malignancies [abstract]. J. Clin. Oncol. 27 (Suppl.), a3554 (2009).
Buck, E. et al. Rapamycin synergizes with the epidermal growth factor receptor inhibitor erlotinib in non-small-cell lung, pancreatic, colon, and breast tumors. Mol. Cancer Ther. 5, 2676–2684 (2006).
Mayer, I. A., Means-Powell, J., Shyr, Y. & Arteaga, C. L. A phase Ib trial of erlotinib, an EGFR inhibitor, and everolimus (RAD001), an mTOR inhibitor, in patients with metastatic breast cancer. 2009 Breast Cancer Symposium (abstract 254) (2009). http://www.asco.org/ASCOv2/Meetings/Abstracts?&vmview=abst_detail_view&confID=70&abstractID=40515.
Papadimitrakopoulou, V. et al. A phase 1/2 study investigating the combination of RAD001 (R) (everolimus) and erlotinib (E) as 2nd and 3rd line therapy in patients (pts) with advanced non-small cell lung cancer (NSCLC) previously treated with chemotherapy (C): phase 1 results [abstract]. J. Clin. Oncol. 26 (Suppl.), a8051 (2008).
Leighl, N. B. et al. Phase II study of everolimus plus erlotinib in previously treated patients with advanced non-small cell lung cancer (NSCLC). J. Clin. Oncol. 28 (Suppl.), a7524 (2010).
Milton, D. T. et al. Phase 1 trial of everolimus and gefitinib in patients with advanced nonsmall-cell lung cancer. Cancer 110, 599–605 (2007).
Price, K. A. et al. Phase II trial of gefitinib and everolimus in advanced non-small cell lung cancer. J. Thorac. Oncol. 5, 1623–1629 (2010).
Reardon, D. A. et al. Phase 2 trial of erlotinib plus sirolimus in adults with recurrent glioblastoma. J. Neurooncol. 96, 219–230 (2010).
Flaig, T. W. et al. Safety and efficacy of the combination of erlotinib and sirolimus for the treatment of metastatic renal cell carcinoma after failure of sunitinib or sorafenib. Br. J. Cancer 103, 796–801 (2010).
Bissler, J. J. et al. Sirolimus for angiomyolipoma in tuberous sclerosis complex or lymphangioleiomyomatosis. N. Engl. J. Med. 358, 140–151 (2008).
Rizell, M. et al. Effects of the mTOR inhibitor sirolimus in patients with hepatocellular and cholangiocellular cancer. Int. J. Clin. Oncol. 13, 66–70 (2008).
Davies, D. M. et al. Sirolimus therapy for angiomyolipoma in tuberous sclerosis and sporadic lymphangioleiomyomatosis: a phase 2 trial. Clin. Cancer Res. 17, 4071–4081 (2011).
Reardon, D. A. et al. Phase 1 trial of gefitinib plus sirolimus in adults with recurrent malignant glioma. Clin. Cancer Res. 12, 860–868 (2006).
Rothenberg, M. L. et al. Randomized phase II trial of the clinical and biological effects of two dose levels of gefitinib in patients with recurrent colorectal adenocarcinoma. J. Clin. Oncol. 23, 9265–9274 (2005).
Jermann, M. et al. A phase II, open-label study of gefitinib (IRESSA) in patients with locally advanced, metastatic, or relapsed renal-cell carcinoma. Cancer Chemother. Pharmacol. 57, 533–539 (2006).
Chang, S. M. et al. Phase I/II study of erlotinib and temsirolimus for patients with recurrent malignant gliomas (MG) (NABTC 04–02) [abstract]. J. Clin. Oncol. 27 (Suppl.), a2004 (2009).
Huang, S., Armstrong, E. A., Benavente, S., Chinnaiyan, P. & Harari, P. M. Dual-agent molecular targeting of the epidermal growth factor receptor (EGFR): combining anti-EGFR antibody with tyrosine kinase inhibitor. Cancer Res. 64, 5355–5362 (2004).
Janjigian, Y. Y. et al. Phase I/II trial of cetuximab and erlotinib in patients with lung adenocarcinoma and acquired resistance to erlotinib. Clin. Cancer Res. 17, 2521–2527 (2011).
Ramalingam, S. et al. Dual inhibition of the epidermal growth factor receptor with cetuximab, an IgG1 monoclonal antibody, and gefitinib, a tyrosine kinase inhibitor, in patients with refractory non-small cell lung cancer (NSCLC): a phase I study. J. Thorac. Oncol. 3, 258–264 (2008).
Baselga, J. et al. A phase I pharmacokinetic (PK) and molecular pharmacodynamic (PD) study of the combination of two anti-EGFR therapies, the monoclonal antibody (MAb) cetuximab and the tyrosine kinase inhibitor (TKI) gefitinib (G), in patients (pts) with advanced colorectal (CRC), head and neck (HNC), and non-small cell lung cancer (NSCLC) [abstract]. J. Clin. Oncol. 24 (Suppl.), a3006 (2006).
Scaltriti, M. et al. Lapatinib, a HER2 tyrosine kinase inhibitor, induces stabilization and accumulation of HER2 and potentiates trastuzumab-dependent cell cytotoxicity. Oncogene 28, 803–814 (2009).
Storniolo, A. M. et al. Phase I dose escalation and pharmacokinetic study of lapatinib in combination with trastuzumab in patients with advanced ErbB2-positive breast cancer. J. Clin. Oncol. 26, 3317–3323 (2008).
Blackwell, K. L. et al. Randomized study of Lapatinib alone or in combination with trastuzumab in women with ErbB2-positive, trastuzumab-refractory metastatic breast cancer. J. Clin. Oncol. 28, 1124–1130 (2010).
Scheuer, W. et al. Strongly enhanced antitumor activity of trastuzumab and pertuzumab combination treatment on HER2-positive human xenograft tumor models. Cancer Res. 69, 9330–9336 (2009).
Portera, C. C. et al. Cardiac toxicity and efficacy of trastuzumab combined with pertuzumab in patients with [corrected] human epidermal growth factor receptor 2-positive metastatic breast cancer. Clin. Cancer Res. 14, 2710–2716 (2008).
Baselga, J. et al. Phase II trial of pertuzumab and trastuzumab in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer that progressed during prior trastuzumab therapy. J. Clin. Oncol. 28, 1138–1144 (2010).
Gianni, L. et al. Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trial. Lancet Oncol. 13, 25–32 (2012).
Normanno, N. et al. Cooperative inhibitory effect of ZD1839 (Iressa) in combination with trastuzumab (Herceptin) on human breast cancer cell growth. Ann. Oncol. 13, 65–72 (2002).
Britten, C. D. et al. A phase I/II trial of trastuzumab plus erlotinib in metastatic HER2-positive breast cancer: a dual ErbB targeted approach. Clin. Breast Cancer 9, 16–22 (2009).
Cobleigh, M. A. et al. Multinational study of the efficacy and safety of humanized anti-HER2 monoclonal antibody in women who have HER2-overexpressing metastatic breast cancer that has progressed after chemotherapy for metastatic disease. J. Clin. Oncol. 17, 2639–2648 (1999).
Vogel, C. L. et al. Efficacy and safety of trastuzumab as a single agent in first-line treatment of HER2-overexpressing metastatic breast cancer. J. Clin. Oncol. 20, 719–726 (2002).
Arteaga, C. L. et al. A phase I-II study of combined blockade of the ErbB receptor network with trastuzumab and gefitinib in patients with HER2 (ErbB2)-overexpressing metastatic breast cancer. Clin. Cancer Res. 14, 6277–6283 (2008).
Baselga, J. et al. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N. Engl. J. Med. 366, 109–119 (2012).
Welch, C. M., Elliot, H., Danuser, G. & Hahn, K. M. Imaging the coordination of multiple signalling activities in living cells. Nat. Rev. Mol. Cell Biol. 12, 749–756 (2011).
Olson, H. et al. Concordance of the toxicity of pharmaceuticals in humans and in animals. Regul. Toxicol. Pharmacol. 32, 56–67 (2000).
Greaves, P., Williams, A. & Eve, M. First dose of potential new medicines to humans: how animals help. Nat. Rev. Drug Discov. 3, 226–236 (2004).
ICH. Guidance for industry: M3(R2) nonclinical safety studies for the conduct of human clinical trials and marketing authorization. U.S. Food and Drug Administration [online] (2010).
Ocana, A., Pandiella, A., Siu, L. L. & Tannock, I. F. Preclinical development of molecular-targeted agents for cancer. Nat. Rev. Clin. Oncol. 8, 200–209 (2010).
Ververis, K., Rodd, A. L., Tang, M. M., El-Osta, A. & Karagiannis, T. C. Histone deacetylase inhibitors augment doxorubicin-induced DNA damage in cardiomyocytes. Cell. Mol. Life Sci. 68, 4101–4114 (2011).
Brown, A. P., Dunstan, R. W., Courtney, C. L., Criswell, K. A. & Graziano, M. J. Cutaneous lesions in the rat following administration of an irreversible inhibitor of erbB receptors, including the epidermal growth factor receptor. Toxicol. Pathol. 36, 410–419 (2008).
Roberts, R. B., Arteaga, C. L. & Threadgill, D. W. Modeling the cancer patient with genetically engineered mice: prediction of toxicity from molecule-targeted therapies. Cancer Cell 5, 115–120 (2004).
Greger, J. G. et al. Combinations of BRAF, MEK, and PI3K/mTOR inhibitors overcome acquired resistance to the BRAF inhibitor GSK2118436 dabrafenib, mediated by NRAS or MEK mutations. Mol. Cancer Ther. 11, 909–920 (2012).
Hatzivassiliou, G. et al. RAF inhibitors prime wild-type RAF to activate the MAPK pathway and enhance growth. Nature 464, 431–435 (2010).
Poulikakos, P. I., Zhang, C., Bollag, G., Shokat, K. M. & Rosen, N. RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature 464, 427–430 (2010).
Corcoran, R. B. et al. BRAF V600 mutant colorectal cancer (CRC) expansion cohort from the phase I/II clinical trial of BRAF inhibitor dabrafenib (GSK2118436) plus MEK inhibitor trametinib (GSK1120212) [abstract]. J. Clin. Oncol. 30 (Suppl.), a3528 (2012).
Weber, J. S. et al. Updated safety and efficacy results from a phase I/II study of the oral BRAF inhibitor dabrafenib (GSK2118436) combined with the oral MEK 1/2 inhibitor trametinib (GSK1120212) in patients with BRAFi-naive metastatic melanoma. J. Clin. Oncol. 30 (Suppl.), a8510 (2012).
Hornberg, J. J., Bruggeman, F. J., Westerhoff, H. V. & Lankelma, J. Cancer: a systems biology disease. Biosystems 83, 81–90 (2006).
Van Cutsem, E. et al. Cetuximab plus irinotecan, fluorouracil, and leucovorin as first-line treatment for metastatic colorectal cancer: updated analysis of overall survival according to tumor KRAS and BRAF mutation status. J. Clin. Oncol. 29, 2011–2019 (2011).
Santoro, A. et al. A phase II randomized multicenter trial of gefitinib plus FOLFIRI and FOLFIRI alone in patients with metastatic colorectal cancer. Ann. Oncol. 19, 1888–1893 (2008).
Postel-Vinay, S. et al. Phase I trials of molecularly targeted agents: should we pay more attention to late toxicities? J. Clin. Oncol. 29, 1728–1735 (2011).
Shimizu, T. et al. The clinical effect of the dual-targeting strategy involving PI3K/AKT/mTOR and RAS/MEK/ERK pathways in patients with advanced cancer. Clin. Cancer Res. 18, 2316–2325 (2012).
Kummar, S. et al. Utilizing targeted cancer therapeutic agents in combination: novel approaches and urgent requirements. Nat. Rev. Drug Discov. 9, 843–856 (2010).
Kreisl, T. N. et al. A pilot study of everolimus and gefitinib in the treatment of recurrent glioblastoma (GBM). J. Neurooncol. 92, 99–105 (2009).
Weickhardt, A. J. et al. DUX study: A phase II study of evaluating dual targeting of the EGFR using the combination of cetuximab and erlotinib in patients with chemotherapy refractory metastatic colorectal cancer. J. Clin. Oncol. 28 (Suppl.), a3533 (2010).
Author information
Authors and Affiliations
Contributions
S. R. Park and S. Kummar made substantial contributions to researching the data, discussion of content, and writing, reviewing and editing of the manuscript before submission. M. Davis contributed to researching the data and writing the article. J. H. Doroshow contributed to writing, reviewing and editing of the manuscript and discussion of the content.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Park, S., Davis, M., Doroshow, J. et al. Safety and feasibility of targeted agent combinations in solid tumours. Nat Rev Clin Oncol 10, 154–168 (2013). https://doi.org/10.1038/nrclinonc.2012.245
Published:
Issue date:
DOI: https://doi.org/10.1038/nrclinonc.2012.245