Abstract
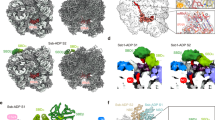
The pocket region of retinoblastoma tumour suppressor (Rb) is essential for tumour suppressing activity. The Rb pocket is primarily composed of two domains, A and B. We have determined the X-ray crystal structure of domain A (residues 378–562) at 2.3 Å resolution. Domain A consists of nine α-helices. The overall arrangement of helices in domain A is remarkably similar to the cyclin-box folds found in the crystal structures of cyclin A and TFIIB. This structure, along with domain B which is predicted to be homologous to the cyclin-box, suggests that the Rb pocket is composed of two cyclin-box fold domains. We present the structural/functional features of the Rb pocket, and the potential binding region for cellular or viral proteins within domain A.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Weinberg, R.A. The retinoblastoma protein and cell cycle control. Cell 81, 323–330 (1995).
Hollingsworth, R.E., Chen, P.-L. & Lee, W.-H. Integration of cell cycle control with transcriptional regulation by the retinoblastoma protein. Curr. Opin. Cell Biol. 5, 194–200 (1993).
Weinberg, R.A. The retinoblastoma gene and gene product. Cancer Surveys 12, 43–57 (1992).
Bookstein, R. et al. Suppression of tumorigenicity of human prostate carcinoma cells by replacing a mutated Rb gene. Science 247, 712–715 (1990).
Huang, H.-J. S. et al. Suppression of the neoplastic phenotype by replacement of the Rb gene in human cancer cells. Science 242, 1563–1566 (1988).
Dyson, N., Howley, P.M., MŸnger, K. & Harlow, E. The human papilloma virus-16 E7 oncoprotein is able to bind to the retinoblastoma gene product. Science 243, 934–937 (1989).
DeCaprio, J.A. et al. SV40 large tumor antigen forms a specific complex with the product of the retinoblastoma susceptibility gene. Cell 54, 275–283 (1988).
Horowitz, J.M. et al. Point mutational inactivation of the retinoblastoma antioncogene. Science 243, 937–940 (1989).
Huang, S. et al. Two distinct and frequently mutated regions of retinoblastoma protein are required for binding to SV40 T antigen. EMBO J. 9, 1815–1822 (1990).
Chellappan, S.P., Hiebert, S., Mudryj, M., Horowitz, J.M. & Nevins, J.R. The E2F transcriptional factor is a cellular target for the RB protein. Cell 65, 1053–1061 (1992).
Gu, W. et al. Interaction of myogenic factors and the retinoblastoma protein mediates muscle cell commitment and differentiation. Cell 72, 309–324 (1993)
White, R.J. et al. Repression of RNA polymerase III transcription by the retinoblastoma protein. Nature 382, 88–90 (1996).
Cavanaugh, A.H. et al. Activity of RNA polymerase I transcription factor UBF blocked by Rb gene product. Nature 374, 177–180 (1995).
Kouarides, T. Transcriptional control by the retinioblastoma protein. Semin.Cancer Biol. 6, 91–98 (1995).
Hagemeier, C., Bannister, A.J., Cook, A. & Kouzarides, T. The activation domain of transcription factor PU.1 binds the retinoblastoma (RB) protein and the transcription factor TFIID in vitro: RB shows sequence similarity to TFIID and TFIIB. Proc Natn. Acad. Sci. USA 90, 1580–1584 (1993).
Brown, N.R. et al. The crystal structure of Cyclin A. Structure 3, 1235–1247 (1995).
Jeffrey, P.D. et al. Mechanism of CDK activation revealed by the structure of a cyclinA–CDK2 complex. Nature 376, 313–320 (1995).
Bagby, S. et al. Solution structure of the C-terminal core domain of human TFIIB: similarity to Cyclin A and interaction with TATA-binding protein. Cell 82, 857–867 (1995).
Nikolov, D.B. et al. Crystal strucutre of a TFIIB-TBP-TATA element ternary complex. Nature 377, 119–128 (1995).
Ewen, M. B., Xing, Y., Lawrence, J. B. & Livingston, D. M. Molecular cloning, chromosomal mapping, and expression of the cDNA for p107, a retinoblastoma gene product-related protein. Cell 66, 1155–1164 (1991).
Li, Y. et al. The adenovirus E1 A-associated p130-kD protein is encoded by a member of the retinoblastoma gene family and physically interacts with cyclins A and E. Genes Dev. 7, 2366–2377 (1993).
Stirdivant, S. M., Ahern, J. D., Oliff, A. & Heimbrook, D. C. Retinoblastoma protein binding properties are dependent on 4 cysteine residues in the protein binding pocket. J. Biol. Chem. 267, 14846–14851 (1992).
Gibson, T. J., Thomson, J. D., Blocker, A. & Kouzarides, T. Evidence for a protein domain superfamily shared by the cyclins, TFIIB and RB/p107. Nucleic Adds Res. 22, 946–952 (1994).
Jones, D.T., Taylor, W.R. & Thornton, J.M. A new approach to protein fold recognition. Nature 358, 86–89 (1992).
Hensey, C. E. et al. Identification of discrete structural domains in the retinoblastoma protein. J. Biol. Chem. 269, 1380–1387 (1994).
Chow, K. N. B & Dean, D. C. Domain A and B in the Rb pocket interact to form a transcriptional represser motif. Mol. Cell. Biol. 16, 4862–4868 (1996).
Lees, J. A., Buchkovich, K. J., Marshak, D. R., Anderson, C. W. & Harlow, E. The retinoblastoma protein is phosphorylated on multiple sites byhuman cdc2. EMBO J. 10, 4279–4290 (1991).
Knudson, E.S. & Wang, J.Y.J. Differential regulation of retinoblastoma protein function by specific phosphorylation sites. J. Biol. Chem. 271, 8313–8320 (1996).
Russo, A.A., Jeffery, P.D. & Pavletich, N.P. Structural basis of cyclin–dependent kinase activation by phosphorylation. Nature Struct. Biol. 3, 696–700 (1996).
Kaelin, W. G., Palas, D.C., DeCaprio, J.A., Kaye, F.J. & Livingston, D. M. Identification of cellular proteins that can interact specifically with the T/E1 A-binding region of the retinoblastoma gene product. Cell 64, 521–532.
Collaborative Computational Project Number 4. The CCP4 Suite: Program for protein crystallography. Acta Crystallogr. D50, 760–763 (1994)
Zhang, K.Y.G. SQUASH-Combining constraints for macromolecular phase refinement and extension. Acta Crystallogr. D49, 213–222 (1993).
Jones, T.A., Zou, J.-Y. & Cowan, S.W. Improved methods of building protein models in electron density maps and the location errors in these models. Acta Crystallogr. A47, 110–119 (1991).
Sack, J.S. CHAIN: a crystallographic modelling program.J. molec. Graphics, 6, 224–225 (1988).
Brünger, A.T. X-PLOR, a system for crystallography and NMR, Version 3.1 (Yale Univ. Press, New Haven, CT, 1992)
Tronrud, D.E., Ten Eyck, L.F. & Matthews, B.W. An efficient general purpose Least-Squares Refinement Program for Macromolecular Structures. Acta Crystallogr. A43, 489–501 (1987).
Merrit, E.A. & Murphy, M.E.P. Raster 3D Version 2.0. A program for photorealistic molecular graphics. Acta Crystallogr. D50, 869–873 (1994).
Kabsch, W. & Sander, C. Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features. Biopolymers. 22, 2577–2637 (1983).
Kraulis, P.J. Molscript: a program to produce both detailed and schematic plots of protein structures. J. appl. Crystallogr. 24, 946–950 (1991).
Nicholls, A., Sharp, K.A. & Honig, B. Protein folding and association: insight from the interracial and thermodynamic properties of hydrocarbons. Proteins:Structure, Function and Genetics, 11, 281–296 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, HY., Cho, Y. Structural similarity between the pocket region of retinoblastoma tumour suppressor and the cyclin-box. Nat Struct Mol Biol 4, 390–395 (1997). https://doi.org/10.1038/nsb0597-390
Received:
Accepted:
Issue date:
DOI: https://doi.org/10.1038/nsb0597-390