Abstract
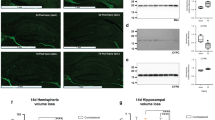
Caspases are believed to play a key role in the delayed neuronal cell death observed in the rat brain after hypoxic-ischemic (HI) insult. Caspase inhibitors have been developed as antiapoptotic agents. Hippocampal damage after HI insult is strongly related to tissue temperature, and systemic hypothermia has been introduced clinically for brain protection. In this study, we examined the effects of a caspase inhibitor and systemic hypothermia on neuronal protection in the developing rat brain. Postnatal d 7 rat pups were subjected to the Rice model of hypoxia for 1 h. Systemic hypothermia was induced with a water bath at 29°C. Before HI insult, a pan-caspase inhibitor, boc-aspartyl-(OMe)-fluoromethyl-ketone (BAF), was injected into the cerebral ventricle. The ipsilateral hippocampus was subjected to caspase assays and histologic assessment. The HI group at 37°C (HI-37°C) showed a peak of caspase-3 activity 16 h after insult. This activity was significantly reduced in the presence of BAF or hypothermia (HI-29°C group, p < 0.05) or by the combination of HI-29°C + BAF (p < 0.01 versus HI-37°C). The number of neuronal cells in the ipsilateral hippocampal CA1 region in the HI-37°C group was significantly decreased (62.9%versus control). The number of neuronal cells was maintained in the HI-37°C + BAF group (82.7%), the HI-29°C group (78.7%), and the combination group (95.2%) (p < 0.05 versus HI-37°C). A combination of systemic hypothermia and BAF produced a strong protective effect against neuronal damage in the developing rat brain, along with a reduction in caspase-3 activity.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- DNCD:
-
delayed neuronal cell death
- PCD:
-
programmed cell death
- PD:
-
postnatal day
- HI:
-
hypoxia and ischemia
- Ac-DEVD-MCA:
-
Ac-Asp-Glu-Val-Asp-MCA
- BAF:
-
boc-aspartyl-(OMe)-fluoromethyl-ketone
- ICV:
-
intracerebroventricular
- f-DNA:
-
fragmented DNA
References
Bruck Y, Bruck W, Kretzschmer HA, Lassmann H 1996 Evidence for neuronal apoptosis in pontosubicular neuron necrosis. Neuropathol Appl Neurobiol 22: 23–29
Pulsinelli WA, Brierley JB, Plum F 1982 Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann Neurol 11: 491–498
Linnik MD, Zobrist RH, Hatfield MD 1993 Evidence supporting a role for programmed cell death in focal cerebral ischemia in rats. Stroke 24: 2002–2008
Pettmann B, Henderson CE 1998 Neuronal cell death. Neuron 20: 633–647
Beilharz EJ, Williams CE, Dragunow M, Sirimanne ES, Gluckman PD 1995 Mechanisms of delayed cell death following hypoxic-ischemic injury in the immature rat: evidence for apoptosis during selective neuronal loss. Brain Res Mol Brain Res 29: 1–14
Tominaga T, Kure S, Narisawa K, Yoshimoto T 1993 Endonuclease activation following focal ischemic injury in the rat brain. Brain Res 608: 21–26
Pravdenkova SV, Basnakian AG, Jill SJ, Andersen BJ 1996 DNA fragmentation and nuclear endonuclease activity in rat brain after severe closed head injury. Brain Res 729: 151–155
Rink A, Fung KM, Trojanowski JQ, Lee VM, Neugebauer E, McIntosh TK 1995 Evidence of apoptotic cell death after experimental traumatic brain injury in the rat. J Pathol 147: 1575–1583
Cotman CW, Anderson AJ 1995 A potential role for apoptosis in neurodegeneration and Alzheimer's disease. Mol Neurobiol 10: 19–45
Mitchell IJ, Lawson S, Moser B, Laidlaw SM, Cooper AJ, Walkinshaw G, Waters CM 1995 Glutamate-induced apoptosis results in a loss of striatal neurons in the Parkinsonian rat. Neuroscience 63: 1–5
Portera-Cailliau C, Hedreen JC, Price DL, Koliatsos VE 1995 Evidence for apoptotic cell death in Huntington's disease and excitotoxic animal models. J Neurosci 15: 3775–3787
Thomas BL, Gates DJ, Richfield EK, O'Brian TF, Schweitzer JB, Steindler DA 1995 DNA end labeling (TUNEL) in Huntington's disease and other neuropathological conditions. Exp Neurol 133: 265–272
Braun JS, Novak R, Herzog KH, Bodner SM, Cleveland JL, Tuomanen EI 1999 Neuroprotection by a caspase inhibitor in acute bacterial meningitis. Nat Med 5: 298–302
Montgomery EME, Bardgett ME, Lall B, Csernansky CA, Csernansky JG 1999 Delayed neuronal loss after administration of intracerebroventricular kainic acid to preweanling rats. Brain Res Dev Brain Res 112: 107–116
Kondratyev A, Gale K 2000 Intracerebral injection of caspase-3 inhibitor prevents neuronal apoptosis after kainic acid-evoked status epilepticus. Brain Res Mol Brain Res 75: 216–224
Pollard H, Chantagrel S, Charriaut-Marlangue C, Moreau J, Ben-Ari Y . 1994 Apoptosis associated DNA fragmentation in epileptic brain damage. Neuroreport 5: 1053–1055
Chen J, Nagayama T, Jin K, Stetler RA, Zhu RL, Graham SH, Simon RP 1998 Induction of caspase-3-like protease may mediate delayed neuronal death in the hippocampus after transient cerebral ischemia. J Neurosci 18: 4914–4928
Gillardon F, Bottiger B, Schmits B, Zimmermann M, Hossmann KA 1997 Activation of CPP-32 protease in hippocampal neurons following ischemia and epilepsy. Brain Res Mol Brain Res 50: 16–22
Cheng Y, Deshmukh M, D'Costa A, Demaro JA, Gidday JM, Shah A, Sun Y, Jacquin MF, Johnson EM, Holtzman DM 1998 Caspase inhibitor affords neuroprotection with delayed administration in a rat model of neonatal hypoxic- ischemic brain injury. J Clin Invest 101: 1992–1999
Fink K, Zhu J, Namura S, Shimizu-Sasamata M, Endres M, Ma J, Dalkara T, Yuan J, Moskowitz MA 1998 Prolonged therapeutic window for ischemic brain damage caused by delayed caspase activation. J Cereb Blood Flow Metab 18: 1071–1076
Deshmukh M, Vasilakos J, Deckwerth TL, Lampe PA, Shivers BD, Johnson EM 1996 Genetic and metabolic status of NGF-derived sympathetic neurons saved by an inhibitor of ICE family proteases. J Cell Biol 135: 1341–1345
Himi T, Ishizaki Y, Murota S 1998 A caspase inhibitor blocks ischaemia-induced delayed neuronal death in the gerbil. Eur J Neurosci 10: 777–781
Ferrand-Drake M, Wieloch T 1999 The time course of DNA fragmentation in the choroid plexus and the CA1 region following transient global ischemia in the rat brain. The effect of intra-ischemic hypothermia. Neuroscience 93: 537–549
Dietrich WD, Busto R, Alonson O, Globus MY-T, Ginsberg MD 1993 Intraischemic but not postischemic brain hypothermia protects chronically following global forebrain ischemia in rats. J Cereb Blood Flow Metab 13: 541–549
Busto R, Dietrich WD, Globus MY-T 1987 Small differences in intraischemic brain temperature critically determine the extent of ischemic neuronal injury. J Cereb Blood Flow Metab 7: 729–738
Minamisawa H, Nordstrom CH, Smith ML, Siesjo BK 1990 The influence of mild body and brain hypothermia on ischemic brain damage. J Cereb Blood Flow Metab 10: 365–374
Welsh FA, Harris VA 1991 Postischemic hypothermia fails to reduce ischemic injury in gerbil hippocampus. J Cereb Blood Flow Metab 11: 617–620
Laptook AR, Corbett RJT, Sterett R, Burns DK, Tollefsbol G, Garcia D 1994 Modest hypothermia provides partial neuroprotection for ischemic neonatal brain. Pediatr Res 35: 436–442
Saeed D, Goetzman BW, Gospe SM 1993 Brain injury and protective effects of hypothermia using triphenyltetrazolium chloride in neonatal rat. Pediatr Neurol 9: 263–267
Towfighi J, Housman C, Heitjan DF, Vannucci RC, Yager JY 1994 The effect of focal cerebral cooling on perinatal hypoxic-ischemic brain damage. Acta Neuropathol Berl 87: 598–604
Yager JY, Towfighi J, Vannucci RC 1993 Influence of mild hypothermia on hypoxic-ischemic brain damage in the immature rat. Pediatr Res 34: 525–529
Bona E, Hagberg H, Loberg EM, Bagenholm R, Thoresen M 1998 Protective effects of moderate hypothermia after neonatal hypoxia-ischemia: short- and long-term outcome. Pediatr Res 43: 738–745
Morita T, Kato H, Iwanaga S, Takada K, Kimura T, Sakakibara S 1977 New fluorogenic substrates for α-thrombin, factor Xa, kallikreins, and urokinase. J Biochem 82: 1495–1498
Rice JE, Vanucci RC, Brierley JB 1981 The influence of immaturity on hypoxic-ischemic brain damage in the rat. Ann Neurol 9: 131–141
Cheng Y, Gidday JM, Yan Q, Shah AR, Holtzman DM 1997 Marked age-dependent neuroprotection by BDNF against neonatal hypoxic-ischemic brain injury. Ann Neurol 41: 521–529
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported, in part, by a Grant-Aid for Scientific Research (No. 11671069) from the Ministry of Education.
Rights and permissions
About this article
Cite this article
Adachi, M., Sohma, O., Tsuneishi, S. et al. Combination Effect of Systemic Hypothermia and Caspase Inhibitor Administration against Hypoxic-Ischemic Brain Damage in Neonatal Rats. Pediatr Res 50, 590–595 (2001). https://doi.org/10.1203/00006450-200111000-00010
Received:
Accepted:
Issue date:
DOI: https://doi.org/10.1203/00006450-200111000-00010
This article is cited by
-
Preserving Continence During Robotic Prostatectomy
Current Urology Reports (2013)
-
Therapeutic Hypothermia for Acute Neurological Injuries
Neurotherapeutics (2012)
-
L’arrêt cardiaque — Protection cérébrale post-arrêt cardiaque
Réanimation (2011)
-
Hypothermia in cardiogenic shock reduces systemic t-PA release
Journal of Thrombosis and Thrombolysis (2011)
-
Cannabinoid as a neuroprotective strategy in perinatal hypoxic-ischemic injury
Neuroscience Bulletin (2011)