Abstract
Background:
Extremely preterm birth is associated with subsequent behavioral problems. We hypothesized that perinatal systemic inflammation, a risk factor for cerebral white matter injury and cognitive impairment, is associated with behavior problems observed at 2 y.
Methods:
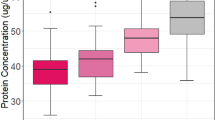
In a cohort of 600 children born before 28 wk gestation, we measured 25 inflammation-related proteins in blood collected on postnatal days 1, 7, and 14, and identified behavior problems using parent responses to the Child Behavior Checklist for Ages 1.5–5 (CBCL/1.5–5) at 2 y of age. A persistent or recurrent protein elevation was defined as a concentration in the highest quartile (for gestational age and postnatal age) on at least 2 d ~1 wk apart. Behavior problems were defined by CBCL/1.5–5 subscale scores at or above the 93rd percentile.
Results:
A single-day elevation of intercellular adhesion molecule-3 was associated with an increased risk of an attention problem, as were persistent or recurrent elevations of myeloperoxidase, interleukin-6, tumor necrosis factor-RI, interleukin-8, intercellular adhesion molecule-3, vascular endothelial growth factor-R1, and vascular endothelial growth factor-R2. These associations persisted among infants without white matter injury and cognitive impairment.
Conclusion:
Among children born extremely prematurely, recurrent, or persistent elevations of inflammation-related proteins in blood during in the first two postnatal weeks are associated with an attention problem at age 2 y.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
O’Shea TM, Downey LC, Kuban KK . Extreme prematurity and attention deficit: epidemiology and prevention. Front Hum Neurosci 2013;7:578.
Kirkhaug B, Drugli MB, Lydersen S, Mørch WT . Associations between high levels of conduct problems and co-occurring problems among the youngest boys and girls in schools: a cross-sectional study. Nord J Psychiatry 2013;67:225–32.
Polderman TJ, Boomsma DI, Bartels M, Verhulst FC, Huizink AC . A systematic review of prospective studies on attention problems and academic achievement. Acta Psychiatr Scand 2010;122:271–84.
Knapp M, King D, Healey A, Thomas C . Economic outcomes in adulthood and their associations with antisocial conduct, attention deficit and anxiety problems in childhood. J Ment Health Policy Econ 2011;14:137–47.
Reiss F . Socioeconomic inequalities and mental health problems in children and adolescents: a systematic review. Soc Sci Med 2013;90:24–31.
Leviton A, Kuban K, O’Shea TM, et al. The relationship between early concentrations of 25 blood proteins and cerebral white matter injury in preterm newborns: the ELGAN study. J Pediatr 2011;158:897–903.e1–5.
O’Shea TM, Allred EN, Kuban KC, et al.; Extremely Low Gestational Age Newborn (ELGAN) Study Investigators. Elevated concentrations of inflammation-related proteins in postnatal blood predict severe developmental delay at 2 years of age in extremely preterm infants. J Pediatr 2012;160:395–401.e4.
Kuban K, O’Shea TM, Allred EN, et al. Systemic inflammation and cerebral palsy risk in extremely preterm infants. J Child Neurol 2014; e-pub ahead of print 18 March 2014.
Reijneveld SA, de Kleine MJ, van Baar AL, et al. Behavioural and emotional problems in very preterm and very low birthweight infants at age 5 years. Arch Dis Child Fetal Neonatal Ed 2006;91:F423–8.
Scott MN, Taylor HG, Fristad MA, et al. Behavior disorders in extremely preterm/extremely low birth weight children in kindergarten. J Dev Behav Pediatr 2012;33:202–13.
Leviton A, Kuban KC, Allred EN, Fichorova RN, O’Shea TM, Paneth N ; ELGAN Study Investigators. Early postnatal blood concentrations of inflammation-related proteins and microcephaly two years later in infants born before the 28th post-menstrual week. Early Hum Dev 2011;87:325–30.
Meyer U, Feldon J, Dammann O . Schizophrenia and autism: both shared and disorder-specific pathogenesis via perinatal inflammation? Pediatr Res 2011;69(5 Pt 2):26R–33R.
Favrais G, van de Looij Y, Fleiss B, et al. Systemic inflammation disrupts the developmental program of white matter. Ann Neurol 2011;70:550–65.
Leviton A, Gressens P . Neuronal damage accompanies perinatal white-matter damage. Trends Neurosci 2007;30:473–8.
Elia J, Sackett J, Turner T, et al. Attention-deficit/hyperactivity disorder genomics: update for clinicians. Curr Psychiatry Rep 2012;14:579–89.
Vuillermot S, Joodmardi E, Perlmann T, Ögren SO, Feldon J, Meyer U . Prenatal immune activation interacts with genetic Nurr1 deficiency in the development of attentional impairments. J Neurosci 2012;32:436–51.
Miller SP, Cozzio CC, Goldstein RB, et al. Comparing the diagnosis of white matter injury in premature newborns with serial MR imaging and transfontanel ultrasonography findings. AJNR Am J Neuroradiol 2003;24:1661–9.
de la Fuente A, Xia SG, Branch C, Li XB . A review of attention-deficit/hyperactivity disorder from the perspective of brain networks. Front Hum Neurosci 2013; 7:192.
Shang CY, Wu YH, Gau SS, Tseng WY . Disturbed microstructural integrity of the frontostriatal fiber pathways and executive dysfunction in children with attention deficit hyperactivity disorder. Psychol Med 2013;43:1093–107.
Johnson S, Hollis C, Kochhar P, Hennessy E, Wolke D, Marlow N . Psychiatric disorders in extremely preterm children: longitudinal finding at age 11 years in the EPICure study. J Am Acad Child Adolesc Psychiatry 2010;49:453–63.e1.
McElrath TF, Hecht JL, Dammann O, et al.; ELGAN Study Investigators. Pregnancy disorders that lead to delivery before the 28th week of gestation: an epidemiologic approach to classification. Am J Epidemiol 2008;168:980–9.
McElrath TF, Fichorova RN, Allred EN, et al.; ELGAN Study Investigators. Blood protein profiles of infants born before 28 weeks differ by pregnancy complication. Am J Obstet Gynecol 2011;204:418.e1–418.e12.
Hecht JL, Fichorova RN, Tang VF, Allred EN, McElrath TF, Leviton A ; Elgan Study Investigators. Relationship Between Neonatal Blood Protein Concentrations and Placenta Histologic Characteristics in Extremely Low GA Newborns. Pediatr Res 2011;69:68–73.
Fichorova RN, Onderdonk AB, Yamamoto H, et al.; Extremely Low Gestation Age Newborns (ELGAN) Study Investigators. Maternal microbe-specific modulation of inflammatory response in extremely low-gestational-age newborns. MBio 2011;2:e00280–10.
Leviton A, O’Shea TM, Bednarek FJ, Allred EN, Fichorova RN, Dammann O ; ELGAN Study Investigators. Systemic responses of preterm newborns with presumed or documented bacteraemia. Acta Paediatr 2012;101:355–9.
Martin CR, Bellomy M, Allred EN, Fichorova RN, Leviton A . Systemic inflammation associated with severe intestinal injury in extremely low gestational age newborns. Fetal Pediatr Pathol 2013;32:222–34.
Bose CL, Laughon MM, Allred EN, et al.; ELGAN Study Investigators. Systemic inflammation associated with mechanical ventilation among extremely preterm infants. Cytokine 2013;61:315–22.
Beloosesky R, Ginsberg Y, Khatib N, et al. Prophylactic maternal N-acetylcysteine in rats prevents maternal inflammation-induced offspring cerebral injury shown on magnetic resonance imaging. Am J Obstet Gynecol 2013;208:213.e1–6.
Gitto E, Reiter RJ, Sabatino G, et al. Correlation among cytokines, bronchopulmonary dysplasia and modality of ventilation in preterm newborns: improvement with melatonin treatment. J Pineal Res 2005;39:287–93.
Leonard CH, Clyman RI, Piecuch RE, Juster RP, Ballard RA, Behle MB . Effect of medical and social risk factors on outcome of prematurity and very low birth weight. J Pediatr 1990;116:620–6.
Thompson RJ Jr, Goldstein RF, Oehler JM, Gustafson KE, Catlett AT, Brazy JE . Developmental outcome of very low birth weight infants as a function of biological risk and psychosocial risk. J Dev Behav Pediatr 1994;15:232–8.
Anselmi L, Barros FC, Teodoro ML, et al. Continuity of behavioral and emotional problems from pre-school years to pre-adolescence in a developing country. J Child Psychol Psychiatry 2008;49:499–507.
O’Shea TM, Allred EN, Dammann O, et al.; ELGAN study Investigators. The ELGAN study of the brain and related disorders in extremely low gestational age newborns. Early Hum Dev 2009;85:719–25.
Kuban K, Adler I, Allred EN, et al. Observer variability assessing US scans of the preterm brain: the ELGAN study. Pediatr Radiol 2007;37:1201–8.
Fichorova RN, Richardson-Harman N, Alfano M, et al. Biological and technical variables affecting immunoassay recovery of cytokines from human serum and simulated vaginal fluid: a multicenter study. Anal Chem 2008;80:4741–51.
Fichorova RN, Trifonova RT, Gilbert RO, et al. Trichomonas vaginalis lipophosphoglycan triggers a selective upregulation of cytokines by human female reproductive tract epithelial cells. Infect Immun 2006;74:5773–9.
Achenbach TM, Rescorla LA . 2000 Manual for the ASEBA Preschool Forms and Profiles. Burlington, Vermont: University of Vermont, Research Center for Children, Youth, and Families, 2000.
Bayley N . Bayley Scales of Infant Development-Second Edition. San Antonio, TX: Psychological Corporation, 1993.
Roberts G, Anderson PJ, Doyle LW ; Victorian Infant Collaborative Study Group. The stability of the diagnosis of developmental disability between ages 2 and 8 in a geographic cohort of very preterm children born in 1997. Arch Dis Child 2010;95:786–90.
Sparrow SS, Balla DA, Cicchetti DV . Vineland Adaptive Behavioral Scales. Circle Pines, MN: American Guidance Service, 1984.
Acknowledgements
The authors gratefully acknowledge the contributions of their subjects, and their subjects’ families, as well as those of their colleagues. The participating institutions and ELGAN Study collaborators who made this report possible are: Children’s Hospital, Boston, Massachusetts: Kathleen Lee, Anne McGovern, Jill Gambardella, Susan Ursprung, Ruth Blomquist Kristen Ecklund, Haim Bassan, Samantha Butler, Adré Duplessis, Cecil Hahn, Catherine Limperopoulos, Omar Khwaja, Janet S. Soul; Baystate Medical Center, Springfield, Massachusetts: Karen Christianson, Frederick Hampf, Herbert Gilmore, Susan McQuiston; Beth Israel Deaconess Medical Center, Boston, Massachusetts: Camilia R. Martin, Colleen Hallisey, Caitlin Hurley, Miren Creixell, Jane Share; Brigham & Women’s Hospital, Boston, Massachusetts: Linda J. Van Marter, Sara Durfee; Massachusetts General Hospital, Boston, Massachusetts: Robert M. Insoft, Jennifer G. Wilson, Maureen Pimental, Sjirk J. Westra, Kalpathy Krishnamoorthy; Floating Hospital for Children at Tufts Medical Center, Boston, Massachusetts: Cynthia Cole, John M. Fiascone, Janet Madden, Ellen Nylen, Anne Furey, Roy McCauley, Paige T. Church, Cecelia Keller, Karen J. Miller; U Mass Memorial Health Care, Worcester, Massachusetts: Francis Bednarek (deceased), Mary Naples, Beth Powers, Jacqueline Wellman, Robin Adair, Richard Bream, Alice Miller, Albert Scheiner, Christy Stine; Yale University School of Medicine, New Haven, Connecticut: Richard Ehrenkranz, Joanne Williams, Elaine Romano; Wake Forest University Baptist Medical Center and Forsyth Medical Center, Winston-Salem, North Carolina: T. Michael O’Shea, Debbie Gordon, Teresa Harold, Barbara Specter, Deborah Allred, Robert Dillard, Don Goldstein, Deborah Hiatt (deceased), Gail Hounshell, Ellen Waldrep, Lisa Washburn, Cherrie D. Welch; University Health Systems of Eastern Carolina, Greenville, North Carolina: Stephen C. Engelke, Sherry Moseley, Linda Pare, Donna Smart, Joan Wilson, Ira Adler, Sharon Buckwald, Rebecca Helms, Kathyrn Kerkering, Scott S. MacGilvray, Peter Resnik; North Carolina Children’s Hospital, Chapel Hill, North Carolina: Carl Bose, Gennie Bose, Lynn A. Fordham, Lisa Bostic, Diane Marshall, Kristi Milowic, Janice Wereszczak; Helen DeVos Children’s Hospital, Grand Rapids, Michigan: Mariel Poortenga, Dinah Sutton, Bradford W. Betz, Steven L. Bezinque, Joseph Junewick, Wendy Burdo-Hartman, Lynn Fagerman, Kim Lohr, Steve Pastyrnak; Sparrow Hospital, Lansing, Michigan: Carolyn Solomon, Ellen Cavenagh, Victoria J. Caine, Nicholas Olomu, Joan Price; Michigan State University, East Lansing, Michgan: Nigel Paneth, Padmani Karna, Madeleine Lenski; University of Chicago Medical Center, Chicago, Illinois: Michael D. Schreiber, Grace Yoon, Kate Feinstein, Leslie Caldarelli, Sunila E. O’Connor, Michael Msall, Susan Plesha-Troyke; William Beaumont Hospital, Royal Oak, Michigan: Daniel Batton, Beth Kring, Karen Brooklier, Melisa J. Oca, Katherine M. Solomon; Arkansas Children’s Hospital, Little Rock, Arkansas: Joanna J. Seibert; Children’s Healthcare of Atlanta: Robert Lorenzo.
Author information
Authors and Affiliations
Consortia
Corresponding author
Supplementary information
Supplementary Table S1 (download DOC )
(DOC 43 kb)
Supplementary Table S2 (download DOC )
(DOC 41 kb)
Supplementary Table S3 (download DOC )
(DOC 57 kb)
Rights and permissions
About this article
Cite this article
O’Shea, T., Joseph, R., Kuban, K. et al. Elevated blood levels of inflammation-related proteins are associated with an attention problem at age 24 mo in extremely preterm infants. Pediatr Res 75, 781–787 (2014). https://doi.org/10.1038/pr.2014.41
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/pr.2014.41
This article is cited by
-
Environmental influences on child health outcomes: cohorts of individuals born very preterm
Pediatric Research (2023)
-
Cortisol and inflammatory biomarker levels in youths with attention deficit hyperactivity disorder (ADHD): evidence from a systematic review with meta-analysis
Translational Psychiatry (2021)
-
COVID-19 threatens maternal mental health and infant development: possible paths from stress and isolation to adverse outcomes and a call for research and practice
Child Psychiatry & Human Development (2021)
-
In-line filtration in very preterm neonates: a randomized controlled trial
Scientific Reports (2020)
-
Early Motor Differences in Infants at Elevated Likelihood of Autism Spectrum Disorder and/or Attention Deficit Hyperactivity Disorder
Journal of Autism and Developmental Disorders (2020)