Abstract
Background:
The aim of this clinical study was to determine the prevalence of SHOX haploinsufficiency in a population of short stature patients and describe their anthropometric measurements.
Methods:
574 short statured patients were evaluated in a single center (1992–2015). SHOX copy number was detected by quantitative polymerase chain reaction (qPCR) in 574 subjects, followed by multiplex ligation-dependent probe amplification (MLPA) and DNA sequencing in subjects with SHOX haploinsufficiency. We evaluated anthropometric measurements at birth, and at first examination. Skeletal abnormalities were recorded for patients with SHOX haploinsufficiency.
Results:
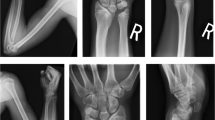
Thirty-two patients were excluded due to Turner syndrome (n = 28), SRY-positive 46,XX male karyotype (n = 1), or lacked clinical follow-up information (n = 3). The prevalence of SHOX haploinsufficiency was 9 out of 542 (1.7%). The nine children had decreased height −2.85 (0.6) SD scores (SDS) (mean (SD)) and weight −2.15 (1.36) SDS, P < 0.001 and P = 0.001, respectively. The sitting height/height ratio was increased, P = 0.04. Madelung deformity was diagnosed in three patients. Mean height was −2.9 (0.4) SDS at baseline and increased by 0.25 (0.2) SDS, P = 0.046, after 1 y of growth hormone (GH) treatment.
Conclusion:
The prevalence of SHOX haploinsufficiency was 1.7%. The clinical findings indicating SHOX haploinsufficiency among the nine children were disproportionate short stature and forearm anomalies.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Cohen P, Rogol AD, Deal CL, et al.; 2007 ISS Consensus Workshop participants. Consensus statement on the diagnosis and treatment of children with idiopathic short stature: a summary of the Growth Hormone Research Society, the Lawson Wilkins Pediatric Endocrine Society, and the European Society for Paediatric Endocrinology Workshop. J Clin Endocrinol Metab 2008;93:4210–7.
Chia DJ. Minireview: mechanisms of growth hormone-mediated gene regulation. Mol Endocrinol 2014;28:1012–25.
Decker E, Durand C, Bender S, et al. FGFR3 is a target of the homeobox transcription factor SHOX in limb development. Hum Mol Genet 2011;20:1524–35.
Rao E, Weiss B, Fukami M, et al. Pseudoautosomal deletions encompassing a novel homeobox gene cause growth failure in idiopathic short stature and Turner syndrome. Nat Genet 1997;16:54–63.
Rappold GA, Fukami M, Niesler B, et al. Deletions of the homeobox gene SHOX (short stature homeobox) are an important cause of growth failure in children with short stature. J Clin Endocrinol Metab 2002;87:1402–6.
Stuppia L, Calabrese G, Gatta V, et al. SHOX mutations detected by FISH and direct sequencing in patients with short stature. J Med Genet 2003;40:E11.
Rappold G, Blum WF, Shavrikova EP, et al. Genotypes and phenotypes in children with short stature: clinical indicators of SHOX haploinsufficiency. J Med Genet 2007;44:306–13.
Huber C, Rosilio M, Munnich A, Cormier-Daire V ; French SHOX GeNeSIS Module. High incidence of SHOX anomalies in individuals with short stature. J Med Genet 2006;43:735–9.
Rosilio M, Huber-Lequesne C, Sapin H, Carel JC, Blum WF, Cormier-Daire V. Genotypes and phenotypes of children with SHOX deficiency in France. J Clin Endocrinol Metab 2012;97:E1257–65.
Binder G, Ranke MB, Martin DD. Auxology is a valuable instrument for the clinical diagnosis of SHOX haploinsufficiency in school-age children with unexplained short stature. J Clin Endocrinol Metab 2003;88:4891–6.
Binder G, Schwarze CP, Ranke MB. Identification of short stature caused by SHOX defects and therapeutic effect of recombinant human growth hormone. J Clin Endocrinol Metab 2000;85:245–9.
Morizio E, Stuppia L, Gatta V, et al. Deletion of the SHOX gene in patients with short stature of unknown cause. Am J Med Genet A 2003;119A:293–6.
Jorge AA, Souza SC, Nishi MY, et al. SHOX mutations in idiopathic short stature and Leri-Weill dyschondrosteosis: frequency and phenotypic variability. Clin Endocrinol (Oxf) 2007;66:130–5.
Caliebe J, Broekman S, Boogaard M, et al. IGF1, IGF1R and SHOX mutation analysis in short children born small for gestational age and short children with normal birth size (idiopathic short stature). Horm Res Paediatr 2012;77:250–60.
Hirschfeldova K, Solc R, Baxova A, et al. SHOX gene defects and selected dysmorphic signs in patients of idiopathic short stature and Léri-Weill dyschondrosteosis. Gene 2012;491:123–7.
Sandoval GT, Jaimes GC, Barrios MC, Cespedes C, Velasco HM. SHOX gene and conserved noncoding element deletions/duplications in Colombian patients with idiopathic short stature. Mol Genet Genomic Med 2014;2:95–102.
Ellison JW, Wardak Z, Young MF, Gehron Robey P, Laig-Webster M, Chiong W. PHOG, a candidate gene for involvement in the short stature of Turner syndrome. Hum Mol Genet 1997;6:1341–7.
Marchini A, Marttila T, Winter A, et al. The short stature homeodomain protein SHOX induces cellular growth arrest and apoptosis and is expressed in human growth plate chondrocytes. J Biol Chem 2004;279:37103–14.
Munns CJ, Haase HR, Crowther LM, et al. Expression of SHOX in human fetal and childhood growth plate. J Clin Endocrinol Metab 2004;89:4130–5.
Binder G, Renz A, Martinez A, et al. SHOX haploinsufficiency and Leri-Weill dyschondrosteosis: prevalence and growth failure in relation to mutation, sex, and degree of wrist deformity. J Clin Endocrinol Metab 2004;89:4403–8.
Zinn AR, Wei F, Zhang L, et al. Complete SHOX deficiency causes Langer mesomelic dysplasia. Am J Med Genet 2002;110:158–63.
Clement-Jones M, Schiller S, Rao E, et al. The short stature homeobox gene SHOX is involved in skeletal abnormalities in Turner syndrome. Hum Mol Genet 2000;9:695–702.
Hertel NT, Scheike T, Juul A, et al. [Body proportions of Danish children. Curves for sitting height ratio, subischial length and arm span]. Ugeskr Laeger 1995;157:6876–81.
Niklasson A, Albertsson-Wikland K. Continuous growth reference from 24th week of gestation to 24 months by gender. BMC Pediatr 2008;8:8.
Tinggaard J, Aksglaede L, Sørensen K, et al. The 2014 Danish references from birth to 20 years for height, weight and body mass index. Acta Paediatr 2014;103:214–24.
Marshall WA, Tanner JM. Variations in the pattern of pubertal changes in boys. Arch Dis Child 1970;45:13–23.
Marshall WA, Tanner JM. Variations in pattern of pubertal changes in girls. Arch Dis Child 1969;44:291–303.
Thodberg HH, Kreiborg S, Juul A, Pedersen KD. The BoneXpert method for automated determination of skeletal maturity. IEEE Trans Med Imaging 2009;28:52–66.
Martin DD, Heckmann C, Jenni OG, Ranke MB, Binder G, Thodberg HH. Metacarpal thickness, width, length and medullary diameter in children–reference curves from the First Zürich Longitudinal Study. Osteoporos Int 2011;22:1525–36.
Ottesen AM, Garn ID, Aksglaede L, Juul A, Rajpert-De Meyts E. A simple screening method for detection of Klinefelter syndrome and other X-chromosome aneuploidies based on copy number of the androgen receptor gene. Mol Hum Reprod 2007;13:745–50.
Gatta V, Palka C, Chiavaroli V, et al. Spectrum of phenotypic anomalies in four families with deletion of the SHOX enhancer region. BMC Med Genet 2014;15:87.
Juul A, Bang P, Hertel NT, et al. Serum insulin-like growth factor-I in 1030 healthy children, adolescents, and adults: relation to age, sex, stage of puberty, testicular size, and body mass index. J Clin Endocrinol Metab 1994;78:744–52.
Jensen RB, Thankamony A, O’Connell SM, et al. A randomised controlled trial evaluating IGF1 titration in contrast to current GH dosing strategies in children born small for gestational age: the North European Small-for-Gestational-Age Study. Eur J Endocrinol 2014;171:509–18.
Sørensen K, Aksglaede L, Petersen JH, Andersson AM, Juul A. Serum IGF1 and insulin levels in girls with normal and precocious puberty. Eur J Endocrinol 2012;166:903–10.
Salmon-Musial AS, Rosilio M, David M, et al. Clinical and radiological characteristics of 22 children with SHOX anomalies and familial short stature suggestive of Léri-Weill Dyschondrosteosis. Horm Res Paediatr 2011;76:178–85.
Blum WF, Crowe BJ, Quigley CA, et al.; SHOX Study Group. Growth hormone is effective in treatment of short stature associated with short stature homeobox-containing gene deficiency: Two-year results of a randomized, controlled, multicenter trial. J Clin Endocrinol Metab 2007;92:219–28.
Ogata T, Onigata K, Hotsubo T, Matsuo N, Rappold G. Growth hormone and gonadotropin-releasing hormone analog therapy in haploinsufficiency of SHOX. Endocr J 2001;48:317–22.
Scalco RC, Melo SS, Pugliese-Pires PN, et al. Effectiveness of the combined recombinant human growth hormone and gonadotropin-releasing hormone analog therapy in pubertal patients with short stature due to SHOX deficiency. J Clin Endocrinol Metab 2010;95:328–32.
Fukami M, Nishi Y, Hasegawa Y, et al. Statural growth in 31 Japanese patients with SHOX haploinsufficiency: support for a disadvantageous effect of gonadal estrogens. Endocr J 2004;51:197–200.
Donze SH, Meijer CR, Kant SG, et al. The growth response to GH treatment is greater in patients with SHOX enhancer deletions compared to SHOX defects. Eur J Endocrinol 2015;173:611–21.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marstrand-Joergensen, M., Jensen, R., Aksglaede, L. et al. Prevalence of SHOX haploinsufficiency among short statured children. Pediatr Res 81, 335–341 (2017). https://doi.org/10.1038/pr.2016.233
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/pr.2016.233
This article is cited by
-
SHOX haploinsufficiency presenting with isolated short long bones in the second and third trimester
European Journal of Human Genetics (2018)