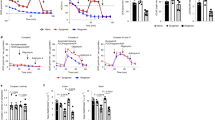
Abstract
Background
The intracellular redox potential of the glutathione (GSH)/glutathione disulfide (GSSG) couple regulates cellular processes. In vitro studies indicate that a reduced GSH/GSSG redox potential favors proliferation, whereas a more oxidized redox potential favors differentiation. Intestinal growth depends upon an appropriate balance between the two. However, how the ontogeny of intestinal epithelial cellular (IEC) GSH/GSSG redox regulates these processes in the developing intestine has not been fully characterized in vivo.
Methods
Ontogeny of intestinal GSH redox potential and growth were measured in neonatal mice.
Results
We show that IEC GSH/GSSG redox potential becomes increasingly reduced (primarily driven by increased GSH concentration) over the first 3 weeks of life. Increased intracellular GSH has been shown to drive proliferation through increased poly-ADP-ribose polymerase (PARP) activity. We show that increasing IEC poly-ADP-ribose chains can be measured over the first 3 weeks of life, indicating an increase in IEC PARP activity. These changes are accompanied by increased intestinal growth and IEC proliferation as assessed by villus height/crypt depth, intestinal length, and Ki67 staining.
Conclusion
Understanding how IEC GSH/GSSG redox potential is developmentally regulated may provide insight into how premature human intestinal redox states can be manipulated to optimize intestinal growth and adaptation.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Marseglia L, D'Angelo G, Manti S et al, Oxidative stress-mediated aging during the fetal and perinatal periods. Oxid Med Cell Longev 2014;2014:358375.
Brion LP, Bell EF, Raghuveer TS . Vitamin E supplementation for prevention of morbidity and mortality in preterm infants. Cochrane Database Syst Rev 2003;3:CD003665.
Darlow BA, Austin NC . Selenium supplementation to prevent short-term morbidity in preterm neonates. Cochrane Database Syst Rev 2003;4:CD003312.
Suresh GK, Davis JM, Soll RF . Superoxide dismutase for preventing chronic lung disease in mechanically ventilated preterm infants. Cochrane Database Syst Rev 2001;1:CD001968.
Veal EA, Day AM, Morgan BA . Hydrogen peroxide sensing and signaling. Mol Cell 2007;26:1–14.
Covarrubias L, Hernandez-Garcia D, Schnabel D, Salas-Vidal E, Castro-Obregon S . Function of reactive oxygen species during animal development: passive or active? Dev Biol 2008;320:1–11.
Das KC . Redox control of premature birth and newborn biology. Antioxid Redox Signal 2004;6:105–7.
Lavoie JC, Chessex P . Development of glutathione synthesis and gamma-glutamyltranspeptidase activities in tissues from newborn infants. Free Radic Biol Med 1998;24:994–1001.
Hernandez-Garcia D, Wood CD, Castro-Obregon S, Covarrubias L . Reactive oxygen species: a radical role in development? Free Radic Biol Med 2010;49:130–43.
Hutter DE, Till BG, Greene JJ . Redox state changes in density-dependent regulation of proliferation. Exp Cell Res 1997;232:435–8.
Imhoff BR, Hansen JM . Differential redox potential profiles during adipogenesis and osteogenesis. Cell Mol Biol Lett 2011;16:149–61.
Nkabyo YS, Ziegler TR, Gu LH, Watson WH, Jones DP . Glutathione and thioredoxin redox during differentiation in human colon epithelial (Caco-2) cells. Am J Physiol Gastrointest Liver Physiol 2002;283:G1352–9.
Touloukian RJ, Smith GJ . Normal intestinal length in preterm infants. J Pediatr Surg 1983;18:720–3.
Kelly N, Friend K, Boyle P et al, The role of the glutathione antioxidant system in gut barrier failure in a rodent model of experimental necrotizing enterocolitis. Surgery 2004;136:557–66.
Pellny TK, Locato V, Vivancos PD et al, Pyridine nucleotide cycling and control of intracellular redox state in relation to poly (ADP-ribose) polymerase activity and nuclear localization of glutathione during exponential growth of Arabidopsis cells in culture. Mol Plant 2009;2:442–56.
Jones DP . Redox potential of GSH/GSSG couple: assay and biological significance. Methods Enzymol 2002;348:93–112.
Lin PW, Myers LE, Ray L et al, Lactobacillus rhamnosus blocks inflammatory signaling in vivo via reactive oxygen species generation. Free Radic Biol Med 2009;47:1205–111.
Hansen JM, Harris C . Glutathione during embryonic development. Biochim Biophys Acta 2014;1850:1527–42.
Ungvari Z, Bailey-Downs L, Gautam T et al, Adaptive induction of NF-E2-related factor-2-driven antioxidant genes in endothelial cells in response to hyperglycemia. Am J Physiol Heart Circ Physiol 2011;300:H1133–40.
Neu J, Walker WA . Necrotizing enterocolitis. N Engl J Med 2011;364:255–64.
Lin PW, Stoll BJ . Necrotising enterocolitis. Lancet 2006;368:1271–83.
Grave GD, Nelson SA, Walker WA et al, New therapies and preventive approaches for necrotizing enterocolitis: report of a research planning workshop. Pediatr Res 2007;62:510–14.
McCracken VJ, Lorenz RG . The gastrointestinal ecosystem: a precarious alliance among epithelium, immunity and microbiota. Cell Microbiol 2001;3:1–11.
Lee JW, Davis JM . Future applications of antioxidants in premature infants. Curr Opin Pediatr 2011;23:161–6.
Qanungo S, Mukherjea M . Ontogenic profile of some antioxidants and lipid peroxidation in human placental and fetal tissues. Mol Cell Biochem 2000;215:11–9.
Perrone S, Tataranno ML, Negro S et al, Early identification of the risk for free radical-related diseases in preterm newborns. Early Hum Dev 2010;86:241–4.
Schafer FQ, Buettner GR . Redox environment of the cell as viewed through the redox state of the glutathione disulfide/glutathione couple. Free Radic Biol Med 2001;30:1191–212.
Denning TL, Takaishi H, Crowe SE, Boldogh I, Jevnikar A, Ernst PB . Oxidative stress induces the expression of Fas and Fas ligand and apoptosis in murine intestinal epithelial cells. Free Radic Biol Med 2002;33:1641–50.
Li N, Ma L, Liu X et al, Arginyl-glutamine dipeptide or docosahexaenoic acid attenuates hyperoxia-induced small intestinal injury in neonatalmice. J Pediatr Gastroenterol Nutr 2012;54:499–504.
Ford H, Watkins S, Reblock K, Rowe M . The role of inflammatory cytokines and nitric oxide in the pathogenesis of necrotizing enterocolitis. J Pediatr Surg 1997;32:275–82.
Santulli TV, Schullinger JN, Heird WC et al, Acute necrotizing enterocolitis in infancy: a review of 64 cases. Pediatrics 1975;55:376–87.
Jilling T, Lu J, Jackson M, Caplan MS . Intestinal epithelial apoptosis initiates gross bowel necrosis in an experimental rat model of neonatal necrotizing enterocolitis. Pediatr Res 2004;55:622–9.
Bell RS, Graham CB, Stevenson JK . Roentgenologic and clinical manifestations of neonatal necrotizing enterocolitis. Experience with 43 cases. Am J Roentgenol Radium Ther Nucl Med 1971;112:123–34.
Gordon PV . Understanding intestinal vulnerability to perforation in the extremely low birth weight infant. Pediatr Res 2009;65:138–44.
Tatekawa Y, Muraji T, Imai Y, Nishijima E, Tsugawa C . The mechanism of focal intestinal perforations in neonates with low birth weight. Pediatr Surg Int 1999;15:549–552.
Martensson J, Jain A, Meister A . Glutathione is required for intestinal function. Proc Natl Acad Sci USA 1990;87:1715–9.
Hagen TM, Wierzbicka GT, Bowman BB, Aw TY, Jones DP . Fate of dietary glutathione: disposition in the gastrointestinal tract. Am J Physiol 1990;259:G530–5.
Aw TY, Wierzbicka G, Jones DP . Oral glutathione increases tissue glutathione in vivo. Chem Biol Interact 1991;80:89–97.
Nkabyo YS, Gu LH, Jones DP, Ziegler TR . Thiol/disulfide redox status is oxidized in plasma and small intestinal and colonic mucosa of rats with inadequate sulfur amino acid intake. J Nutr 2006;136:1242–8.
Shyntum Y, Iyer SS, Tian J et al, Dietary sulfur amino acid supplementation reduces small bowel thiol/disulfide redox state and stimulates ileal mucosal growth after massive small bowel resection in rats. J Nutr 2009;139:2272–8.
Jonas CR, Gu LH, Nkabyo YS et al, Glutamine and KGF each regulate extracellular thiol/disulfide redox and enhance proliferation in Caco-2 cells. Am J Physiol Regul Integr Comp Physiol 2003;285:R1421–9.
Acknowledgements
We would like to acknowledge Loren S. Myers for her technical support toward completion of the studies reported.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Reid, G., Berardinelli, A., Ray, L. et al. Timing of developmental reduction in epithelial glutathione redox potential is associated with increased epithelial proliferation in the immature murine intestine. Pediatr Res 82, 362–369 (2017). https://doi.org/10.1038/pr.2017.49
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/pr.2017.49