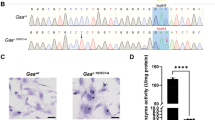
Abstract
A newborn screening program for Pompe disease using dried blood spots (DBSs) was initiated in Japan. Here, we summarized this screening program and described the results of the GAA gene analysis. From April 2013 to November 2016, 103,204 newborns were screened; 71 had low acid alpha-glucosidase (AαGlu) activity. GAA sequencing showed that 32 (45.1%) and 37 (52.1%) of these newborns were homozygous and heterozygous for pseudodeficiency alleles c.[1726G>A; 2965G>A], respectively. Moreover, 24 of 32 newborns with homozygous c.[1726G>A; 2965G>A] alleles had no mutations, and the other eight had one mutation each. Thirty-five of 37 newborns with heterozygous c.[1726G>A; 2965G>A] alleles had one mutation, and the other two had two mutations each. Only one newborn who had two mutations did not harbor c.[1726G>A; 2965G>A] alleles. Thus, it was difficult to distinguish newborns with c.[1726G>A; 2965G>A] alleles from newborns with pre-symptomatic Pompe disease using AαGlu assays in DBSs or fibroblasts; GAA gene sequencing was necessary. Seventy-one newborns had 50 variants, including 21 mutations or predictably pathogenic variants, and 29 polymorphisms or predictably non-pathogenic variants. Four of 21 mutations or predictably pathogenic variants and four of 29 polymorphisms or predictably non-pathogenic variants were novel. No infantile-onset Pompe disease was detected, and three newborns were diagnosed with potential late-onset Pompe disease. In the literature, 156 variants have been reported for 296 patients from 277 families in 41 articles from Japan, Korea, Taiwan, and China. Our results provide insights into GAA gene mutation profiles and the relationship between GAA and Pompe disease in Asian populations.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Martiniuk F, Mehler M, Pellicer A, Tzall S, La Badie G, Hobart C, et al. Isolation of a cDNA for human acid α-glucosidase and detection of genetic heterogeneity for mRNA in three α-glucosidase deficient patients. Proc Natl Acad Sci USA. 1986;83:9641–4.
Hagemans MLC, Winkel LPF, Hop WCJ, Reuser AJJ, Van Doorn PA, Van der Ploeg AT. Disease severity in children and adults with Pompe disease related to age and disease duration. Neurology. 2005;64:2139–41.
Kishnani PS, Corzo D, Nicolino M, Byrne B, Mandel H, Hwu WL, et al. Recombinant human acid alpha-glucosidase: major clinical benefits in infantile-onset Pompe disease. Neurology. 2007;68:99–109.
Van der Ploeg AT, Clemens PR, Corzo D, Escolar DM, Florence J, Groeneveld G, et al. A randomized study of alglucosidase alfa in late-onset Pompe's disease. N Engl J Med. 2010;15:1396–406.
Kishnani PS, Corzo D, Leslie ND, Gruskin D, Van der Ploeg A, Clancy JP, et al. Early treatment with alglucosidase alpha prolongs long-term survival of infants with Pompe disease. Pedia Res. 2009;66:329–35.
Prater SN, Banugaria SG, DeArmey SM, Botha EG, Stege EM, Case LE, et al. The emerging phenotype of long-term survivors with infantile Pompe disease. Genet Med. 2012;14:800–10.
Chien Y-H, Lee N-C, Thurberg BL, Chiang S-C, Zhang XK, Keutzer J, et al. Pompe Disease in Infants: improving the prognosis by newborn screening and early treatment. Pediatrics. 2009;124:e1116.
Yang C-F, Liu H-C, Hsu T-R, Tsai F-C, Chiang S-F, Chiang C-C, et al. A large-scale nationwide newborn screening program for Pompe disease in Taiwan: towards effective diagnosis and treatment. Am J Med Genet A. 2014;164A:54–61.
Kroos MA, Mullaart RA, Van Vliet L, Pomponio RJ, Amartino H, Kolodny EH, et al. p.[G576S; E689K]: pathogenic combination or polymorphism in Pompe disease? Eur J Hum Genet. 2008;16:875–9.
Kumamoto S, Katafuchi T, Nakamura K, Endo F, Oda E, Okuyama T, et al. High frequency of acid α-glucosidase pseudodeficiency complicates newborn screening for glycogen storage disease type II in the Japanese population. Mol Genet Metab. 2009;97:190–5.
Labrousse P, Chien Y-H, Pomponio RJ, Keutzer J, Lee N-C, Akmaev VR, et al. Genetic heterozygosity and pseudodeficiency in the Pompe disease newborn screening pilot program. Mol Genet Metab. 2010;99:379–83.
Shigeto S, Katafuchi T, Okuda Y, Nakamura K, Endo F, Okuyama T, et al. Improved assay for differential diagnosis between Pompe disease and acid α-glucosidase pseudodeficiency on dried blood spots. Mol Genet Metab. 2011;103:12–7.
Kishnani PS, Steiner RD, Bali D, Berger K, Byrne BJ, Case L, et al. Pompe disease diagnosis and management guideline. Genet Med. 2006;8:267–88.
Yoshida S. Genetic testing of lysosomal storage diseases by next-generation sequencer. Sci Rep Chemo-Sero-Ther Res Inst. 2015;24:14–31. https://www.kaketsuken.org/pdf/003-reimei-24.pdf
Tsujino S, Huie M, Kanazawa N, Sugie H, Goto Y, Kawai M, et al. Frequent mutations in Japanese patients with acid maltase deficiency. Neuromuscul Disord. 2000;10:599–603.
Landrum MJ, Lee JM, Benson M, Brown G, Chao C, Chitipiralla S, et al. ClinVar: public archive of interpretations of clinically relevant variants. Nucleic Acids Res. 2016;44:D862–D868.
Adzhubei I, Jordan DM, Sunyaev SR. Predicting functional effect of human missense mutations using PolyPhen-2. Curr Protoc Hum Genet. 2013;76:7.20.1–41.
Desmet F-O, Hamroun D, Lalande M, Collod-Beroud G, Claustres M, Beroud C. Human splicing finder: an online bioinformatics tool to predict splicing signals. Nucleic Acids Res. 2009;37:e67.
Schwarz JM, Cooper DN, Schuelke M, Seelow D. MutationTaster2: mutation prediction for the deep-sequencing age. Nat Methods. 2014;11:361–2.
Hijikata A, Raju R, Keerthikumar S, et al. Mutation@A Glance: an integrative Web application for analyzing mutations from human genetic diseases. DNA Res. 2010;17:197–208.
Qiu J-J, Wei M, Zhang W-M, Shi H-P. Clinical and molecular genetic study on two patients of the juvenile form of Pompe disease in China. Chin J Pedia. 2007;45:760–4.
Eto Y ed. Pompe disease (glycogen storage disease type II) diagnosis and management guideline for Japanese (revised edition). (2013) (En Medix Co. Ltd, Tokyo, Japan).
Tarnopolsky M, Katzberg H, Petrof BJ, Sirrs S, Sarnat HB, Myers K, et al. Pompe disease: diagnosis and management. Evidence-based guidelines from a Canadian expert panel. Can J Neurol Sci. 2016;43:472–85.
Bodamer OA, Scott R, Giugliani R. Newborn screening for Pompe disease. Pediatrics. 2017;140:S4–13.
Ausems MGEM, Verbiest J, Hermans MMP, Kroos MA, Beemer FA, Wokke JHJ, et al. Frequency of glycogen storage disease type II in the Netherlands: implications for diagnosis and genetic counseling. Eur J Hum Genet. 1999;7:713–6.
Becker JA, Vlach J, Raben N, Nagaraju K, Adams EM, Hermans MM, et al. The African origin of the common mutation in African American patients with glycogen-storage disease type II. Am J Hum Genet. 1998;62:991–4.
Huie ML, Chen AS, Tsujino S, Shanske S, DiMauro S, Engel AG, et al. Aberrant splicing in adult-onset glycogen storage disease type II (GSDII): molecular identification of an IVS1 (-13T->G) mutation in a majority of patients and a novel IVS10 (+1GT->CT) mutation. Hum Mol Genet. 1994;3:2231–6.
Tsuburaya RS, Monma K, Oya Y, Nakayama T, Fukuda T, Sugie H, et al. Acid phosphatase-positive globular inclusions is a good diagnostic marker for two patients with adult-onset Pompe disease lacking disease-specific pathology. Neuromuscul Disord. 2012;22:389–93.
Shieh J-J, Lin C-Y. Frequent mutation of Chinese patients with infantile type of GSD II in Taiwan: evidence for a founder effect. Hum Mutat. 1998;11:306–12.
Wan L, Lee C-C, Hsu C-M, Hwu W-L, Yang C-C, Tsai C-H, Tsai F-J. Identification of eight novel mutations of the acid α-glucosidase gene causing the infantile or juvenile form of glycogen storage disease type II. J Neurol. 2008;255:831–8.
Mashima R, Sakai E, Kosuga M, Okuyama T. Levels of enzyme activities in six lysosomal storage diseases in Japanese neonates determined by liquid chromatography-tandem mass spectrometry. Mol Genet Metab Rep. 2016;9:6–11.
Lin N, Huang J, Violante S, Orsini JJ, Caggana M, Hughes EE, et al. Liquid chromatography-tandem mass spectrometry assay of leukocyte acid α-glucosidase for post-newborn screening evaluation of Pompe disease. Clin Chem. 2017;63:842–51.
Acknowledgements
This study was supported in part by a Grant-in-Aid for Research on Rare and Intractable Diseases, Health and Labor Sciences Research; a Grant-in-Aid for Pediatric Research from the Ministry of Health, Labor and Welfare; and a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology. We are grateful to Ms. Fumiko Nozaki, Ms. Naomi Yano, Ms. Ayako Tateishi, Ms. Emi Harakawa, Ms. Yasuyo Sakamoto, and Ms. Matsumi Harada for their technical support in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Ethics Committee of Kumamoto University (approval no. 1537). Written informed consent was obtained from the parents or legal guardians of newborns.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Momosaki, K., Kido, J., Yoshida, S. et al. Newborn screening for Pompe disease in Japan: report and literature review of mutations in the GAA gene in Japanese and Asian patients. J Hum Genet 64, 741–755 (2019). https://doi.org/10.1038/s10038-019-0603-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s10038-019-0603-7
This article is cited by
-
Japanese experience of newborn screening for lysosomal storage diseases and adrenoleukodystrophy
Orphanet Journal of Rare Diseases (2025)
-
Effect of newborn genomic screening for lysosomal storage disorders: a cohort study in China
Genome Medicine (2025)
-
Late-onset Pompe’s disease in pediatrics: results from an Italian national survey on 38 patients and proposal of a targeted diagnostic algorithm
Orphanet Journal of Rare Diseases (2025)
-
Global carrier frequency and predicted genetic prevalence of patients with pathogenic sequence variants in autosomal recessive genetic neuromuscular diseases
Scientific Reports (2024)
-
Current status of newborn screening for Pompe disease in Japan
Orphanet Journal of Rare Diseases (2021)