Abstract
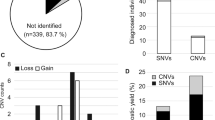
We aimed to study the diagnostic yield and clinical impact of trio exome sequencing (tES) in children with autism spectrum disorder (ASD). Participants (n = 137) between 2 and 18 years with syndromic and non-syndromic ASD underwent tES, after excluding karyotype-detectable cytogenetic abnormalities and fragile X syndrome. The diagnostic yield was 22/137 (16.1%) when considering only pathogenic (P) and likely-pathogenic (LP) variants in known disease-causing genes. We reported 23 significant (P, LP) variants in 22 individuals, with one participant (AGS041) harbouring a dual genetic diagnosis. Nearly half of these (12/23, 52.2%) were novel, while 21/23 (91.3%) occurred de novo. 20/23 (86.9%) of the variants were single nucleotide variants, while 3/23 (13.1%) were copy number variants. The diagnostic yield in syndromic ASD (14/40, 35%) was significantly higher than the non-syndromic group (8/97, 8.2%, p = 0.000258). Variants of uncertain significance in two participants were considered to be likely causative for the phenotype, given the strong clinical correlation (likely-causative variant of uncertain significance, LcVUS). On considering these two participants and an additional 28 participants with significant variants in autism candidate genes (vACG), the net diagnostic yield increased to 37.9%. The clinical benefits among those receiving a definite genetic diagnosis (P/LP variants only) included better prognostication (100%), availing reproductive counselling (100%), disease-specific surveillance (86.4%), and therapeutic implications (27.3%). Thus, in conclusion, in our cohort of 137 children with ASD, tES provided a definite genetic diagnosis in 16.1% of the participants, the yield being higher in syndromic ASD. A confirmed genetic diagnosis aided in holistic clinical care, extending beyond reproductive counselling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study is available on request from the corresponding author. The data is not publicly available due to privacy or ethical restrictions.
References
Lord C, Elsabbagh M, Baird G, Veenstra-Vanderweele J. Autism Spectrum Disorder. Nat Rev Dis Prim. 2020;6:5.
Lai MC, Lombardo MV, Baron-Cohen S. Autism. Lancet. 2014;383:896–910.
Gaugler T, Klei L, Sanders SJ, Bodea CA, Goldberg AP, Lee AB, et al. Most Genetic Risk For Autism Resides With Common Variation. Nat Genet. 2014;46:881–5.
Sandin S, Lichtenstein P, Kuja-Halkola R, Larsson H, Hultman CM, Reichenberg A. The Familial Risk Of Autism. JAMA. 2014;311:1770–7.
Sandin S, Lichtenstein P, Kuja-Halkola R, Hultman CM, Larsson H, Reichenberg A. The Heritability of Autism Spectrum Disorder. JAMA. 2017;318:1182–4.
Chattopadhyay N. Autism Screening in India: Many a Chasm to Bridge. Indian Pediatr. 2024;61:321–2.
Nakatsuka N, Moorjani P, Rai N, Sarkar B, Tandon A, Patterson N, et al. The Promise of Discovering Population-Specific Disease-Associated Genes in South Asia. Nat Genet. 2017;49:1403–7.
Sheth F, Shah J, Jain D, Shah S, Patel H, Patel K, et al. Comparative Yield of Molecular Diagnostic Algorithms For Autism Spectrum Disorder Diagnosis in India: Evidence Supporting Whole Exome Sequencing As First-Tier Test. BMC Neurol. 2023;23:292.
Arora K, Balwani M, Wakchaure A, Passi GR. Yield of Genetic Testing in Children With Autism Spectrum Disorder: A Single-Center Experience. Ann Indian Acad Neurol. 2025;28:253–6.
Manickam K, McClain MR, Demmer LA, Biswas S, Kearney HM, Malinowski J, et al. ACMG Board of Directors. Exome and genome sequencing for pediatric patients with congenital anomalies or intellectual disability: An evidence-based clinical guideline of the American College of Medical Genetics and Genomics (ACMG). Genet Med. 2021;23:2029–37.
Arteche-López A, Gómez Rodríguez MJ, Sánchez Calvin MT, Quesada-Espinosa JF, Lezana Rosales JM, Palma Milla C, et al. Towards a Change In The Diagnostic Algorithm Of Autism Spectrum Disorders: Evidence Supporting Whole Exome Sequencing as a First-Tier Test. Genes (Basel). 2021;12:560.
Schaefer GB, Mendelsohn NJ, Professional P, Guidelines C. Clinical Genetics Evaluation In Identifying The Etiology Of Autism Spectrum Disorders: 2013 Guideline Revisions. Genet Med. 2013;15:399–407.
Hellquist A, Tammimies K. Access, Utilization, And Awareness For Clinical Genetic Testing In Autism Spectrum Disorder in Sweden: A survey study. Autism. 2022;26:1795–804.
Smith HS, Franciskovich R, Lewis AM, Gerard A, Littlejohn RO, Nugent K, et al. Outcomes Of Prior Authorization Requests For Genetic Testing In Outpatient Pediatric Genetics Clinics. Genet Med. 2021;23:950–5.
Stafford CF, Sanchez-Lara PA. Impact Of Genetic And Genomic Testing On The Clinical Management Of Patients With Autism Spectrum Disorder. Genes (Basel). 2022;13:585.
Chen J, Bardes EE, Aronow BJ, Jegga AG. ToppGene Suite for Gene List Enrichment Analysis And Candidate Gene Prioritization. Nucleic Acids Res. 2009;ume 37:W305–W311.
Online Mendelian Inheritance in Man, OMIM®. McKusick-Nathans Institute of Genetic Medicine, Johns Hopkins University (Baltimore, MD) [Internet]. [cited 2024 Dec 20]. Available from: https://omim.org/.
Singh A, Balasundaram MK, Gupta D. Trofinetide in Rett Syndrome: A Brief Review of Safety And Efficacy. Intractable Rare Dis Res. 2023;12:262–6. https://doi.org/10.5582/irdr.2023.01060.
Tedesco Silva LM, Sharma S, Schrauwen I, Margolesky J, Detyniecki K. A de novo HECW2 Variant In A Patient With Acetazolamide-Responsive Episodic Ataxia. Cerebellum. 2023;22:1029–33.
Mercimek-Andrews S. STXBP1 encephalopathy with epilepsy. 2016 Dec 1 [updated 2023 Sep 28]. In: Adam MP, Feldman J, Mirzaa GM, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK396561/
Freibauer A, Wohlleben M, Boelman C. STXBP1-Related Disorders: Clinical Presentation, Molecular Function, Treatment, And Future Directions. Genes (Basel). 2023;14:2179.
Feliciano P, Zhou X, Astrovskaya I, Turner TN, Wang T, Brueggeman L, et al. Exome Sequencing of 457 Autism Families Recruited Online Provides Evidence For Autism Risk Genes. NPJ Genom Med. 2019;4:19–22.
Martinez-Granero F, Blanco-Kelly F, Sanchez-Jimeno C, Avila-Fernandez A, Arteche A, Bustamante-Aragones A, et al. Comparison of The Diagnostic Yield Of Acgh And Genome-Wide Sequencing Across Different Neurodevelopmental Disorders. NPJ Genom Med. 2021;6:25.
Shen Y, Dies KA, Holm IA, Bridgemohan C, Sobeih MM, Caronna EB, et al. Clinical Genetic Testing For Patients With Autism Spectrum Disorders. Pediatrics. 2010;125:e727–35.
Tammimies K, Marshall CR, Walker S, Kaur G, Thiruvahindrapuram B, Lionel AC, et al. Molecular Diagnostic Yield Of Chromosomal Microarray Analysis And Whole-Exome Sequencing In Children With Autism Spectrum Disorder. JAMA. 2015;314:895–903.
Miyake N, Tsurusaki Y, Fukai R, Kushima I, Okamoto N, Ohashi K, et al. Molecular Diagnosis Of 405 Individuals With Autism Spectrum Disorder. Eur J Hum Genet. 2024;32:1551–8.
Bruno LP, Doddato G, Valentino F, Baldassarri M, Tita R, Fallerini C, et al. New Candidates for Autism/Intellectual Disability Identified by Whole-Exome Sequencing. Int J Mol Sci. 2021;22:13439.
Du X, Gao X, Liu X, Shen L, Wang K, Fan Y, et al. Genetic Diagnostic Evaluation of Trio-Based Whole Exome Sequencing Among Children With Diagnosed or Suspected Autism Spectrum Disorder. Front Genet. 2018;9:594.
Leppa VM, Kravitz SN, Martin CL, Andrieux J, Le Caignec C, Martin-Coignard D, et al. Rare Inherited and De Novo CNVs Reveal Complex Contributions to ASD Risk in Multiplex Families. Am J Hum Genet. 2016;99:540–54.
Satterstrom FK, Kosmicki JA, Wang J, Breen MS, De Rubeis S, An JY, et al. Large-Scale Exome Sequencing Study Implicates Both Developmental and Functional Changes in the Neurobiology of Autism. Cell. 2020;180:568–584.e23.
Cirnigliaro M, Chang TS, Arteaga SA, Pérez-Cano L, Ruzzo EK, Gordon A, et al. The Contributions Of Rare Inherited And Polygenic Risk To ASD In Multiplex Families. Proc Natl Acad Sci USA. 2023;120:e2215632120.
Fan H, Lu J, Guo Y, Li D, Zhang ZM, Tsai YH, et al. BAHCC1 Binds H3k27me3 Via A Conserved Bah Module To Mediate Gene Silencing And Oncogenesis. Nat Genet. 2020;52:1384–96.
Hezroni H, Ben-Tov Perry R, Gil N, Degani N, Ulitsky I. Regulation of Neuronal Commitment In Mouse Embryonic Stem Cells By The Reno1/Bahcc1 Locus. EMBO Rep. 2020;21:e51264.
De Rubeis S, He X, Goldberg AP, Poultney CS, Samocha K, Cicek AE, et al. Synaptic, Transcriptional And Chromatin Genes Disrupted In Autism. Nature. 2014;515:209–15.
Helsmoortel C, Vulto-van Silfhout AT, Coe BP, Vandeweyer G, Rooms L, van den Ende J, et al. SWI/SNF-Related Autism Syndrome Caused By De Novo Mutations In ADNP. Nat Genet. 2014;46:380–4.
Garrido-Torres N, Marqués Rodríguez R, Alemany-Navarro M, Sánchez-García J, García-Cerro S, Ayuso MI, et al. Exploring Genetic Testing Requests, Genetic Alterations And Clinical Associations In A Cohort Of Children With Autism Spectrum Disorder. Eur Child Adolesc Psychiatry. 2024;33:3829–40.
Amiet C, Couchon E, Carr K, Carayol J, Cohen D. Are There Cultural Differences In Parental Interest In Early Diagnosis And Genetic Risk Assessment For Autism Spectrum Disorder? Front Pediatr. 2014;2:32.
Wang L, Wang B, Wu C, Wang J, Sun M. Autism Spectrum Disorder: Neurodevelopmental Risk Factors, Biological Mechanism, and Precision Therapy. Int J Mol Sci. 2023;24:1819.
Clark MM, Stark Z, Farnaes L, Tan TY, White SM, Dimmock D, et al. Meta-Analysis of The Diagnostic And Clinical Utility Of Genome And Exome Sequencing And Chromosomal Microarray In Children With Suspected Genetic Diseases. NPJ Genom Med. 2018;3:16.
Acknowledgements
We would like to thank Ms. Niharika Jadeja and Ms. Tasneem Shaikh for their technical support in drafting the manuscript. The study was supported by an academic grant from MedGenome Labs, Bangalore.
Author information
Authors and Affiliations
Contributions
Shruti Bajaj: conceptualization, methodology, data analysis, data curation, clinical collaborator, drafting and critical review of the manuscript; Shreya Gandhi: data collection and analysis, drafting the manuscript; Thenral S. Geetha and Malini Venkata: conceptualization, methodology, data analysis, data curation, drafting and critical review of the manuscript; Anita Chitre: conceptualization, data curation, neuropsychological analysis of the participants, critical review of the manuscript; Nazema Sagi: project conceptualization, data curation, neuropsychological analysis of the participants, critical review of the manuscript; Nisha Agrawal: project conceptualization, data curation, neuropsychological analysis of the participants, critical review of the manuscript; Suhani Shah: data collection and analysis, drafting the manuscript; Ruta Deo: data collection and analysis, critical review of the manuscript; Sudharshana Pai, Kripa Saira Jacob, Sakthivel Murugan, Ramesh Menon, Ravi Gupta, Jeevana Praharsha Athota, Nagaraja M. Phani, Akshi Bassi, Sandeep Charugulla: data analysis, data curation, critical review of the manuscript; Vishakha Mali: data analysis, data curation, drafting the manuscript; Koyeli Sengupta, Puja Mehta, Vrajesh Udani, Pradnya Gadgil: clinical collaborator, data curation, critical review of the manuscript; Ramprasad Vedam: conceptualization, methodology, data analysis and curation, critical review of the manuscript; Anaita Udwadia Hegde: conceptualization, methodology, clinical collaborator, data analysis, drafting and critical review of the manuscript. All authors have contributed significantly to the preparation of this manuscript, have reviewed its contents, and have approved the final version for submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The authors confirm that the ethical policies of the journal, as noted on the journal’s author guidelines page, have been adhered to. The study was approved by an ethics committee, study number: R-202112.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bajaj, S., Gandhi, S., Geetha, T.S. et al. Prospective study to analyze the yield and clinical impact of trio exome sequencing in 137 Indian children with autism spectrum disorder. J Hum Genet 70, 611–624 (2025). https://doi.org/10.1038/s10038-025-01368-4
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s10038-025-01368-4