Abstract
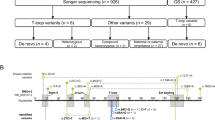
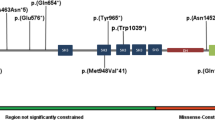
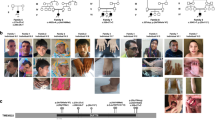
Tenascin-R (TNR) is an extracellular matrix glycoprotein that is essential for the formation of perineuronal nets in the central nervous system and is critical for neurite outgrowth, synaptic plasticity, and neural stem cell proliferation and differentiation. Biallelic TNR variants were reported to cause neurodevelopmental disorders with developmental delay, hypotonia, spasticity, and a variety of motor abnormalities. Here, we describe two Japanese siblings sharing novel compound heterozygous TNR missense variants (NM_003285.3:c.[1783 G > A];[3766 C > T] p.[(Asp595Asn)];[(Arg1256Cys)]) identified by exome and Sanger sequencing. The elder brother had dystonia, while the younger sister was asymptomatic except for adult-onset restless legs syndrome. Their development and intellect were normal. A total of 15 patients, including 13 previously reported patients, showed diverse phenotypic variability and severity, even among individuals sharing the same variants, indicating variable expressivity and reduced penetrance possibly influenced by genetic or environmental modifiers. Our findings extend the clinical spectrum of TNR-related disease and highlight the need for further accumulation of clinical cases and functional studies to understand genotype–phenotype correlations and the pathogenesis of diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The relevant genomic data have not been deposited to public databases but are available on request to the corresponding author.
References
di Biase L, Di Santo A, Caminiti ML, Pecoraro PM, Carbone SP, Di Lazzaro V. Dystonia diagnosis: clinical neurophysiology and genetics. J Clin Med. 2022;11:4184.
Lange LM, Gonzalez-Latapi P, Rajalingam R, Tijssen MAJ, Ebrahimi-Fakhari D, Gabbert C, et al. Nomenclature of genetic movement disorders: recommendations of the International Parkinson and Movement Disorder Society Task Force - An update. Mov Disord. 2022;37:905–35.
Thomsen M, Lange LM, Zech M, Lohmann K. Genetics and pathogenesis of dystonia. Annu Rev Pathol. 2024;19:99–131.
Galtrey CM, Fawcett JW. The role of chondroitin sulfate proteoglycans in regeneration and plasticity in the central nervous system. Brain Res Rev. 2007;54:1–18.
Pesheva P, Spiess E, Schachner M. J1-160 and J1-180 are oligodendrocyte-secreted nonpermissive substrates for cell adhesion. J Cell Biol. 1989;109:1765–78.
Bruckner G, Grosche J, Schmidt S, Hartig W, Margolis RU, Delpech B, et al. Postnatal development of perineuronal nets in wild-type mice and in a mutant deficient in tenascin-R. J Comp Neurol. 2000;428:616–29.
Bukalo O, Schachner M, Dityatev A. Modification of extracellular matrix by enzymatic removal of chondroitin sulfate and by lack of tenascin-R differentially affects several forms of synaptic plasticity in the hippocampus. Neuroscience. 2001;104:359–69.
Xu JC, Xiao MF, Jakovcevski I, Sivukhina E, Hargus G, Cui YF, et al. The extracellular matrix glycoprotein tenascin-R regulates neurogenesis during development and in the adult dentate gyrus of mice. J Cell Sci. 2014;127:641–52.
Montag-Sallaz M, Montag D. Severe cognitive and motor coordination deficits in tenascin-R-deficient mice. Genes Brain Behav. 2003;2:20–31.
Christen M, Gutierrez-Quintana R, James M, Faller KME, Lowrie M, Rusbridge C, et al. A TNR frameshift variant in Weimaraner dogs with an exercise-induced paroxysmal movement disorder. Mov Disord. 2023;38:1094–99.
Sethi MK, Zaia J. Extracellular matrix proteomics in schizophrenia and Alzheimer’s disease. Anal Bioanal Chem. 2017;409:379–94.
Dong Y, Zhao K, Qin X, Du G, Gao L. The mechanisms of perineuronal net abnormalities in contributing aging and neurological diseases. Ageing Res Rev. 2023;92:102092.
Wagner M, Levy J, Jung-Klawitter S, Bakhtiari S, Monteiro F, Maroofian R, et al. Loss of TNR causes a nonprogressive neurodevelopmental disorder with spasticity and transient opisthotonus. Genet Med. 2020;22:1061–68.
Utsuno Y, Hamada K, Hamanaka K, Miyoshi K, Tsuchimoto K, Sunada S, et al. Novel missense variants cause intermediate phenotypes in the phenotypic spectrum of SLC5A6-related disorders. J Hum Genet. 2024;69:69–77.
Sinha A, Kawakami J, Cole KS, Ladutska A, Nguyen MY, Zalmai MS, et al. Protein-protein interactions between tenascin-R and RPTPζ/phosphacan are critical to maintain the architecture of perineuronal nets. J Biol Chem. 2023;299:104952.
Abramson J, Adler J, Dunger J, Evans R, Green T, Pritzel A, et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature. 2024;630:493–500.
Guerois R, Nielsen JE, Serrano L. Predicting changes in the stability of proteins and protein complexes: a study of more than 1000 mutations. J Mol Biol. 2002;320:369–87.
Schymkowitz J, Borg J, Stricher F, Nys R, Rousseau F, Serrano L. The FoldX web server: an online force field. Nucleic Acids Res. 2005;33:W382–8.
Ramos VF, Karp BI, Hallett M. Tricks in dystonia: ordering the complexity. J Neurol Neurosurg Psychiatry. 2014;85:987–93.
Carnemolla B, Leprini A, Borsi L, Querze G, Urbini S, Zardi L. Human tenascin-R. Complete primary structure, pre-mRNA alternative splicing and gene localization on chromosome 1q23-q24. J Biol Chem. 1996;271:8157–60.
Michetti C, Falace A, Benfenati F, Fassio A. Synaptic genes and neurodevelopmental disorders: From molecular mechanisms to developmental strategies of behavioral testing. Neurobiol Dis. 2022;173:105856.
DeLong MR. Primate models of movement disorders of basal ganglia origin. Trends Neurosci. 1990;13:281–5.
Simonyan K, Cho H, Hamzehei Sichani A, Rubien-Thomas E, Hallett M. The direct basal ganglia pathway is hyperfunctional in focal dystonia. Brain. 2017;140:3179–90.
Earley CJ, Hyland K, Allen RP. Circadian changes in CSF dopaminergic measures in restless legs syndrome. Sleep Med. 2006;7:263–8.
Allen RP. Restless Leg Syndrome/Willis-Ekbom Disease Pathophysiology. Sleep Med Clin. 2015;10:207–14.
Morawski M, Dityatev A, Hartlage-Rübsamen M, Blosa M, Holzer M, Flach K, et al. Tenascin-R promotes assembly of the extracellular matrix of perineuronal nets via clustering of aggrecan. Philos Trans R Soc Lond B Biol Sci. 2014;369:20140046.
Gwilt MA, Hodgson AR, Axelsson SFA, Cockcroft GJ, McIver LB, Hird M, et al. Hippocampal perineuronal net degradation identifies prefrontal and striatal circuits involved in schizophrenia-like changes in marmosets. Sci Adv. 2025;11:eadu0975.
Ozelius LJ, Hewett JW, Page CE, Bressman SB, Kramer PL, Shalish C, et al. The early-onset torsion dystonia gene (DYT1) encodes an ATP-binding protein. Nat Genet. 1997;17:40–8.
Carecchio M, Invernizzi F, Gonzàlez-Latapi P, Panteghini C, Zorzi G, Romito L, et al. Frequency and phenotypic spectrum of KMT2B dystonia in childhood: a single-center cohort study. Mov Disord. 2019;34:1516–27.
Wiessner M, Roos A, Munn CJ, Viswanathan R, Whyte T, Cox D, et al. Mutations in INPP5K, encoding a phosphoinositide 5-phosphatase, cause congenital muscular dystrophy with cataracts and mild cognitive impairment. Am J Hum Genet. 2017;100:523–36.
Yousaf S, Sheikh SA, Riazuddin S, Waryah AM, Ahmed ZM. INPP5K variant causes autosomal recessive congenital cataract in a Pakistani family. Clin Genet. 2018;93:682–6.
Acknowledgements
We thank the individuals and their family for their participation in this study. We also thank N. Watanabe, T. Miyama, M. Sato, S. Sugimoto, and K. Takabe for their technical assistance. We thank Edanz (https://jp.edanz.com/ac) for editing the English text of a draft of this manuscript. This work was supported by the Japan Agency for Medical Research and Development (AMED) (grant numbers JP25ek0109674, JP25ek0109760, JP25ek0109617, JP25ek0109648, and JP25ek0109677 to N. Matsumoto); the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Scientific Research (grant number JP22K15901 to A. Fujita, JP23H02829 to S. Miyatake, JP23H02877 to T. Mizuguchi, JP22K15901 to K. Hamanaka, JP23K07229 to Y. Uchiyama, JP23K15353 to N. Tsuchida, JP21K07869 to E. Koshimizu, JP24K18862 to M. Sakamoto, and JP24K02230 to N. Matsumoto); the Takeda Science Foundation (T. Mizuguchi and N. Matsumoto); and Kawano Masanori Memorial Public Interest Incorporated Foundation for the Promotion of Pediatrics (S. Miyatake).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ozaki, A., Sakamoto, M., Kumada, S. et al. Biallelic variants in TNR cause neurodevelopmental disorders with variable expressivity. J Hum Genet 71, 231–237 (2026). https://doi.org/10.1038/s10038-025-01431-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s10038-025-01431-0