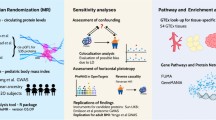
Abstract
Objective
To investigate the causal relationship between plasma metabolites and ferroptosis-related genes in childhood obesity and to explore the potential mediating role of ferroptosis-related genes in the association between plasma metabolites and childhood obesity risk.
Methods
A bidirectional two-step Mendelian randomization (MR) approach was applied, leveraging publicly available genome-wide association study (GWAS) datasets to analyze the causal relationship among 1400 plasma metabolites, 159 ferroptosis-related genes, and childhood obesity. In the first step, protein quantitative trait loci (pQTL) data corresponding to ferroptosis-related genes were identified as mediators to evaluate the causal effects of plasma metabolites and ferroptosis-related genes on childhood obesity. In the second step, MR analysis was conducted on ferroptosis-related genes and plasma metabolites identified in the first step to confirm their causal association. The inverse-variance weighted (IVW) method was primarily used for meta-analysis, while MR-PRESSO was employed to detect pleiotropy and outliers.
Results
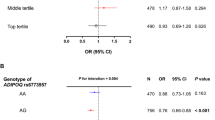
Four ferroptosis-related genes (SMPD1 and SIRT3 suppressing obesity, GSTZ1 and ADAMTS13 promoting obesity) and nine plasma metabolites were found to be significantly associated with childhood obesity (six negatively correlated and three positively correlated). Further mediation analysis indicated that the ferroptosis mechanism regulated by SMPD1 and SIRT3 partially mediated the association between specific plasma metabolites and childhood obesity, with the highest mediation proportion reaching 9.62%. Sensitivity analysis confirmed the robustness of the results (no heterogeneity or horizontal pleiotropy), and reverse Mendelian randomization ruled out causal interference.
Conclusion
This study is the first to reveal, through Mendelian randomization analysis, the potential mediating role of ferroptosis-related genes in the association between plasma metabolites and childhood obesity. It suggests that the ferroptosis mechanism may influence childhood obesity risk by regulating specific metabolites. These findings contribute to understanding the role of ferroptosis in the pathological mechanisms of childhood obesity and provide novel molecular targets and intervention strategies for obesity prevention and treatment in children.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets analyzed during the current study are available in the Finngen database (https://www.finngen.fi/en) and IEU OpenGWAS (https://gwas.mrcieu.ac.uk/)、Ferroptosis-related genes were retrieved from FerrDb (http://www.zhounan.org/ferrdb/current/).
References
Kelly AS, Armstrong SC, Michalsky MP, Fox CK. Obesity in adolescents: a review. JAMA. 2024;332:738–48.
Myette RL, Flynn JT. The ongoing impact of obesity on childhood hypertension. Pediatr Nephrol. 2024;39:2337–46.
Yuan C, Dong Y, Chen H, Ma L, Jia L, Luo J, et al. Determinants of childhood obesity in China. Lancet Public Health. 2024;9:e1105–14.
Iona A, Yao P, Pozarickij A, Kartsonaki C, Said S, Wright N, et al. Proteo-genomic analyses in relatively lean Chinese adults identify proteins and pathways that affect general and central adiposity levels. Commun Biol. 2024;7:1327.
Dixon SJ, Olzmann JA. The cell biology of ferroptosis. Nat Rev Mol Cell Biol. 2024;25:424–42.
Dai E, Chen X, Linkermann A, Jiang X, Kang R, Kagan VE, et al. A guideline on the molecular ecosystem regulating ferroptosis. Nat Cell Biol. 2024;26:1447–57.
Li S, Zhang G, Hu J, Tian Y, Fu X. Ferroptosis at the nexus of metabolism and metabolic diseases. Theranostics. 2024;14:5826–52.
Ma W, Jia L, Xiong Q, Du H. Iron Overload Protects from Obesity by Ferroptosis. Foods. 2021;10:1787.
Gijbels A, Jardon KM, Trouwborst I, Manusama KC, Goossens GH, Blaak EE, et al. Fasting and postprandial plasma metabolite responses to a 12-wk dietary intervention in tissue-specific insulin resistance: a secondary analysis of the PERSonalized glucose Optimization through Nutritional intervention (PERSON) randomized trial. Am J Clin Nutr. 2024;120:347–59.
Burgess S, Thompson SG. Use of allele scores as instrumental variables for Mendelian randomization. Int J Epidemiol. 2013;42:1134–44.
Sekula P, Del GMF, Pattaro C, Kottgen A. Mendelian Randomization as an Approach to Assess Causality Using Observational Data. J Am Soc Nephrol. 2016;27:3253–65.
Chen Y, Lu T, Pettersson-Kymmer U, Stewart ID, Butler-Laporte G, Nakanishi T, et al. Genomic atlas of the plasma metabolome prioritizes metabolites implicated in human diseases. Nat Genet. 2023;55:44–53.
Bradfield JP, Taal HR, Timpson NJ, Scherag A, Lecoeur C, Warrington NM, et al. A genome-wide association meta-analysis identifies new childhood obesity loci. Nat Genet. 2012;44:526–31.
Ferkingstad E, Sulem P, Atlason BA, Sveinbjornsson G, Magnusson MI, Styrmisdottir EL, et al. Large-scale integration of the plasma proteome with genetics and disease. Nat Genet. 2021;53:1712–21.
Bowden J, Holmes MV. Meta-analysis and Mendelian randomization: A review. Res Synth Methods. 2019;10:486–96.
Burgess S, Small DS, Thompson SG. A review of instrumental variable estimators for Mendelian randomization. Stat Methods Med Res. 2017;26:2333–55.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266–82.
Sun Y, Chen P, Zhai B, Zhang M, Xiang Y, Fang J, et al. The emerging role of ferroptosis in inflammation. Biomed Pharmacother. 2020;127:110108.
Chen X, Kang R, Kroemer G, Tang D Ferroptosis in infection, inflammation, and immunity. J Exp Med. 2021;218:e20210518.
Cho CH, Patel S, Rajbhandari P. Adipose tissue lipid metabolism: lipolysis. Curr Opin Genet Dev. 2023;83:102114.
Pope LE, Dixon SJ. Regulation of ferroptosis by lipid metabolism. Trends Cell Biol. 2023;33:1077–87.
Lin Z, Liu J, Kang R, Yang M, Tang D. Lipid Metabolism in Ferroptosis. Adv Biol. 2021;5:e2100396.
Zhou L, Pinho R, Gu Y, Radak Z. The Role of SIRT3 in Exercise and Aging. Cells. 2022;11:2596.
Diao Z, Ji Q, Wu Z, Zhang W, Cai Y, Wang Z, et al. SIRT3 consolidates heterochromatin and counteracts senescence. Nucleic Acids Res. 2021;49:4203–19.
Green MF, Hirschey MD. SIRT3 weighs heavily in the metabolic balance: a new role for SIRT3 in metabolic syndrome. J Gerontol A Biol Sci Med Sci. 2013;68:105–7.
Huang Y, Sulek K, Stinson SE, Holm LA, Kim M, Trost K, et al. Lipid profiling identifies modifiable signatures of cardiometabolic risk in children and adolescents with obesity. Nat Med. 2025;31:294–305.
Ueda S, Manabe Y, Kubo N, Morino N, Yuasa H, Shiotsu M, et al. Early secretory pathway-resident Zn transporter proteins contribute to cellular sphingolipid metabolism through activation of sphingomyelin phosphodiesterase 1. Am J Physiol Cell Physiol. 2022;322:C948–59.
Gorelik A, Illes K, Heinz LX, Superti-Furga G, Nagar B. Crystal structure of mammalian acid sphingomyelinase. Nat Commun. 2016;7:12196.
Acknowledgements
We would like to thank all the authors who contributed to the drafting of the manuscript.
Funding
This study was funded by the Guangxi Science and Technology Program Project (Guike AD22035121).
Author information
Authors and Affiliations
Contributions
Ji-Gan Wang designed the study and interpreted the results. Ji-Gan Wang and Xiu-Hua Pan was responsible for the conceptualization, methodology, data analysis, and manuscript writing. Xiu-Hua Pan and Yan Li participated in supervising the study, project management, and funding acquisition, and reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
In this MR study, we used publicly available aggregate data; therefore, no separate ethical approval is required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, JG., Pan, XH. & Li, Y. Plasma metabolites may inhibit childhood obesity by regulating ferroptosis through SMPD1 and SIRT3. Int J Obes 50, 414–420 (2026). https://doi.org/10.1038/s41366-025-01951-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41366-025-01951-x