Abstract
Objective
Pharmacokinetic (PK) data to guide cefazolin dosing in premature infants are virtually non-existent. Therefore, we aimed to characterize cefazolin PK in infants aged ≤32 weeks of gestation at birth.
Study Design
We conducted a prospective, open-label PK and safety study of cefazolin in infants ≤32 weeks gestation from a University Medical Center. We administered intravenous cefazolin and collected both timed and scavenged blood samples. We analyzed data using non-linear mixed effect modeling and simulated several dosage regimens to achieve target concentrations against methicillin-susceptible Staphylococcus aureus.
Results
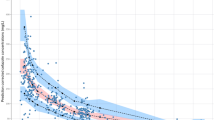
We analyzed 40 samples from nine infants and observed that premature infants had lower clearance and greater volume of distribution for cefazolin compared to older children. The median (range) individual Bayesian estimates were 0.03 L/h/kg (0.01–0.08) for clearance and 0.39 L/kg (0.31–0.52) for volume.
Conclusion
Simulations suggested reduced cefazolin dosing based on postmenstrual age achieve target concentrations and potentially reduce unnecessary exposure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Cohen-Wolkowiez M, Watt KM, Zhou C, Bloom BT, Poindexter B, Castro L, et al. Developmental pharmacokinetics of piperacillin and tazobactam using plasma and dried blood spots from infants. Antimicrob Agents Chemother. 2014;58:2856–65.
Product Information. Cefazolin Injection. Sagent Pharmaceuticals. Schaumburg, IL. 2017.
Drusano GL. Prevention of resistance: a goal for dose selection for antimicrobial agents. Clin Infect Dis. 2003;36(Suppl 1):S42–S50.
De Cock RFW, Smits A, Allegaert K, de Hoon J, Saegeman V, Danhof M, et al. Population pharmacokinetic modelling of total and unbound cefazolin plasma concentrations as a guide for dosing in preterm and term neonates. J Antimicrob Chemoth. 2014;69:1330–8.
Schmitz ML, Blumer JL, Cetnarowski W, Rubino CM. Determination of appropriate weight-based cutoffs for empiric cefazolin dosing using data from a phase 1 pharmacokinetics and safety study of cefazolin administered for surgical prophylaxis in pediatric patients aged 10 to 12 years. Antimicrob Agents Chemother. 2015;59:4173–80.
Koshida R, Nakashima E, Ichimura F, Nakano O, Watanabe R, Taniguchi N, et al. Comparative distribution kinetics of cefazolin and tobramycin in children. J Pharmacobiodyn. 1987;10:436–42.
Nahata MC, Durrell DE, Ginn-Pease ME, King DR. Pharmacokinetics and tissue concentrations of cefazolin in pediatric patients undergoing gastrointestinal surgery. Eur J Drug Metab Pharmacokinet. 1991;16:49–52.
Deguchi Y, Koshida R, Nakashima E, Watanabe R, Taniguchi N, Ichimura F, et al. Interindividual changes in volume of distribution of cefazolin in newborn infants and its prediction based on physiological pharmacokinetic concepts. J Pharm Sci. 1988;77:674–8.
Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing. 27th ed. CLSI supplement M100. Wayne, PA: Clinical and Laboratory Standards Institute; 2017. Accessed 28 December 2017.
Pedreira C, Pinto F, Pereira S, Costa E. Birth weight patterns by gestational age in Brazil. An Acad Bras Cienc. 2011;83:619–25.
Sulemanji M, Vakili K. Neonatal renal physiology. Semin Pediatr Surg. 2013;22:195–8.
Acknowledgements
This work was supported by NIH-1K23HD060040-04 (NICHD), 2T32GM086330-06 (NIGMS/NICHD), K23 HD091398-01 (NICHD), R35 GM122576 (NIGMS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
SJB received salary and/or research support from the National Institutes of Health (2T32GM086330-06, 5R01-HD076676-04, HHSN275201000003I, HHSN275201800003I, 5U24-TR001608-03), the Rheumatology Research Foundation’s Scientist Development Award, and the Thrasher Research Fund. The National Institutes of Health sponsor open access. PBS receives support from industry (www.dcri.duke.edu/research/coi.jsp). HW receives salary support for research from the National Institutes of Health Clinical and Translational Science Award (5UL1TR001117-05). KLRB receives funding from NIGMS, R35 GM122576. KOZ receives support for research from the National Institute for Child Health and Human Development (NICHD) (HHSN275201000003I and K23HD091398) and the Duke Clinical and Translational Science Awards (KL2TR001115). NDR-C received funding from training grant T32 from the National Institute of Child Health and Human Development (T32GM086330-06). DKBJr receives support from the National Institutes of Health (award 2K24HD058735-10, National Institute of Child Health and Human Development (HHSN275201000003I), National Institute of Allergy and Infectious Diseases (HHSN272201500006I), ECHO Program (1U2COD023375-02), and the National Center for Advancing Translational Sciences (1U24TR001608-03); he also receives research support from Cempra Pharmaceuticals (subaward to HHSO100201300009C) and industry for neonatal and pediatric drug development (www.dcri.duke.edu/research/coi.jsp). MC-W receives support for research from the NIH (5R01-HD076676, HHSN275201000003I), NIAID/NIH (HHSN272201500006I), FDA (1U18-FD006298), the Biomedical Advanced Research and Development Authority (HHSO1201300009C), and from the industry for the drug development in adults and children (www.dcri.duke.edu/research/coi.jsp). The remaining author declare that he has no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Balevic, S.J., Smith, P.B., Testoni, D. et al. Cefazolin pharmacokinetics in premature infants. J Perinatol 39, 1213–1218 (2019). https://doi.org/10.1038/s41372-019-0368-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41372-019-0368-z