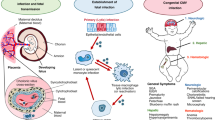
Abstract
Objective
To determine the frequency of abnormal findings on evaluation of neonates with congenital CMV infection who have a normal physical examination
Study design
Retrospective, 2-center study (1996–2017) that reviewed results of complete blood cell count and platelets, serum alanine aminotransferase (ALT) and bilirubin concentrations, eye examination, cranial ultrasonography or other neuroimaging, and brainstem evoked responses performed on neonates with congenital CMV infection and a normal physical examination
Results
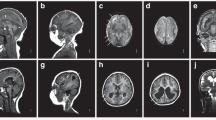
Of 34 infants with congenital CMV infection and a normal physical examination, 56% (19/34) had ≥1 abnormality: 39%, elevated ALT concentration; 45%, abnormal neuroimaging (five, lenticulostriate vasculopathy; six, intraventricular hemorrhage; four, calcifications); 12%, anemia; 16%, thrombocytopenia; and 3%, chorioretinitis. Seven (21%) infants had sensorineural hearing loss, and 18 infants received antiviral therapy.
Conclusion
Some infants with congenital CMV infection and a normal physical examination had abnormalities on laboratory or neuroimaging evaluation, which in some cases prompted antiviral treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Manicklal S, Emery VC, Lazzarotto T, Boppana SB, Gupta RK. The “silent” global burden of congenital cytomegalovirus. Clin Microbiol Rev. 2013;26:86–102.
Dollard SC, Grosse SD, Ross DS. New estimates of the prevalence of neurological and sensory sequelae and mortality associated with congenital cytomegalovirus infection. Rev Med Virol. 2007;17:355–63.
Boppana SB, Ross SA, Shimamura M, Palmer AL, Ahmed A, Michaels MG, et al. Saliva polymerase-chain-reaction assay for cytomegalovirus screening in newborns. New Engl J Med. 2011;364:2111–8.
Foulon I, Naessens A, Foulon W, Casteels A, Gordts F. A 10-year prospective study of sensorineural hearing loss in children with congenital cytomegalovirus infection. J Pediatr. 2008;153:84–88.
Madden C, Wiley S, Schleiss M, Benton C, Meinzen-Derr J, Greinwald J, et al. Audiometric, clinical and educational outcomes in a pediatric symptomatic congenital cytomegalovirus (CMV) population with sensorineural hearing loss. Int J Pediatr Otorhinolaryngol. 2005;69:1191–8.
Cantey JB, Sanchez PJ. Overview of congenital infections: the prominence of cytomegalovirus. Infect Disord Drug Targets. 2011;11:426–31.
Fowler KB. Congenital cytomegalovirus infection: audiologic outcome. Clin Infect Dis. 2013;57:S182–4.
Rivera LB, Boppana SB, Fowler KB, Britt WJ, Stagno S, Pass RF. Predictors of hearing loss in children with symptomatic congenital cytomegalovirus infection. Pediatrics. 2002;110:762–7.
Ross SA, Ahmed A, Palmer AL, Michaels MG, Sanchez PJ, Bernstein DI, et al. Detection of congenital cytomegalovirus infection by real-time polymerase chain reaction analysis of saliva or urine specimens. J Infect Dis. 2014;210:1415–8.
Boppana SB, Ross SA, Novak Z, Shimamura M, Tolan RW Jr, Palmer AL, et al. Dried blood spot real-time polymerase chain reaction assays to screen newborns for congenital cytomegalovirus infection. JAMA. 2010;303:1375–82.
Stehel EK, Shoup AG, Owen KE, Jackson GL, Sendelbach DM, Boney LF, et al. Newborn hearing screening and detection of congenital cytomegalovirus infection. Pediatrics. 2008;121:970–5.
Duryea EL, Sanchez PJ, Sheffield JS, Jackson GL, Wendel GD, McElwee BS, et al. Maternal human immunodeficiency virus infection and congenital transmission of cytomegalovirus. Pediatr Infect Dis J. 2010;29:915–8.
Ellington SR, Clarke KE, Kourtis AP. Cytomegalovirus Infection in Human Immunodeficiency Virus (HIV)-Exposed and HIV-Infected Infants: a systematic review. J Infect Dis. 2016;213:891–900.
Jopling J, Henry E, Wiedmeier SE, Christensen RD. Reference ranges for hematocrit and blood hemoglobin concentration during the neonatal period: data from a multihospital health care system. Pediatrics. 2009;123:e333–7.
Northern JL, Downs MP. Hearing in children. USA: Lippincott Williams & Wilkins; 2001.
Rawlinson WD, Boppana SB, Fowler KB, Kimberlin DW, Lazzarotto T, Alain S, et al. Congenital cytomegalovirus infection in pregnancy and the neonate: consensus recommendations for prevention, diagnosis, and therapy. Lancet Infect Dis. 2017;17:e177–88.
Shoji K, Ito N, Ito Y, Inoue N, Adachi S, Fujimaru T, et al. Is a 6-week course of ganciclovir therapy effective for chorioretinitis in infants with congenital cytomegalovirus infection? J Pediatr. 2010;157:331–3.
Coors LE, Spencer R. Delayed presentation of cytomegalovirus retinitis in an infant with severe congenital cytomegalovirus infection. Retina. 2010;30:S59–62.
Ghekiere S, Allegaert K, Cossey V, Van Ranst M, Cassiman C, Casteels I. Ophthalmological findings in congenital cytomegalovirus infection: when to screen, when to treat? J Pediatr Ophthalmol Strabismus. 2012;49:274–82.
Cantey JB, Sisman J. The etiology of lenticulostriate vasculopathy and the role of congenital infections. Early Hum Dev. 2015;91:427–30.
El Ayoubi M, de Bethmann O, Monset-Couchard M. Lenticulostriate echogenic vessels: clinical and sonographic study of 70 neonatal cases. Pediatr Radiol. 2003;33:697–703.
Bilavsky E, Schwarz M, Pardo J, Attias J, Levy I, Haimi-Cohen Y, et al. Lenticulostriated vasculopathy is a high-risk marker for hearing loss in congenital cytomegalovirus infections. Acta Paediatr. 2015;104:e388–94.
Amir J, Atias J, Linder N, Pardo J. Follow-up of infants with congenital cytomegalovirus and normal fetal imaging. Arch Dis Child Fetal Neonatal Ed. 2016;101:F428–32.
Amir J, Schwarz M, Levy I, Haimi-Cohen Y, Pardo J. Is lenticulostriated vasculopathy a sign of central nervous system insult in infants with congenital CMV infection? Arch Dis Child. 2011;96:846–50.
Goderis J, De Leenheer E, Smets K, Van Hoecke H, Keymeulen A, Dhooge I. Hearing loss and congenital CMV infection: a systematic review. Pediatrics. 2014;134:972–82.
Bergevin A, Zick CD, McVicar SB, Park AH. Cost-benefit analysis of targeted hearing directed early testing for congenital cytomegalovirus infection. Int J Pediatr Otorhinolaryngol. 2015;79:2090–3.
Fowler KB, McCollister FP, Sabo DL, Shoup AG, Owen KE, Woodruff JL, et al. A targeted approach for congenital cytomegalovirus screening within newborn hearing screening. Pediatrics. 2017;139:e20162128.
Diener ML, Zick CD, McVicar SB, Boettger J, Park AH. Outcomes from a hearing-targeted cytomegalovirus screening program. Pediatrics. 2017;139:e20160789.
Cannon MJ, Griffiths PD, Aston V, Rawlinson WD. Universal newborn screening for congenital CMV infection: what is the evidence of potential benefit? Rev Med Virol. 2014;24:291–307.
Ronchi A, Shimamura M, Malhotra PS, Sanchez PJ. Encouraging postnatal cytomegalovirus (CMV) screening: the time is NOW for universal screening! Expert Rev Anti Infective Ther. 2017;15:417–9.
Gantt S, Dionne F, Kozak FK, Goshen O, Goldfarb DM, Park AH, et al. Cost-effectiveness of universal and targeted newborn screening for congenital cytomegalovirus infection. JAMA Pediatr. 2016;107:906.
Kimberlin DW, Jester PM, Sanchez PJ, Ahmed A, Arav-Boger R, Michaels MG, et al. Valganciclovir for symptomatic congenital cytomegalovirus disease. New Engl J Med. 2015;372:933–43.
Oliver SE, Cloud GA, Sanchez PJ, Demmler GJ, Dankner W, Shelton M, et al. Neurodevelopmental outcomes following ganciclovir therapy in symptomatic congenital cytomegalovirus infections involving the central nervous system. J Clin Virol. 2009;46:S22–6.
Kimberlin DW, Lin CY, Sanchez PJ, Demmler GJ, Dankner W, Shelton M, et al. Effect of ganciclovir therapy on hearing in symptomatic congenital cytomegalovirus disease involving the central nervous system: a randomized, controlled trial. J Pediatr. 2003;143:16–25.
Forner G, Abate D, Mengoli C, Palu G, Gussetti N. High cytomegalovirus (CMV) DNAemia predicts CMV sequelae in asymptomatic congenitally infected newborns born to women with primary infection during pregnancy. J Infect Dis. 2015;212:67–71.
Funding
AR received grant support from “A. Griffini—J. Miglierina” Fundation—Provincia di Varese Piazza Libertà 1- 21100 Varese-Italy. The Fundation was not involved in the (1) study design, (2) the collection, analysis, and interpretation of data; (3) the writing of the report; and (4) the decision to submit the paper for publication. AR wrote the first draft of the paper. The other authors have no financial relationships relevant to this article to disclose.
Author information
Authors and Affiliations
Contributions
AR, had full access to all study data and is responsible for the data integrity and accuracy of the data analysis. He participated in the study concept and design, acquisition of data, analysis and interpretation of data, initial and subsequent drafts of the paper, and approved the final paper as submitted. FZ, participated in the study concept and design, acquisition of data, critical revision of the paper, and approved the final paper as submitted. LE.L, participated in the study concept and design, acquisition of data, critical revision of the paper, and approved the final paper as submitted. KE.O, participated in the study design, acquisition of data, critical revision of the paper, and approved the final paper as submitted. AG.S, participated in the study concept and design, acquisition of data, critical revision of the paper, and approved the final paper as submitted. FG, had full access to all study data and is responsible for the data integrity and accuracy of the data analysis. She participated in the study concept and design, acquisition of data, analysis and interpretation of data, initial and subsequent drafts of the paper, and approved the final paper as submitted. LN.V, had full access to all study data and is responsible for the data integrity and accuracy of the data analysis. She participated in the study concept and design, acquisition of data, analysis and interpretation of data, initial and subsequent drafts of the paper, and approved the final paper as submitted. JB.C, had full access to all study data and is responsible for the data integrity and accuracy of the data analysis. He participated in the study concept and design, acquisition of data, analysis and interpretation of data, initial and subsequent drafts of the paper, and approved the final paper as submitted. SV, participated in the acquisition of data, critical revision of the paper, and approved the final paper as submitted. LP, supervised the overall study by analysis and interpretation of data, critical revision of the paper for important intellectual content, and approved the final paper as submitted. FM, supervised the overall study by analysis and interpretation of data, critical revision of the paper for important intellectual content, and approved the final paper as submitted. PJ.S, had full access to all study data and is responsible for the data integrity and accuracy of the data analysis. He supervised the overall study by developing the study concept and design, acquisition of data, analysis and interpretation of data, initial and subsequent drafts of the paper, and approved the final paper as submitted.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The study was presented in part at the 4th Congenital Cytomegalovirus Conference, San Francisco, CA, October 29–31, 2012, and at the Pediatric Academic Societies’ Annual Meeting, Vancouver, BC, May 3–6, 2014
Rights and permissions
About this article
Cite this article
Ronchi, A., Zeray, F., Lee, L.E. et al. Evaluation of clinically asymptomatic high risk infants with congenital cytomegalovirus infection. J Perinatol 40, 89–96 (2020). https://doi.org/10.1038/s41372-019-0501-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41372-019-0501-z
This article is cited by
-
Asymptomatic viruses detectable in saliva in the first year of life: a narrative review
Pediatric Research (2024)
-
Loop-mediated isothermal amplification assay for screening congenital cytomegalovirus infection in newborns
Applied Microbiology and Biotechnology (2023)
-
An observational study for appraisal of clinical outcome and risk of mother-to-child SARS-CoV-2 transmission in neonates provided the benefits of mothers’ own milk
European Journal of Pediatrics (2022)
-
Blood genome expression profiles in infants with congenital cytomegalovirus infection
Nature Communications (2020)