Abstract
Objective
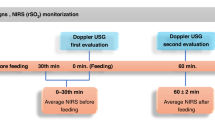
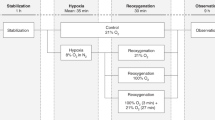
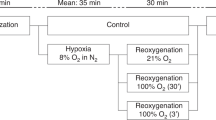
To compare regional splanchnic oxygenation (SrSO2) levels and clinical outcomes between infants who received minimal enteral nutrition (MEN) during Therapeutic Hypothermia (TH), and those who did not.
Study design
A prospective, interventional cohort study with a historic control group at two Regional Perinatal Centers (NCT05471336).
Results
Participant demographics and clinical illness severity were similar between MEN (n = 40) and control (n = 40) groups. There were no significant safety events. SrSO2 levels were normal in both groups throughout. Subjects that received MEN experienced fewer central line days (5.8 v 6.9, p = 0.005) and higher rates of human milk feeding (77% v 46%, p = 0.010), and achieved full oral feeds sooner (9.9 v 13.4 days, p = 0.043).
Conclusion
Splanchnic tissue oxygenation was normal in both groups, and similar between groups throughout TH. Providing MEN during TH appears safe and effective, resulting in several important clinical benefits.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The dataset that supports the findings of this study is not currently available in a public repository, but will be made available to reviewers or readers upon reasonable request, by contacting the corresponding author via e-mail.
References
Jacobs SE, Berg M, Hunt R, Tarnow-Mordi WO, Inder TE, Davis PG. Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst Rev. 2013;2013:CD003311.
Russ JB, Simmons R, Glass HC. Neonatal encephalopathy: beyond hypoxic-ischemic encephalopathy. Neoreviews. 2021;22:e148–62.
Beltempo M, Wintermark P, Mohammad K, Jabbour E, Afifi J, Shivananda S, et al. Variations in practices and outcomes of neonates with hypoxic ischemic encephalopathy treated with therapeutic hypothermia across tertiary NICUs in Canada. J Perinatol. 2022;42:898–906.
Mohammad K, McIntosh S, Lee KS, Beltempo M, Afifi J, Tremblay S, et al. Variations in care of neonates during therapeutic hypothermia: call for care practice bundle implementation. Pediatr Res. 2023;94:321–30.
Chang LL, Wynn JL, Pacella MJ, Rossignol CC, Banadera F, Alviedo N, et al. Enteral feeding as an adjunct to hypothermia in neonates with hypoxic-ischemic encephalopathy. Neonatology. 2018;113:347–52.
Alburaki W, Scringer-Wilkes M, Dawoud F, Oliver N, Lind J, Zein H, et al. Feeding during therapeutic hypothermia is safe and may improve outcomes in newborns with perinatal asphyxia. J Matern Fetal Neonatal Med. 2022;35:9440–4.
Gale C, Longford NT, Jeyakumaran D, Ougham K, Battersby C, Ojha S, et al. Feeding during neonatal therapeutic hypothermia, assessed using routinely collected National Neonatal Research Database data: a retrospective, UK population-based cohort study. Lancet Child Adolesc Health. 2021;5:408–16.
Kumar J, Anne RP, Meena J, Sundaram V, Dutta S, Kumar P. To feed or not to feed during therapeutic hypothermia in asphyxiated neonates: a systematic review and meta-analysis. Eur J Pediatr. 2023;182:2759–73.
Marin T, Moore J. Understanding near-infrared spectroscopy. Adv Neonatal Care. 2011;11:382–8.
Bailey SM, Hendricks-Munoz KD, Mally P. Cerebral, renal, and splanchnic tissue oxygen saturation values in healthy term newborns. Am J Perinatol. 2014;31:339–44.
El-Dib M, Abend NS, Austin T, Boylan G, Chock V, Cilio MR, et al. Neuromonitoring in neonatal critical care part I: neonatal encephalopathy and neonates with possible seizures. Pediatr Res. 2023;94:64–73.
Metcalfe KHM, Stienstra R, McHoney M. NIRS as a biomarker of bowel ischaemia & surgical pathology: a meta-analysis of studies in newborns. Early Hum Dev. 2021;161:105437.
Palleri E, van der Heide M, Hulscher JBF, Bartocci M, Wester T, Kooi EMW. Clinical usefulness of splanchnic oxygenation in predicting necrotizing enterocolitis in extremely preterm infants: a cohort study. BMC Pediatr. 2023;23:336.
Martini S, Corvaglia L. Splanchnic NIRS monitoring in neonatal care: rationale, current applications and future perspectives. J Perinatol. 2018;38:431–43.
Sarnat HB, Sarnat MS. Neonatal encephalopathy following fetal distress. A clinical and electroencephalographic study. Arch Neurol. 1976;33:696–705.
Shankaran S, Barnes PD, Hintz SR, Laptook AR, Zaterka-Baxter KM, McDonald SA, et al. Brain injury following trial of hypothermia for neonatal hypoxic-ischaemic encephalopathy. Arch Dis Child Fetal Neonatal Ed. 2012;97:F398–404.
Hu Y, Chen F, Xiang X, Wang F, Hua Z, Wei H. Early versus delayed enteral nutrition for neonatal hypoxic-ischemic encephalopathy undergoing therapeutic hypothermia: a randomized controlled trial. Ital J Pediatr. 2022;48:146.
Ojha S, Dorling J, Battersby C, Longford N, Gale C. Optimising nutrition during therapeutic hypothermia. Arch Dis Child Fetal Neonatal Ed. 2019;104:F230–F1.
Hazeldine B, Thyagarajan B, Grant M, Chakkarapani E. Survey of nutritional practices during therapeutic hypothermia for hypoxic-ischaemic encephalopathy. BMJ Paediatr Open. 2017;1:e000022.
Thyagarajan B, Tillqvist E, Baral V, Hallberg B, Vollmer B, Blennow M. Minimal enteral nutrition during neonatal hypothermia treatment for perinatal hypoxic-ischaemic encephalopathy is safe and feasible. Acta Paediatr. 2015;104:146–51.
Warchol A, Kwinta P. Nutrition of newborns with hypoxic-ischaemic encephalopathy during therapeutic hypothermia - a survey of practice in polish neonatal care units. J Mother Child. 2024;28:8–13.
Sharma S, Kallesh A, Aradhya AS, Diggikar S, Veeraiah PS, Subbareddy NN, et al. Feasibility of minimal enteral nutrition during therapeutic hypothermia for perinatal asphyxia: a five-year multicenter experience from South India. Indian J Pediatr. 2023;90:513–5.
Sakhuja P, More K, Ting JY, Sheth J, Lapointe A, Jain A, et al. Gastrointestinal hemodynamic changes during therapeutic hypothermia and after rewarming in neonatal hypoxic-Ischemic encephalopathy. Pediatr Neonatol. 2019;60:669–75.
Forman E, Breatnach CR, Ryan S, Semberova J, Miletin J, Foran A, et al. Noninvasive continuous cardiac output and cerebral perfusion monitoring in term infants with neonatal encephalopathy: assessment of feasibility and reliability. Pediatr Res. 2017;82:789–95.
Bailey SM, Mally PV. Review of splanchnic oximetry in clinical medicine. J Biomed Opt. 2016;21:091306.
Bailey SM, Hendricks-Munoz KD, Mally P. Splanchnic-cerebral oxygenation ratio (SCOR) values in healthy term infants as measured by near-infrared spectroscopy (NIRS). Pediatr Surg Int. 2013;29:591–5.
Howarth CN, Leung TS, Banerjee J, Eaton S, Morris JK, Aladangady N. Regional cerebral and splanchnic tissue oxygen saturation in preterm infants - Longitudinal normative measurements. Early Hum Dev. 2022;165:105540.
Thomas RA, Ballard MR, Aladangady N, Banerjee J. Abdominal Near Infrared Spectroscopy can be reliably used to measure splanchnic oxygenation changes in preterm infants. J Perinatol. 2023;43:716–21.
van der Heide M, Dotinga BM, Stewart RE, Kalteren WS, Hulscher JBF, Reijneveld SA, et al. Regional splanchnic oxygen saturation for preterm infants in the first week after birth: reference values. Pediatr Res. 2021;90:882–7.
Fortune PM, Wagstaff M, Petros AJ. Cerebro-splanchnic oxygenation ratio (CSOR) using near infrared spectroscopy may be able to predict splanchnic ischaemia in neonates. Intensive Care Med. 2001;27:1401–7.
Gillam-Krakauer M, Cochran CM, Slaughter JC, Polavarapu S, McElroy SJ, Hernanz-Schulman M, et al. Correlation of abdominal rSO2 with superior mesenteric artery velocities in preterm infants. J Perinatol. 2013;33:609–12.
DeWitt AG, Charpie JR, Donohue JE, Yu S, Owens GE. Splanchnic near-infrared spectroscopy and risk of necrotizing enterocolitis after neonatal heart surgery. Pediatr Cardiol. 2014;35:1286–94.
Ravikumar C, Pandey R. Feeding infants with hypoxic ischemic encephalopathy during therapeutic hypothermia. J Perinatol. 2023;43:124–7.
Dave V, Brion LP, Campbell DE, Scheiner M, Raab C, Nafday SM. Splanchnic tissue oxygenation, but not brain tissue oxygenation, increases after feeds in stable preterm neonates tolerating full bolus orogastric feeding. J Perinatol. 2009;29:213–8.
Hosseini SM, Talaei-Khozani T, Sani M, Owrangi B. Differentiation of human breast-milk stem cells to neural stem cells and neurons. Neurol Res Int. 2014;2014:807896.
Kersin SG, Ozek E. Breast milk stem cells: are they magic bullets in neonatology? Turk Arch Pediatr. 2021;56:187–91.
Ninkina N, Kukharsky MS, Hewitt MV, Lysikova EA, Skuratovska LN, Deykin AV, et al. Stem cells in human breast milk. Hum Cell. 2019;32:223–30.
Damianos A, Sammour I. Barriers in translating stem cell therapies for neonatal diseases. Semin Perinatol. 2023;47:151731.
Dix LM, van Bel F, Baerts W, Lemmers PM. Comparing near-infrared spectroscopy devices and their sensors for monitoring regional cerebral oxygen saturation in the neonate. Pediatr Res. 2013;74:557–63.
Acknowledgements
We would like to acknowledge the support of the Neonatology faculty, fellows, residents, APPs, Nursing and Ancillary staff in the Neonatal Intensive Care Units at Hassenfeld Children’s and Bellevue Hospitals.
Author information
Authors and Affiliations
Contributions
TAN and EW developed the study concept and design, and were responsible for patient recruitment. TAN was responsible for data collection and analysis. TAN, EVW and SMB interpreted results. MC served as safety monitor as designated by the institutional review board. All authors contributed to manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was performed in accordance with the Declaration of Helsinki. Informed consent was sought for each infant assigned to the interventional arm. The study protocol and informed consent form were approved by our local institutional review boards prior to the study’s conductance (NYU Langone Health Office of Science and Research Institutional Review Board—ref #i22-00861, Bellevue Hospital Institutional Review Board—ref STUDY00003666). Need for consent was waived for the control group, as they were a retrospective sample.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nuzum, T.A., Bailey, S.M., Caprio, M. et al. A prospective study describing splanchnic NIRS and clinical outcomes in encephalopathic neonates receiving minimal enteral nutrition during therapeutic hypothermia. J Perinatol 45, 1087–1092 (2025). https://doi.org/10.1038/s41372-025-02270-9
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41372-025-02270-9