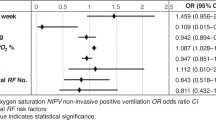
Abstract
Objective
To characterize racial disparities in retinopathy of prematurity (ROP) requiring treatment and to investigate specific treatment patterns across racial groups.
Study design
Retrospective analysis of de-identified data from ROP Check®, a cloud-based electronic medical record system for neonatology screening and ophthalmology tracking ROP examinations and treatment. Data from 28 hospitals from 2011 to 2022 included 3631 surviving premature infants with recorded race.
Results
American Indian/Alaska Native (AI/AN), Asian, and Pacific Islander (PI) infants had the highest rates of ROP treatment at both early (21–25 weeks) and later gestational ages (26–30 weeks) while African American infants had the lowest and most pronounced at later gestational ages (26–30 weeks). AI/AN infants had earlier and more multiple treatments.
Conclusions
Our study identifies PI race, alongside AI/AN and Asian backgrounds, as high-risk categories for treatment-warranted ROP. The inclusion of race to gestational age and birthweight enhances the prediction of treatment-warranted ROP.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
References
Wallace ME, Mendola P, Kim SS, Epps N, Chen Z, Smarr M, et al. Racial/ethnic differences in preterm perinatal outcomes. Am J Obstet Gynecol. 2017;216:306.e1–306.e12.
Janevic T, Zeitlin J, Auger N, Egorova NN, Hebert P, Balbierz A, et al. Association of race/ethnicity with very preterm neonatal morbidities. JAMA Pediatr. 2018;172:1061–9.
Ying GS, Bell EF, Donohue P, Tomlinson LA, Binenbaum G, G-ROP Research Group. Perinatal risk factors for the retinopathy of prematurity in postnatal growth and Rop Study. Ophthal Epidemiol. 2019;26:270–8.
Yang MB, Donovan EF, Wagge JR. Race, gender, and clinical risk index for babies (CRIB) score as predictors of severe retinopathy of prematurity. J AAPOS. 2006;10:253–61.
Cheng QE, Daniel E, Pan W, Baumritter A, Quinn GE, Ying GS, et al. Plus disease in telemedicine approaches to evaluating acute-phase ROP (e-ROP) study: characteristics, predictors, and accuracy of image grading. Ophthalmology. 2019;126:868–75.
Tadesse M, Dhanireddy R, Mittal M, Higgins RD. Race, Candida sepsis, and retinopathy of prematurity. Biol Neonate. 2002;81:86–90.
Wang J, Ying GS, Yu Y, Tomlinson L, Binenbaum G. Racial differences in retinopathy of prematurity. Ophthalmic Epidemiol. 2023;30:523–31.
Karmouta R, Altendahl M, Romero T, Piersante T, Langston S, Khitri M, et al. Association between social determinants of health and retinopathy of prematurity outcomes. JAMA Ophthalmol. 2022;140:496–502.
Lang DM, Blackledge J, Arnold RW. Is Pacific race a retinopathy of prematurity risk factor? Arch Pediatr Adolesc Med. 2005;159:771–3.
Arnold RW, Kesler K, Avila E. Susceptibility to retinopathy of prematurity in Alaskan Natives. J Pediatr Ophthalmol Strabismus. 1994;31:192–4.
Fierson WM.American Academy oF PEDIATRICS Section on Ophthalmology. et al. Screening examination of premature infants for retinopathy of prematurity. Pediatrics. 2018;142:1–9.
Arnold RW, Jacob J, Matrix Z. Toward achieving 100% adherence for retinopathy of prematurity screening guidelines. J Pediatr Ophthalmol Strabismus. 2017;54:356–62.
Brown DR, Biglan AW, Stretavsky MM. Retinopathy of prematurity: the relationship with intraventricular hemorrhage and bronchopulmonary dysplasia. J Pediatr Ophthalmol Strabismus. 1990;27:268–71.
Glaser K, Härtel C, Klingenberg C, Herting E, Fortmann MI, Speer CP, et al. Neonatal sepsis episodes and retinopathy of prematurity in very preterm infants. JAMA Netw Open. 2024;7:e2423933.
Fundora JB, Binenbaum G, Tomlinson L, Yu Y, Ying GS, Maheshwari A, et al. Association of surgical necrotizing enterocolitis and its timing with retinopathy of prematurity. Am J Perinatol. 2023;40:1178–84.
Arnaiz-Villena A, Parga-Lozano C, Moreno E, Areces C, Rey D, Gomez-Prieto P. The origin of Amerindians and the peopling of the Americas according to HLA genes: admixture with Asian and Pacific people. Curr Genom. 2010;11:103–14.
Anderson KG, Spicer P, Peercy MT. Obesity, diabetes, and birth outcomes among American Indians and Alaska Natives. Matern Child Health J. 2016;20:2548–56.
Yuan J, Gu X, Yang J, Lin X, Hu J, Jiang S, et al. Impact of maternal diabetes mellitus on neonatal outcomes among infants <32 weeks of gestation in China: a multicenter cohort study. Am J Perinatol. 2024;41:e2474–84.
Persson M, Shah PS, Rusconi F, Reichman B, Modi N, Kusuda S, et al. Association of maternal diabetes with neonatal outcomes of very preterm and very low-birth-weight infants: an international cohort study. JAMA Pediatr. 2018;172:867–75.
Opara CN, Akintorin M, Byrd A, Cirignani N, Akintorin S, Soyemi K. Maternal diabetes mellitus as an independent risk factor for clinically significant retinopathy of prematurity severity in neonates less than 1500g. PLoS ONE. 2020;15:e0236639.
Boghossian NS, Geraci M, Edwards EM, Horbar JD. In-hospital outcomes in large for gestational age infants at 22-29 weeks of gestation. J Pediatr. 2018;198:174–80.
Gimenez LG, Gili JA, Elias DE, Sagula R, Comas B, Santos MR, et al. Genetic susceptibility for retinopathy of prematurity and its associated comorbidities. Pediatr Res. 2024;96:1325–31.
Mohamed S, Schaa K, Cooper ME, Ahrens E, Alvarado A, Colaizy T, et al. Genetic contributions to the development of retinopathy of prematurity. Pediatr Res. 2009;65:193–7.
Paradis H, Werdyani S, Gendron RL, Gendron RL, Tabrizchi R, McGovern M, et al. Genetic variants of the beta-adrenergic receptor pathways as both risk and protective factors for retinopathy of prematurity. Am J Ophthalmol. 2024;263:179–87.
Li X, Owen LA, Taylor KD, Ostmo S, Chen YI, Coyner AS, et al. Genome-wide association identifies novel ROP risk loci in a multi-ethnic cohort. Commun Biol. 2024;7:107.
Author information
Authors and Affiliations
Contributions
Jack Jacob designed the work, collected data, drafted the manuscript, approves and agrees to be accountable for this work. Robert Arnold analyzed the data, critically reviewed the manuscript, and approves and agrees to be accountable for this work.
Corresponding author
Ethics declarations
Competing interests
JJ and RA developed ROP Check® software but no longer own it. They both serve as consultants for NeoLight.
Ethics approval and consent to participate
This study complies with the Declaration of Helsinki and HIPAA. IRB exemption (45 CFR § 46.104(d)(4),) was obtained for using the de-identified data set from WCG IRB on 3/4/24. Parental written consent was waived.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jacob, J., Arnold, R.W. The characteristics of racial disparity in retinopathy of prematurity outcomes. J Perinatol (2025). https://doi.org/10.1038/s41372-025-02355-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41372-025-02355-5